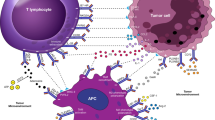
Abstract
Immunological evasion is one of the defining characteristics of cancers, as the immune modification of an immune checkpoint (IC) confers immune evasion capabilities to tumor cells. Multiple ICs, such as programmed cell death protein-1 (PD-1) and cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4), can bind to their respective receptors and reduce tumor immunity in a variety of ways, including blocking immune cell activation signals. IC blockade (ICB) therapies targeting these checkpoint molecules have demonstrated significant clinical benefits. This is because antibody-based IC inhibitors and a variety of specific small molecule inhibitors can inhibit key oncogenic signaling pathways and induce durable tumor remission in patients with a variety of cancers. Deciphering the roles and regulatory mechanisms of these IC molecules will provide crucial theoretical guidance for clinical treatment. In this review, we summarize the current knowledge on the functional and regulatory mechanisms of these IC molecules at multiple levels, including epigenetic regulation, transcriptional regulation, and post-translational modifications. In addition, we provide a summary of the medications targeting various nodes in the regulatory pathway, and highlight the potential of newly identified IC molecules, focusing on their potential implications for cancer diagnostics and immunotherapy.
概要
包括程序性细胞死亡蛋白1(PD-1)和细胞毒性T淋巴细胞相关抗原4(CTLA-4)在内的免疫检查点蛋白,可以与各自的受体结合,以阻断免疫细胞的激活信号等方式促进肿瘤细胞免疫逃逸。这种由免疫检查点的免疫修饰赋予的肿瘤细胞免疫规避能力,使得免疫逃逸成为肿瘤的重要特征之一。靶向上述分子的免疫检查点阻断(ICB)疗法通过抗体与特定小分子抑制剂抑制肿瘤关键信号通路,破解肿瘤患者的免疫耐受,已表现出优异的临床效益。因此,破译这些免疫检查点分子的作用和调节机制将为临床治疗提供关键的理论指导。在这篇综述中,我们总结了目前关于这些免疫检查点分子在多个层面上的功能和调节机制,包括表观遗传调节、转录调节和翻译后修饰。此外,我们还对针对调控途径中各个节点的药物进行了总结,进一步阐明新鉴定的免疫检查点分子的临床潜力,重点介绍了它们对癌症诊断和免疫治疗的潜在意义。
Similar content being viewed by others
References
Agata Y, Kawasaki A, Nishimura H, et al., 1996. Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int Immunol, 8(5):765–772. https://doi.org/10.1093/intimm/8.5.765
Ahn HK, Kim S, Kwon D, et al., 2019. MET receptor tyrosine kinase regulates the expression of co-stimulatory and co-inhibitory molecules in tumor cells and contributes to PD-L1-mediated suppression of immune cell function. Int J Mol Sci, 20(17):4287. https://doi.org/10.3390/ijms20174287
Akbay EA, Koyama S, Carretero J, et al., 2013. Activation of the PD-1 pathway contributes to immune escape in EGFR-driven lung tumors. Cancer Discov, 3(12): 1355–1363. https://doi.org/10.1158/2159-8290.Cd-13-0310
Albitar M, Sudarsanam S, Ma WL, et al., 2018. Correlation of MET gene amplification and TP53 mutation with PD-L1 expression in non-small cell lung cancer. Oncotarget, 9(17):13682–13693. https://doi.org/10.18632/oncotarget.24455
Alegre ML, Noel PJ, Eisfelder BJ, et al., 1996. Regulation of surface and intracellular expression of CTLA4 on mouse T cells. J Immunol, 157(11):4762–4770.
