Abstract
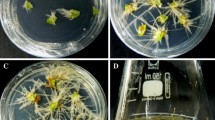
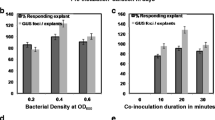
A rapid and efficient Agrobacterium rhizogenes mediated transformation system for Ocimum tenuiflorum L., a traditional Indian medicinal plant that occurs in red and green forma, was developed. The plant is a repertoire of several pharmaceutically and nutraceutically important metabolites. Three different types of explants i.e. leaves, hypocotyls and excised shoots, obtained from shoot cultures of in vitro germinated red and green forma plants were transformed using Agrobacterium rhizogenes strain ATCC 15834. The transformation efficiency was equal between similar explants of both forma. Transformation efficiency was best in leaves of 4 days while excised shoots and hypocotyls had 6 and 8 days respectively. Transformation frequency of green forma leaves was the highest (70.6%) among all explants. Excised shoots of green forma plants exhibited better transformation (58.3%) than the red forma excised shoots (42.59%). Red forma hypocotyl explants displayed marginally better (26.27%) transformation frequency than green hypocotyl explants (21.14%). Transformation with hairy root was confirmed by the presence of rolC gene through PCR amplification and Southern hybridization. The development of hairy root-based transgenic system for O. tenuiflorum will pave the way for in vitro production of important secondary metabolites.
Similar content being viewed by others
References
Aarrouf J., Castro-Quezada P., Mallard S., Caromel B., Lizzi Y. & Lefebvre V. 2012. Agrobacterium rhizogenes-dependent production of transformed roots from foliar explants of pepper (Capsicum annuum): a new and efficient tool for functional analysis of genes. Plant Cell Rep. 31: 391–401.
Bais H.P., Walker T.S., Schweizer H.P. & Vivanco J.M. 2002. Root specific elicitation and antimicrobial activity of rosmarinic acid in hairy root cultures of Ocimum basilicum. Plant Physiol. Biochem. 40: 983–995.
Bettini P., Baraldi R., Rapparini F., Melani L., Mauro M.L., Bindi D. & Buiatti M. 2010. The insertion of the Agrobacterium rhizogenes rolC gene in tomato (Solanum lycopersicum L.) affects plant architecture and endogenous auxin and abscisic acid levels. Sci. Hortic. 123: 323–328.
Chaudhuri K.N., Ghosh B., Tepfer D. & Jha S. 2005. Genetic transformation of Tylophora indica with Agrobacterium rhizogenes A4: growth and tylophorine productivity in different transformed root clones. Plant Cell Rep. 24: 25–35.
Danphitsanuparn P., Boonsnongcheep P., Boribonkaset T., Chintapakorn Y. & Prathanturarug S. 2012. Effect of Agrobacterium rhizogenes strains and other parameters on production of isoflavonoids in hairy roots of Pueraria candollei Grah. Ex Benth.Var. candollei. Plant Cell Tissue Organ Cult. 111: 315–322.
Fu C.X., Zhao D.X., Xue X.F., Jin Z.P., Ma F.S. 2005. Transformation of Saussurea involucrata by Agrobacterium rhizogenes: Hairy root induction and syringin production. Process Biochem. 40: 3789–3794.
Galvano F., Fauci L.L., Lazzarino G., Fogliano V., Ritieni A., Ciapellano S., Battistini N.C., Tavazzi B. & Galvano G. 2004. Cyanidins: metabolism and biological properties. J. Nut. Biochem. 15: 2–11.
Gangopadhyay M., Chakraborty D., Bhattacharyya S. & Bhattacharya S. 2010. Regeneration of transformed plants from hairy root of Plumbago indica. Plant Cell Tissue Organ Cult. 102: 109–114.
Gelvin S.B. 2000. Agrobacterium and plant genes involved in TDNA transfer and integration. Ann. Rev. Plant Physiol. Plant Mol. Biol. 51: 223–256.
Guillon S., Guiller J.T., Pati P.K., Rideau M. & Gantet P. 2006. Harnessing the potential of hairy roots: dawn of a new era. Trends Biotechnol. 24: 403–409.
Jirovetz L., Buchbauer G., Shafi M.P. & Kaniampady M.M. 2003. Chemotaxonomical analysis of the essential oils aroma compounds of four different Ocimum species from southern India. Eur. Food Res. Technol. 217: 120–124.
Marzouk A.M. 2009. Hepatoprotective triterpenes from hairy root cultures of Ocimum basilicum L. Z Naturforsch C. 64: 201–209.
