Abstract
Background
Hypersensitivity reactions (HSR) to pegylated liposomal doxorubicin (PLD; Caelyx®) have been reported, and symptoms usually resolve with drug withdrawal. However, the risk of relapse of severe HSR and prevention remain poorly described.
Objectives
To report the management and outcome in four patients with HSR due to PLD.
Materials & Methods
Patient characteristics, premedication regimen, rate of infusion, time between onset and HSR, clinical manifestations, and management were documented.
Results
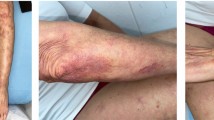
A first cycle of PLD was received for cutaneous T-cell lymphoma (n = 3) and Kaposi sarcoma (n = 1). The drug was diluted in 250 mL 5% glucose and administered over one hour (4.17 mL dilution/min, i.e. 0.6 mg PLD/min for 1.8m2 body surface area [BSA]). Grade 3 HSR occurred in the first minutes in the four patients. Because of the absence of alternative treatment for the underlying disease, PLD was resumed. Premedication was reinforced with 300 mg oral ranitidine and 50 mg hydroxyzine the night before and the morning of infusion. The rate of infusion was 1 mL dilution/min (0.14 mg PLD/min for 1.8 m2 BSA) for the first 15 minutes. No HSR occurred in three patients. In contrast, severe symptoms appeared in the first seconds of resumption in one patient.
Conclusion
To minimise HSR to PLD, an initial reduced rate of infusion of 0.1-0.2 mg of PLD/min is warranted. In the event of HSR, alternative therapy must be privileged, and if necessary, careful re-challenge with PLD may be attempted, however relapse of HSR may occur.
Similar content being viewed by others
References
Gabizon A, Shmeeda H, Barenholz Y. Pharmacokinetics of pegylatedliposomal Doxorubicin: review of animal and human studies. Clin Pharmacokinet 2003; 42: 419–36.
Coukell AJ, Spencer CM. Polyethylene glycol-liposomal doxorubicin. A review of its pharmacodynamic and pharmacokineticproperties, and therapeutic efficacy in the management of AIDS-relatedKaposi’s sarcoma. Drugs 1997; 53: 520–38.
Régnier-Rosencher E, Guillot B, Dupin N. Treatments for classicKaposi sarcoma: a systematic review of the literature. J Am AcadDermatol 2013; 68: 313–31.
Quereux G, Marques S, Nguyen JM, et al. Prospective multicenterstudy of pegylated liposomal doxorubicin treatment in patients withadvanced or refractory mycosis fungoides or Sézary syndrome. ArchDermatol 2008; 144: 727–33.
Dummer R, Quaglino P, Becker JC, et al. Prospective internationalmulticenter phase II trial of intravenous pegylated liposomal doxorubicinmonochemotherapy in patients with stage IIB, IVA, or IVBadvanced mycosis fungoides: final results from EORTC 21012. J ClinOncol 2012; 30: 4091–7.
Duggan ST, Keating GM. Pegylated liposomal doxorubicin: areview of its use in metastatic breast cancer, ovarian cancer, multiplemyeloma and AIDS-related Kaposi’s sarcoma. Drugs 2011; 71: 2531–58.
Chanan-Khan A, Szebeni J, Savay S, et al. Complement activationfollowing first exposure to pegylated liposomal doxorubicin (Doxil):possible role in hypersensitivity reactions. Ann Oncol 2003; 14: 1430–7.
Szebeni J, Muggia F, Gabizon A, Barenholz Y. Activation ofcomplement by therapeutic liposomes and other lipid excipient-basedtherapeutic products: prediction and prevention. Adv Drug Deliv Rev 2011; 63: 1020–30.
Kintzel PE. Prophylaxis for paclitaxel hypersensitivity reactions. AnnPharmacother 2001; 35: 1114–7.
Skubitz KM, Skubitz AP. Mechanism of transient dyspneainduced by pegylated-liposomal doxorubicin (Doxil). Anticancer Drugs 1998; 9: 45–50.
Szebeni J. Complement activation-related pseudoallergy: anew class of drug-induced acute immune toxicity. Toxicology 2005; 216: 106–21.
Decuyper II, Ebo DG, Uyttebroek AP, et al. Quantification ofspecific IgE antibodies in immediate drug hypersensitivity: more shortcomingsthan potentials? Clin Chim Acta 2016; 460: 184–9.
Limsuwan T, Castells MC. Outcomes and safety of rapid desensitizationfor chemotherapy hypersensitivity. Expert Opin Drug Saf 2010; 9: 39–53.
Castells MC, Tennant NM, Sloane DE, et al. Hypersensitivity reactionsto chemotherapy: outcomes and safety of rapid desensitization in413 cases. J Allergy Clin Immunol 2008; 122: 574–80.
Balsari A, Lombardo N, Ghione M. Skin and perivascular toxicityinduced experimentally by doxorubicin. J Chemother 1989; 1: 324–9.
Mangodt EA, Van Gasse AL, Bastiaensen A, et al. Flow-assistedbasophil activation tests in immediate drug hypersensitivity: twodecades of Antwerp experience. Acta Clin Belg 2016; 71: 19–25.
Piva E, Chieco-Bianchi F, Krajcar V, et al. Adverse reactions inpatients with B-cell lymphomas during combined treatment with rituximab:in vitro evaluation of rituximab hypersensitivity by basophilactivation test. Am J Hematol 2012; 87: E130–1.
Author information
Authors and Affiliations
Corresponding author
Additional information
To cite this article: Ingen-Housz-Oro S, Pham-Ledard A, Brice P, Lebrun-Vignes B, Zehou O, Reitter D, Ram-Wolff C, Dupin N, Bagot M, Chosidow O, Beylot-Barry M. Immediate hypersensitivity reaction to pegylated liposomal doxorubicin: management and outcome in four patients. Eur J Dermatol 2017; 27(3): 271-4 doi:10.1684/ejd.2017.2986
About this article
Cite this article
Ingen-Housz-Oro, S., Pham-Ledard, A., Brice, P. et al. Immediate hypersensitivity reaction to pegylated liposomal doxorubicin: management and outcome in four patients. Eur J Dermatol 27, 271–274 (2017). https://doi.org/10.1684/ejd.2017.2986
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2017.2986