al Emran A, Chatterjee A, Rodger EJ, et al., 2019. Targeting DNA methylation and EZH2 activity to overcome melanoma resistance to immunotherapy. Trends Immunol, 40(4): 328–344. https://doi.org/10.1016/j.it.2019.02.004
Anastasiadou E, Stroopinsky D, Alimperti S, et al., 2019. Epstein-Barr virus-encoded EBNA2 alters immune checkpoint PD-L1 expression by downregulating miR-34a in B-cell lymphomas. Leukemia, 33(1):132–147. https://doi.org/10.1038/s41375-018-0178-x
Anderson AC, Joller N, Kuchroo VK, 2016. Lag-3, Tim-3, and TIGIT: co-inhibitory receptors with specialized functions in immune regulation. Immunity, 44(5):989–1004. https://doi.org/10.1016/j.immuni.2016.05.001
Ashizawa M, Okayama H, Ishigame T, et al., 2019. miRNA-148a-3p regulates immunosuppression in DNA mismatch repair-deficient colorectal cancer by targeting PD-L1. Mol Cancer Res, 17(6):1403–1413. https://doi.org/10.1158/1541-7786.Mcr-18-0831
Atsaves V, Tsesmetzis N, Chioureas D, et al., 2017. PD-L1 is commonly expressed and transcriptionally regulated by STAT3 and MYC in ALK-negative anaplastic large-cell lymphoma. Leukemia, 31(7):1633–1637. https://doi.org/10.1038/leu.2017.103
Baixeras E, Huard B, Miossec C, et al., 1992. Characterization of the lymphocyte activation gene 3-encoded protein. A new ligand for human leukocyte antigen class II antigens. J Exp Med, 176(2):327–337. https://doi.org/10.1084/jem.176.2.327
Barsoum IB, Smallwood CA, Siemens DR, et al., 2014. A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Cancer Res, 74(3):665–674. https://doi.org/10.1158/0008-5472.Can-13-0992
Baumeister SH, Freeman GJ, Dranoff G, et al., 2016. Coinhibitory pathways in immunotherapy for cancer. Annu Rev Immunol, 34:539–573. https://doi.org/10.1146/annurev-immunol-032414-112049
Borst J, Busselaar J, Bosma DMT, et al., 2021. Mechanism of action of PD-1 receptor/ligand targeted cancer immunotherapy. Eur J Immunol, 51(8):1911–1920. https://doi.org/10.1002/eji.202048994
Boussiotis VA, 2016. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med, 375(18): 1767–1778. https://doi.org/10.1056/NEJMra1514296
Bu LL, Yu GT, Wu L, et al., 2017. STAT3 induces immunosuppression by upregulating PD-1/PD-L1 in HNSCC. J Dent Res, 96(9):1027–1034. https://doi.org/10.1177/0022034517712435
Buisson S, Triebel F, 2005. LAG-3 (CD223) reduces macrophage and dendritic cell differentiation from monocyte precursors. Immunology, 114(3):369–374. https://doi.org/10.1111/j.1365-2567.2004.02087.x
Burr ML, Sparbier CE, Chan YC, et al., 2017. CMTM6 maintains the expression of PD-L1 and regulates anti-tumour immunity. Nature, 549(7670):101–105. https://doi.org/10.1038/nature23643
Butte MJ, Keir ME, Phamduy TB, et al., 2007. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity, 27(1):111–122. https://doi.org/10.1016/j.immuni.2007.05.016
Casey SC, Tong L, Li YL, et al., 2016. MYC regulates the antitumor immune response through CD47 and PD-L1. Science, 352(6282):227–231. https://doi.org/10.1126/science.aac9935
Castellanos JR, Purvis IJ, Labak CM, et al., 2017. B7-H3 role in the immune landscape of cancer. Am J Clin Exp Immunol, 6(4):66–75.
Ceeraz S, Nowak EC, Noelle RJ, 2013. B7 family checkpoint regulators in immune regulation and disease. Trends Immunol, 34(11):556–563. https://doi.org/10.1016/j.it.2013.07.003
Cerezo M, Guemiri R, Druillennec S, et al., 2018. Translational control of tumor immune escape via the eIF4F-STAT1-PD-L1 axis in melanoma. Nat Med, 24(12):1877–1886. https://doi.org/10.1038/s41591-018-0217-1
Cesano A, Warren S, 2018. Bringing the next generation of immuno-oncology biomarkers to the clinic. Biomedicines, 6:14. https://doi.org/10.3390/biomedicines6010014
Cha JH, Yang WH, Xia WY, et al., 2018. Metformin promotes antitumor immunity via endoplasmic-reticulum-associated degradation of PD-L1. Mol Cell, 71(4):606–620.E7. https://doi.org/10.1016/j.molcel.2018.07.030
Chan LC, Li CW, Xia WY, et al., 2019. IL-6/JAK1 pathway drives PD-L1 Y112 phosphorylation to promote cancer immune evasion. J Clin Invest, 129(8):3324–3338. https://doi.org/10.1172/jci126022
Chang LC, Chen TP, Kuo WK, et al., 2018. The protein expression of PDL1 is highly correlated with those of eIF2α and ATF4 in lung cancer. Dis Markers, 2018:5068701. https://doi.org/10.1155/2018/5068701