Medina-Bolivar F., Condori J., Rimando A.M., Hubstenberger J., Shelton K., O’Keefe S.F., Bennett S. & Dolan M.C. 2007. Production and secretion of resveratrol in hairy root cultures of peanut. Phytochemistry 68: 1992–2003.
Murashige T. & Skoog F.A. 1962. Revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–497.
Nishiyama M. & Yamakawa T. 2004. Effect of medium composition on production of anthocyanins by hairy root cultures of Ipomoea batatas. Plant Biotechnol. 21: 411–414.
Ooi C.T., Syahida A., Stanslas J. & Maziah M. 2013. Efficiency of different Agrobacterium rhizogenes strains on hairy root induction in Solanum mammosum. World J. Microbiol. Biotechnol. 29: 421–430.
Paton A., Harley M.R. & Harley M.M. 2001. Ocimum: An overview of classification and relationships, pp. 1–38. In: Hiltunen R. & Holm Y. (eds), Basil: The Genus Ocimum, Harwood Academic Publisher, Amsterdam.
Renu I.K., Haque I., Kumar M., Poddar R., Bandopadhyay R., Rai A., Mukhopadhyay K. 2014. Characterization and functional analysis of Eugenol O-methyltransferase gene reveal metabolite shifts, chemotype specific differential expression and developmental regulation in Ocimum tenuiflorum L. Mol. Biol. Rep. doi: 10.1007/s11033-014-3035-7.
Satdive R.K., Fulzele D.P. & Eapen S. 2007. Enhanced production of azadirachtin by hairy root cultures of Azadirachta indicia. Juss by elicitation and media optimization. J. Biotechnol. 128: 281–289.
Sharafi A., Sohi H.H., Mousavi A., Azadi P., Razavi K. & Ntui V.O. 2013. A reliable and efficient protocol for inducing hairy roots in Papaver bracteatum. Plant Cell Tissue Organ Cult. 113: 1–9.
Sheikholeslam S.N. & Weeks D.P. 1987. Acetosyringone promotes high efficiency transformation of Arabidopsis thaliana explants by Agrobacterium tumefaciens. Plant Mol. Biol. 8: 291–298.
Simon J.E., Morales M.R., Phippen W.B., Vieira R.F. & Hao Z. 1999. Basil: A source of aroma compounds and a popular culinary and ornamental herb, pp. 499–505. In: Janick J. (ed.), Perspectives on New Crops and New Uses, ASHS Press, Alexandria, VA.
Sudha C.G., Sherina T.V., Anand V.P., Reji J.V., Padmesh P. & Sonia E.V. 2012. Agrobacterium rhizogenes mediated transformation of the medicinal plant Decalepis arayalpathra and production of 2-hydroxy-4-methoxy benzaldehyde. Plant Cell Tissue Organ Cult. 112: 217–226.
Tada H., Murakami Y., Omoto T., Shimomura K. & Ishimaru K. 1995. Rosamarinic acid and related phenolics in hairy root cultures of Ocimum basilicum. Pergamon 42: 431–434.
Vervliet G., Holsters M., Teuchy H., Van M.M. & Schell J. 1975. Characterization of different plaque-forming and defective temperate phages in Agrobacterium. J. Gen. Virol. 26: 33–48.
Viña A. & Murillo E. 2003. Essential oil composition from twelve varieties of Basil (Ocimum spp) grown in Colombia. J. Braz. Chem. Soc. 14: 744–749.
White F.F. & Nester E.W. 1980. Hairy root: Plasmid encodes virulence TrqKts in Agrobacterium rhizogenes. J. Bact. 141: 1134–1141.
WHO monographs on selected medicinal plants. 2002. Folium Ocimi Sancti 2: 206–216.
Wu J., Wang Y., Zhang L.X., Zhang X.Z., Kong J., Lu J. & Han Z.H. 2012. High-efficiency regeneration of Agrobacterium rhizogenes-induced hairy root in apple rootstock Malus baccata (L.) Borkh. Plant Cell Tissue Organ Cult. 111: 183–189.
Xu T.F., Zhang H.M. & Zhang W. 2000. Establishing better lines and regenerating system of hairy roots of Isatis indigotica with Ri T-DNA. Acad. J. Sec. Mil. Med. Univ. 21: 907–910.
Zhao B., Agblevor F.A., Ritesh K.C. & Jelesko J.G. 2013. Enhanced production of the alkaloid nicotine in hairy root cultures of Nicotiana tabacum L. Plant Cell Tissue Organ Cult. 113: 121–129.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vyas, P., Mukhopadhyay, K. Development of a rapid and high frequency Agrobacterium rhizogenes mediated transformation protocol for Ocimum tenuiflorum . Biologia 69, 765–770 (2014). https://doi.org/10.2478/s11756-014-0375-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-014-0375-7