Abstract
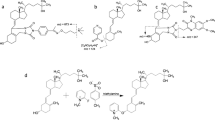
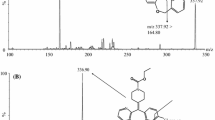
In this study, a rapid and reliable high-performance liquid chromatography–tandem mass spectrometry (HPLC-MS/MS) method for the determination of ambroxol in human plasma was developed and validated using palmatine as an internal standard (IS). Ambroxol and IS were extracted from 200 μL of human plasma via a simple protein precipitation preparation. Chromatographic separation was achieved on a Platisil C18 column (150 × 4.6 mm, 5 μm) using methanol–0.01% formic acid (70:30, v/v) as the mobile phase at a flow rate of 0.6 mL/min under an isocratic condition. The MS acquisition m/z 379 → 264 for ambroxol and 352 → 336 for IS was performed by atmospheric-pressure chemical ionization (APCI) mass spectrometry in selected reaction monitoring mode. The calibration curve for ambroxol was linear over the concentration range of 2.500–180.0 ng/mL. The matrix effects of ambroxol ranged from 104.6 to 112.7%. This fully validated method was successfully applied to a pharmacokinetic study of ambroxol in humans after oral administration of ambroxol at a single dose of 75 mg.
Similar content being viewed by others
References
G. H. Guyatt, M. Townsend, F. Kazim, and M. T. Newhouse, Chest, 1987, 92, 618.
J. L. M. Santos, A. Clausse, J. L. F. C. Lima, M. L. M. F. S. Saraiva, and A. O. S. Rangel, Anal. Sci., 2005, 27, 461.
M. Yamaya, H. Nishimura, L.K. Nadine, C. Ota, H. Kubo, and R. Nagatomi, Arch. Pharm. Res., 2014, 37, 520.
X. Wang, L. Wang, H. Wang, and H. Zhang, Cell Biochem. Biophys., 2015, 73, 281.
A. McNeill, J. Magalhaes, C. Shen, K. Y. Chau, D. Hughes, A. Mehta, T. Foltynie, J. M. Cooper, A. Y. Abramov, M. Gegg, and A. H. Schapira, Brain, 2014, 737, 1481.
T. J. Hang, M. Zhang, M. Song, J. P. Shen, and Y. D. Zhang, Clin. Chim. Acta, 2007, 382, 20.
F. Su, F. Wang, W. Gao, and H. Li, J. Chromatogr. B, 2007, 853, 364.
W. Hu, Y. Xu, F. Liu, A. Liu, and Q. Guo, Biomed. Chromatogr., 2008, 22, 1108.
A. Wen, T. Hang, S. Chen, Z. Wang, L. Ding, Y. Tian, M. Zhang, and X. Xu, J. Pharm. Biomed. Anal., 2008, 48, 829.
X. Dong, L. Ding, X. Cao, L. Jiang, and S. Zhong, Biomed. Chromatogr., 2013, 27, 520.
Z. Guo, Y. Chen, X. Ding, C. Huang, and L. Miao, Biomed. Chromatogr., 2016, 30, 1789.
E. Beltrán, M. Ibánez, J. V. Sancho, and F. Hernandez, Food Chem., 2014, 742, 400.
A. ter Halle, C. Claparols, J. C. Garrigues, S. Franceschi-Messant, and E. Perez, J. Chromatogr. A, 2015, 1414, 1.
L. Chen, F. Song, Z. Liu, Z. Zheng, J. Xing, and S. Liu, Anal. Bioanal. Chem., 2013, 406, 1481.
Food and Drug Administration, “Guidance for Industry: Bioanalytical Method Validation”, 2013, US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm368107.pdf, Accessed 16th Feb. 2015.
A. Van Eeckhaut, K. Lanckmans, S. Sarre, I. Smolders, and Y. Michotte, J. Chromatogr. B, 2009, 877, 2198.
A. C. Isaguirre, R. A. Olsina, L. D. Martinez, A. V. Lapierre, and S. Cerutti, Microchem. J., 2016, 129, 362.
J. Ding, G. Jin, G. Jin, A. Shen, Z. Guo, B. Yu, Y. Jiao, J. Yan, and X. Liang, Food Anal. Methods, 2016, 9, 2856.
J. Zander, B. Maier, A. Suhr, M. Zoller, L. Frey, D. Teupser, and M. Vogeser, Clin. Chem. Lab. Med., 2015, 53, 781.
N. Lindegardh, A. Annerberg, N. J. White, and N. P. Day, J. Chromatogr. B, 2008, 862, 227.
M. Tsuchiya, Anal. Sci., 1998, 14, 661.
O. A. Ismaiel, M. S. Halquist, M. Y. Elmamly, A. Shalaby, and H. Thomas Karnes, J. Chromatogr. B, 2008, 875, 333.
Acknowledgments
We thank Ms. Na Zhao and Ms. Yang Liu of Yiling Medical Technology Co., Ltd. for their technical help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mao, Z., Wang, X., Di, X. et al. Quantitative Detection of Ambroxol in Human Plasma Using HPLC-APCI-MS/MS: Application to a Pharmacokinetic Study. ANAL. SCI. 33, 1099–1103 (2017). https://doi.org/10.2116/analsci.33.1099
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.33.1099