Chang RM, Xiao S, Lei X, et al., 2017. miRNA-487a promotes proliferation and metastasis in hepatocellular carcinoma. Clin Cancer Res, 23(10):2593–2604. https://doi.org/10.1158/1078-0432.Ccr-16-0851
Chapoval AI, Ni J, Lau JS, et al., 2001. B7-H3: a costimulatory molecule for T cell activation and IFN-γ production. Nat Immunol, 2(3):269–274. https://doi.org/10.1038/85339
Chauvin JM, Pagliano O, Fourcade J, et al., 2015. TIGIT and PD-1 impair tumor antigen-specific CD8+ T cells in melanoma patients. J Clin Invest, 125(5):2046–2058. https://doi.org/10.1172/jci80445
Chen BJ, Chapuy B, Ouyang J, et al., 2013. PD-L1 expression is characteristic of a subset of aggressive B-cell lymphomas and virus-associated malignancies. Clin Cancer Res, 19(13):3462–3473. https://doi.org/10.1158/1078-0432.Ccr-13-0855
Chen C, Liu JM, Luo YP, 2020, MicroRNAs in tumor immunity: functional regulation in tumor-associated macrophages. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(1):12–28. https://doi.org/10.1631/jzus.B1900452
Chen LM, Gibbons DL, Goswami S, et al., 2014. Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression. Nat Commun, 5:5241. https://doi.org/10.1038/ncomms6241
Chen MX, Pockaj B, Andreozzi M, et al., 2018. JAK2 and PD-L1 amplification enhance the dynamic expression of PD-L1 in triple-negative breast cancer. Clin Breast Cancer, 18(5):E1205–E1215. https://doi.org/10.1016/j.clbc.2018.05.006
Cheng HY, Borczuk A, Janakiram M, et al., 2018. Wide expression and significance of alternative immune checkpoint molecules, B7x and HHLA2, in PD-L1-negative human lung cancers. Clin Cancer Res, 24(8):1954–1964. https://doi.org/10.1158/1078-0432.Ccr-17-2924
Cheng PY, Eksioglu EA, Chen XH, et al., 2019. S100A9-induced overexpression of PD-1/PD-L1 contributes to ineffective hematopoiesis in myelodysplastic syndromes. Leukemia, 33(8):2034–2046. https://doi.org/10.1038/s41375-019-0397-9
Clarke JM, George DJ, Lisi S, et al., 2018. Immune checkpoint blockade: the new frontier in cancer treatment. Targ Oncol, 13(1):1–20. https://doi.org/10.1007/s11523-017-0549-7
Coelho MA, de Carné Trécesson S, Rana S, et al., 2017. Oncogenic RAS signaling promotes tumor immunoresistance by stabilizing PD-L1 mRNA. Immunity, 47(6):1083–1099.e6. https://doi.org/10.1016/j.immuni.2017.11.016
Dai XM, Bu X, Gao Y, et al., 2021. Energy status dictates PD-L1 protein abundance and anti-tumor immunity to enable checkpoint blockade. Mol Cell, 81(11):2317–2331.e6. https://doi.org/10.1016/j.molcel.2021.03.037
Dangaj D, Lanitis E, Zhao AZ, et al., 2013. Novel recombinant human B7-H4 antibodies overcome tumoral immune escape to potentiate T-cell antitumor responses. Cancer Res, 73(15):4820–4829. https://doi.org/10.1158/0008-5472.Can-12-3457
D’Arrigo P, Russo M, Rea A, et al., 2017. A regulatory role for the co-chaperone FKBP51s in PD-L1 expression in glioma. Oncotarget, 8(40):68291–68304. https://doi.org/10.18632/oncotarget.19309
Darvin P, Nair VS, Elkord E, 2019. PD-L1 expression in human breast cancer stem cells is epigenetically regulated through posttranslational histone modifications. J Oncol, 2019:3958908. https://doi.org/10.1155/2019/3958908
David JM, Dominguez C, Mccampbell KK, et al., 2017. A novel bifunctional anti-PD-L1/TGF-β trap fusion protein (M7824) efficiently reverts mesenchymalization of human lung cancer cells. Oncoimmunology, 6(10):e1349589. https://doi.org/10.1080/2162402x.2017.1349589
Demuth C, Andersen MN, Jakobsen KR, et al., 2017. Increased PD-L1 expression in erlotinib-resistant NSCLC cells with MET gene amplification is reversed upon MET-TKI treatment. Oncotarget, 8(40):68221–68229. https://doi.org/10.18632/oncotarget.19920
de Vos L, Grünwald I, Bawden EG, et al., 2020. The landscape of CD28, CD80, CD86, CTLA4, and ICOS DNA methylation in head and neck squamous cell carcinomas. Epigenetics, 15(11):1195–1212. https://doi.org/10.1080/15592294.2020.1754675
Dorand RD, Nthale J, Myers JT, et al., 2016. Cdk5 disruption attenuates tumor PD-L1 expression and promotes antitumor immunity. Science, 353(6297):399–403. https://doi.org/10.1126/science.aae0477
Du LY, Lee JH, Jiang HF, et al., 2020. β-Catenin induces transcriptional expression of PD-L1 to promote glioblastoma immune evasion. J Exp Med, 217(11):e20191115. https://doi.org/10.1084/jem.20191115
Emaldi M, Nunes-Xavier CE, 2022. B7-H4 immune checkpoint protein affects viability and targeted therapy of renal cancer cells. Cells, 11(9):1448. https://doi.org/10.3390/cells11091448
Fiedler M, Schulz D, Piendl G, et al., 2020. Buparlisib modulates PD-L1 expression in head and neck squamous cell carcinoma cell lines. Exp Cell Res, 396(1):112259. https://doi.org/10.1016/j.yexcr.2020.112259
Flem-Karlsen K, Fodstad Ø, Nunes-Xavier CE, 2020. B7-H3 immune checkpoint protein in human cancer. Curr Med Chem, 27(24):4062–4086. https://doi.org/10.2174/0929867326666190517115515
Flies DB, Wang SD, Xu HY, et al., 2011. Cutting edge: a monoclonal antibody specific for the programmed death-1 homolog prevents graft-versus-host disease in mouse models. J Immunol, 187(4):1537–1541. https://doi.org/10.4049/jimmunol.1100660
Franzen A, Vogt TJ, Müller T, et al., 2018. PD-L1 (CD274) and PD-L2 (PDCD1LG2) promoter methylation is associated with HPV infection and transcriptional repression in head and neck squamous cell carcinomas. Oncotarget, 9(1):641–650. https://doi.org/10.18632/oncotarget.23080
Fuhrman CA, Yeh WI, Seay HR, et al., 2015. Divergent phenotypes of human regulatory T cells expressing the receptors TIGIT and CD226. J Immunol, 195(1):145–155. https://doi.org/10.4049/jimmunol.1402381
Gainor JF, Shaw AT, Sequist LV, et al., 2016. EGFR mutations and ALK rearrangements are associated with low response rates to PD-1 pathway blockade in non-small cell lung cancer: a retrospective analysis. Clin Cancer Res, 22(18):4585–4593. https://doi.org/10.1158/1078-0432.Ccr-15-3101
Gao L, Xia LL, Ji WX, et al., 2021. Knockdown of CDK5 down-regulates PD-L1 via the ubiquitination-proteasome pathway and improves antitumor immunity in lung adenocarcinoma. Transl Oncol, 14(9):101148. https://doi.org/10.1016/j.tranon.2021.101148
Gao Y, Nihira NT, Bu X, et al., 2020. Acetylation-dependent regulation of PD-L1 nuclear translocation dictates the efficacy of anti-PD-1 immunotherapy. Nat Cell Biol, 22(9):1064–1075. https://doi.org/10.1038/s41556-020-0562-4
Garcia-Diaz A, Shin DS, Moreno BH, et al., 2017. Interferon receptor signaling pathways regulating PD-L1 and PD-L2 expression. Cell Rep, 19(6):1189–1201. https://doi.org/10.1016/j.celrep.2017.04.031
Gibson HM, Hedgcock CJ, Aufiero BM, et al., 2007. Induction of the CTLA-4 gene in human lymphocytes is dependent on NFAT binding the proximal promoter. J Immunol 179(6):3831–3840. https://doi.org/10.4049/jimmunol.179.6.3831
Goltz D, Gevensleben H, Dietrich J, et al., 2017. PD-L1 (CD274) promoter methylation predicts survival in colorectal cancer patients. Oncoimmunology, 6(1):e1257454. https://doi.org/10.1080/2162402x.2016.1257454
Goltz D, Gevensleben H, Vogt TJ, et al., 2018. CTLA4 methylation predicts response to anti-PD-1 and anti-CTLA-4 immunotherapy in melanoma patients. JCI Insight, 3(13): e96793. https://doi.org/10.1172/jci.insight.96793
Green MR, Rodig S, Juszczynski P, et al., 2012. Constitutive AP-1 activity and EBV infection induce PD-L1 in Hodgkin lymphomas and posttransplant lymphoproliferative disorders: implications for targeted therapy. Clin Cancer Res, 18(6):1611–1618. https://doi.org/10.1158/1078-0432.Ccr-11-1942
Greenwald RJ, Freeman GJ, Sharpe AH, 2005. The B7 family revisited. Annu Rev Immunol, 23:515–548. https://doi.org/10.1146/annurev.immunol.23.021704.115611
Gros A, Robbins PF, Yao X, et al., 2014. PD-1 identifies the patient-specific CD8+ tumor-reactive repertoire infiltrating human tumors. J Clin Invest, 124(5):2246–2259. https://doi.org/10.1172/JCI73639
Guo W, Tan FW, Huai QL, et al., 2021. Comprehensive analysis of PD-L1 expression, immune infiltrates, and m6A RNA methylation regulators in esophageal squamous cell carcinoma. Front Immunol, 12:669750. https://doi.org/10.3389/fimmu.2021.669750
Harjunpää H, Guillerey C, 2020. TIGIT as an emerging immune checkpoint. Clin Exp Immunol, 200(2):108–119. https://doi.org/10.1111/cei.13407
Hegde PS, Chen DS, 2020. Top 10 challenges in cancer immunotherapy. Immunity, 52(1):17–35. https://doi.org/10.1016/j.immuni.2019.12.011
Hou JW, Zhao RC, Xia WY, et al., 2020. PD-L1-mediated gasdermin C expression switches apoptosis to pyroptosis in cancer cells and facilitates tumour necrosis. Nat Cell Biol, 22(10):1264–1275. https://doi.org/10.1038/s41556-020-0575-z
Howe FS, Fischl H, Murray SC, et al., 2017. Is H3K4me3 instructive for transcription activation? BioEssays, 39(1):1–12. https://doi.org/10.1002/bies.201600095
Hsu JM, Xia WY, Hsu YH, et al., 2018. STT3-dependent PD-L1 accumulation on cancer stem cells promotes immune evasion. Nat Commun, 9:1908. https://doi.org/10.1038/s41467-018-04313-6
Huang KCY, Chiang SF, Chen WTL, et al., 2020. Decitabine augments chemotherapy-induced PD-L1 upregulation for PD-L1 blockade in colorectal cancer. Cancers, 12(2):462. https://doi.org/10.3390/cancers12020462
Huang SF, Bai XF, Fang TY, et al., 2021. Gastrointestinal toxicities associated with immune checkpoint inhibitors: a disproportionality analysis leveraging VigiBase, the WHO Adverse Drug Reaction Database. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 22(2):156–164. https://doi.org/10.1631/jzus.B2000449
Huard B, Tournier M, Hercend T, et al., 1994. Lymphocyte-activation gene 3/major histocompatibility complex class II interaction modulates the antigenic response of CD4+ T-lymphocytes. Eur J Immunol, 24(12):3216–3221. https://doi.org/10.1002/eji.1830241246
Ikeda S, Okamoto T, Okano S, et al., 2016. PD-L1 is upregulated by simultaneous amplification of the PD-L1 and JAK2 genes in non-small cell lung cancer. J Thorac Oncol, 11(1):62–71. https://doi.org/10.1016/j.jtho.2015.09.010
Iñarrairaegui M, Melero I, Sangro B, 2018. Immunotherapy of hepatocellular carcinoma: facts and hopes. Clin Cancer Res, 24(7):1518–1524. https://doi.org/10.1158/1078-0432.Ccr-17-0289
Janakiram M, Chinai JM, Fineberg S, et al., 2015. Expression, clinical significance, and receptor identification of the newest B7 family member HHLA2 protein. Clin Cancer Res, 21(10):2359–2366. https://doi.org/10.1158/1078-0432.Ccr-14-1495
Jebbawi F, Fayyad-Kazan H, Merimi M, et al., 2014. A microRNA profile of human CD8+ regulatory T cells and characterization of the effects of microRNAs on Treg cell-associated genes. J Transl Med, 12:218. https://doi.org/10.1186/s12967-014-0218-x
Jia L, Xi Q, Wang HF, et al., 2017. miR-142-5p regulates tumor cell PD-L1 expression and enhances anti-tumor immunity. Biochem Biophys Res Commun, 488(2):425–431. https://doi.org/10.1016/j.bbrc.2017.05.074
Jiang XF, Zhou J, Giobbie-Hurder A, et al., 2013. The activation of MAPK in melanoma cells resistant to BRAF inhibition promotes PD-L1 expression that is reversible by MEK and PI3K inhibition. Clin Cancer Res, 19(3):598–609. https://doi.org/10.1158/1078-0432.Ccr-12-2731
Jiang XG, 2020. Lymphatic vasculature in tumor metastasis and immunobiology. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(1):3–11. https://doi.org/10.1631/jzus.B1800633
Jiao SP, Xia WY, Yamaguchi H, et al., 2017. PARP inhibitor upregulates PD-L1 expression and enhances cancer-associated immunosuppression. Clin Cancer Res, 23(14): 3711–3720. https://doi.org/10.1158/1078-0432.Ccr-16-3215
Johnston RJ, Comps-Agrar L, Hackney J, et al., 2014. The immunoreceptor TIGIT regulates antitumor and antiviral CD8+ T cell effector function. Cancer Cell, 26(6):923–937. https://doi.org/10.1016/j.ccell.2014.10.018
Kim EY, Kim A, Kim SK, et al., 2017. MYC expression correlates with PD-L1 expression in non-small cell lung cancer. Lung Cancer, 110:63–67. https://doi.org/10.1016/j.lungcan.2017.06.006
Klein O, Kee D, Markman B, et al., 2021. Evaluation of TMB as a predictive biomarker in patients with solid cancers treated with anti-PD-1/CTLA-4 combination immunotherapy. Cancer Cell, 39(5):592–593. https://doi.org/10.1016/j.ccell.2021.04.005
Koh J, Jang JY, Keam B, et al., 2016. EML4-ALK enhances programmed cell death-ligand 1 expression in pulmonary adenocarcinoma via hypoxia-inducible factor (HIF)-1α and STAT3. Oncoimmunology, 5(3):e1108514. https://doi.org/10.1080/2162402x.2015.1108514
Korman AJ, Peggs KS, Allison JP, 2006. Checkpoint blockade in cancer immunotherapy. Adv Immunol, 90: 297–339.
Kouo T, Huang LQ, Pucsek AB, et al., 2015. Galectin-3 shapes antitumor immune responses by suppressing CD8 T cells via LAG-3 and inhibiting expansion of plasmacytoid dendritic cells. Cancer Immunol Res, 3(4):412–423. https://doi.org/10.1158/2326-6066.Cir-14-0150
Kourepini E, Paschalidis N, Simoes DCM, et al., 2016. TIGIT enhances antigen-specific Th2 recall responses and allergic disease. J Immunol, 196(9):3570–3580. https://doi.org/10.4049/jimmunol.1501591
Kurtulus S, Sakuishi K, Ngiow SF, et al., 2015. TIGIT predominantly regulates the immune response via regulatory T cells. J Clin Invest, 125(11):4053–4062. https://doi.org/10.1172/jci81187
Lamano JB, Lamano JB, Li YD, et al., 2019. Glioblastoma-derived IL6 induces immunosuppressive peripheral myeloid cell PD-L1 and promotes tumor growth. Clin Cancer Res, 25(12):3643–3657. https://doi.org/10.1158/1078-0432.Ccr-18-2402
Lastwika KJ, Wilson III W, Li QK, et al., 2016. Control of PD-L1 expression by oncogenic activation of the AKT-mTOR pathway in non-small cell lung cancer. Cancer Res, 76(2):227–238. https://doi.org/10.1158/0008-5472.Can-14-3362
Lee YH, Martin-Orozco N, Zheng PL, et al., 2017. Inhibition of the B7-H3 immune checkpoint limits tumor growth by enhancing cytotoxic lymphocyte function. Cell Res, 27(8):1034–1045. https://doi.org/10.1038/cr.2017.90
Leitner J, Klauser C, Pickl WF, et al., 2009. B7-H3 is a potent inhibitor of human T-cell activation: no evidence for B7-H3 and TREML2 interaction. Eur J Immunol, 39(7): 1754–1764. https://doi.org/10.1002/eji.200839028
Leong SR, Liang WC, Wu Y, et al., 2015. An anti-B7-H4 antibody-drug conjugate for the treatment of breast cancer. Mol Pharm, 12(6):1717–1729. https://doi.org/10.1021/mp5007745
Li CW, Lim SO, Xia WY, et al., 2016. Glycosylation and stabilization of programmed death ligand-1 suppresses T-cell activity. Nat Commun, 7:12632. https://doi.org/10.1038/ncomms12632
Li CW, Lim SO, Chung EM, et al., 2018. Eradication of triple-negative breast cancer cells by targeting glycosylated PD-L1. Cancer Cell, 33(2):187–201.e10. https://doi.org/10.1016/j.ccell.2018.01.009
Li FY, Zhao X, Zhang YL, et al., 2021. TFH cells depend on Tcf1-intrinsic HDAC activity to suppress CTLA4 and guard B-cell help function. Proc Natl Acad Sci USA, 118(2): e2014562118. https://doi.org/10.1073/pnas.2014562118
Li H, Li CW, Li XQ, et al., 2019. Met inhibitors promote liver tumor evasion of the immune response by stabilizing PDL1. Gastroenterology, 156(6): 1849–1861.e13. https://doi.org/10.1053/j.gastro.2019.01.252
Li M, Xia PY, Du Y, et al., 2014. T-cell immunoglobulin and ITIM domain (TIGIT) receptor/poliovirus receptor (PVR) ligand engagement suppresses interferon-γ production of natural killer cells via β-arrestin 2-mediated negative signaling. J Biol Chem, 289(25):17647–17657. https://doi.org/10.1074/jbc.M114.572420
Liang MQ, Yu FQ, Chen C, 2020. C-Myc regulates PD-L1 expression in esophageal squamous cell carcinoma. Am J Transl Res, 12(2):379–388.
Lim SO, Li CW, Xia WY, et al., 2016. Deubiquitination and stabilization of PD-L1 by CSN5. Cancer Cell, 30(6):925–939. https://doi.org/10.1016/j.ccell.2016.10.010
Lim TS, Chew V, Sieow JL, et al., 2016. PD-1 expression on dendritic cells suppresses CD8+ T cell function and antitumor immunity. Oncoimmunology, 5(3):e1085146. https://doi.org/10.1080/2162402x.2015.1085146
Linsley PS, Bradshaw J, Greene J, et al., 1996. Intracellular trafficking of CTLA-4 and focal localization towards sites of TCR engagement. Immunity, 4(6):535–543. https://doi.org/10.1016/s1074-7613(00)80480-x
Liu J, Yuan Y, Chen WN, et al., 2015. Immune-checkpoint proteins VISTA and PD-1 nonredundantly regulate murine T-cell responses. Proc Natl Acad Sci USA, 112(21):6682–6687. https://doi.org/10.1073/pnas.1420370112
Lu CW, Paschall AV, Shi HD, et al., 2017. The MLL1-H3K4me3 axis-mediated PD-L1 expression and pancreatic cancer immune evasion. J Natl Cancer Inst, 109(6): djw283. https://doi.org/10.1093/jnci/djw283
Luan XY, Yu WZ, Cao QZ, et al., 2009. The effects of B7H4 on human bone marrow mesenchymal stem cell inhibiting proliferation of PHA activated T cells. Chin J Hematol, 30(10):689–693 (in Chinese). https://doi.org/10.3760/cma.j.issn.0253-2727.2009.10.013
Maher CM, Thomas JD, Haas DA, et al., 2018. Small-molecule Sigma1 modulator induces autophagic degradation of PD-L1. Mol Cancer Res, 16(2):243–255. https://doi.org/10.1158/1541-7786.Mcr-17-0166
Mahnke K, Ring S, Johnson TS, et al., 2007. Induction of immunosuppressive functions of dendritic cells in vivo by CD4+CD25+ regulatory T cells: role of B7-H3 expression and antigen presentation. Eur J Immunol, 37(8):2117–2126. https://doi.org/10.1002/eji.200636841
Mao XB, Ou MT, Karuppagounder SS, et al., 2016. Pathological α-synuclein transmission initiated by binding lymphocyte-activation gene 3. Science, 353(6307):eaah3374. https://doi.org/10.1126/science.aah3374
Marwitz S, Scheufele S, Perner S, et al., 2017. Epigenetic modifications of the immune-checkpoint genes CTLA4 and PDCD1 in non-small cell lung cancer results in increased expression. Clin Epigenetics, 9:51. https://doi.org/10.1186/s13148-017-0354-2
Marzec M, Zhang Q, Goradia A, et al., 2008. Oncogenic kinase NPM/ALK induces through STAT3 expression of immunosuppressive protein CD274 (PD-L1, B7-H1). Proc Natl Acad Sci USA, 105(52):20852–20857. https://doi.org/10.1073/pnas.0810958105
Mead KI, Zheng Y, Manzotti CN, et al., 2005. Exocytosis of CTLA-4 is dependent on phospholipase D and ADP ribosylation factor-1 and stimulated during activation of regulatory T cells. J Immunol, 174(8):4803–4811. https://doi.org/10.4049/jimmunol.174.8.4803
Mezzadra R, Sun C, Jae LT, et al., 2017. Identification of CMTM6 and CMTM4 as PD-L1 protein regulators. Nature, 549(7670):106–110. https://doi.org/10.1038/nature23669
Micevic G, Thakral D, Mcgeary M, et al., 2019. PD-L1 methylation regulates PD-L1 expression and is associated with melanoma survival. Pigment Cell Melanoma Res, 32(3): 435–440. https://doi.org/10.1111/pcmr.12745
Mittendorf EA, Philips AV, Meric-Bernstam F, et al., 2014. PD-L1 expression in triple-negative breast cancer. Cancer Immunol Res, 2(4):361–370. https://doi.org/10.1158/2326-6066.Cir-13-0127
Nagaraju K, Raben N, Villalba ML, et al., 1999. Costimulatory markers in muscle of patients with idiopathic inflammatory myopathies and in cultured muscle cells. Clin Immunol, 92(2):161–169. https://doi.org/10.1006/clim.1999.4743
Nguyen LT, Ohashi PS, 2015. Clinical blockade of PD1 and LAG3-potential mechanisms of action. Nat Rev Immunol, 15(1):45–56. https://doi.org/10.1038/nri3790
Ni L, Dong C, 2017. New checkpoints in cancer immunotherapy. Immunol Rev, 276(1):52–65. https://doi.org/10.1111/imr.12524
Ni XY, Sui HX, Liu Y, et al., 2012. TGF-β of lung cancer microenvironment upregulates B7H1 and GITRL expression in dendritic cells and is associated with regulatory T cell generation. Oncol Rep, 28(2):615–621. https://doi.org/10.3892/or.2012.1822
Noguchi T, Ward JP, Gubin MM, et al., 2017. Temporally distinct PD-L1 expression by tumor and host cells contributes to immune escape. Cancer Immunol Res, 5(2):106–117. https://doi.org/10.1158/2326-6066.Cir-16-0391
Norris S, Coleman A, Kuri-Cervantes L, et al., 2012. PD-1 expression on natural killer cells and CD8+ T cells during chronic HIV-1 infection. Viral Immunol, 25(4):329–332. https://doi.org/10.1089/vim.2011.0096
Ota K, Azuma K, Kawahara A, et al., 2015. Induction of PD-L1 expression by the EML4-ALK oncoprotein and downstream signaling pathways in non-small cell lung cancer. Clin Cancer Res, 21(17):4014–4021. https://doi.org/10.1158/1078-0432.Ccr-15-0016
Ou DW, Wang XJ, Metzger DL, et al., 2006. Suppression of human T-cell responses to β-cells by activation of B7-H4 pathway. Cell Transplant, 15(5):399–410. https://doi.org/10.3727/000000006783981837
Ou JN, Wiedeman AE, Stevens AM, 2012. TNF-α and TGF-β counter-regulate PD-L1 expression on monocytes in systemic lupus erythematosus. Sci Rep, 2:295. https://doi.org/10.1038/srep00295
Oyewole-Said D, Konduri V, Vazquez-Perez J, et al., 2020. Beyond T-cells: functional characterization of CTLA-4 expression in immune and non-immune cell types. Front Immunol, 11:608024. https://doi.org/10.3389/fimmu.2020.608024
Paiva P, Lockhart MG, Girling JE, et al., 2016. Identification of genes differentially expressed in menstrual breakdown and repair. Mol Hum Reprod, 22(12):898–912. https://doi.org/10.1093/molehr/gaw060
Pan Y, Fei QL, Xiong P, et al., 2019. Synergistic inhibition of pancreatic cancer with anti-PD-L1 and c-Myc inhibitor JQ1. Oncoimmunology, 8(5):e1581529. https://doi.org/10.1080/2162402x.2019.1581529
Pandey P, Khan F, Qari HA, et al., 2022. Revolutionization in cancer therapeutics via targeting major immune checkpoints PD-1, PD-L1 and CTLA-4. Pharmaceuticals, 15(3): 335. https://doi.org/10.3390/ph15030335
Pardoll DM, 2012. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer, 12(4):252–264. https://doi.org/10.1038/nrc3239
Peguero JA, Bajaj P, Carcereny E, et al., 2019. A multicenter, phase II study of soluble LAG-3 (Eftilagimod alpha) in combination with pembrolizumab (TACTI-002) in patients with advanced non-small cell lung cancer (NSCLC) or head and neck squamous cell carcinoma (HNSCC). J Clin Oncol, 37(S15):TPS2667. https://doi.org/10.1200/JCO.2019.37.15_suppl.TPS2667
Peng SL, Wang R, Zhang XJ, et al., 2019. EGFR-TKI resistance promotes immune escape in lung cancer via increased PD-L1 expression. Mol Cancer, 18:165. https://doi.org/10.1186/s12943-019-1073-4
Petroni G, Formenti SC, Chen-Kiang S, et al., 2020. Immunomodulation by anticancer cell cycle inhibitors. Nat Rev Immunol, 20(11):669–679. https://doi.org/10.1038/s41577-020-0300-y
Podlesnykh SV, Abramova KE, Gordeeva A, et al., 2021. Peptide blocking CTLA-4 and B7-1 interaction. Molecules, 26(2):253. https://doi.org/10.3390/molecules26020253
Podojil JR, Miller SD, 2017. Potential targeting of B7-H4 for the treatment of cancer. Immunol Rev, 276(1):40–51. https://doi.org/10.1111/imr.12530
Qiu XY, Yang S, Wang S, et al., 2021. M6A demethylase ALKBH5 regulates PD-L1 expression and tumor immunoenvironment in intrahepatic cholangiocarcinoma. Cancer Res, 81(18):4778–4793. https://doi.org/10.1158/0008-5472.Can-21-0468
Qu FJ, Ye JQ, Pan XW, et al., 2019. MicroRNA-497-5p down-regulation increases PD-L1 expression in clear cell renal cell carcinoma. J Drug Target, 27(1):67–74. https://doi.org/10.1080/1061186x.2018.1479755
Quandt D, Jasinski-Bergner S, Müller U, et al., 2014. Synergistic effects of IL-4 and TNFα on the induction of B7-H1 in renal cell carcinoma cells inhibiting allogeneic T cell proliferation. J Transl Med, 12:151. https://doi.org/10.1186/1479-5876-12-151
Radichev IA, Maneva-Radicheva LV, Amatya C, et al., 2016. Loss of peripheral protection in pancreatic islets by proteolysis-driven impairment of VTCN1 (B7-H4) presentation is associated with the development of autoimmune diabetes. J Immunol, 194(4):1495–1506. https://doi.org/10.4049/jimmunol.1403251
Robert C, 2020. A decade of immune-checkpoint inhibitors in cancer therapy. Nat Commun, 11:3801. https://doi.org/10.1038/s41467-020-17670-y
Rotte A, 2019. Combination of CTLA-4 and PD-1 blockers for treatment of cancer. J Exp Clin Cancer Res, 38:255. https://doi.org/10.1186/s13046-019-1259-z
Rowshanravan B, Halliday N, Sansom DM, 2018. CTLA-4: a moving target in immunotherapy. Blood, 131(1):58–67. https://doi.org/10.1182/blood-2017-06-741033
Ruan ZY, Liang MH, Lai MX, et al., 2020. KYA1797K down-regulates PD-L1 in colon cancer stem cells to block immune evasion by suppressing the β-catenin/STT3 signaling pathway. Int Immunopharmacol, 78:106003. https://doi.org/10.1016/j.intimp.2019.106003
Schneider H, Martin M, Agarraberes FA, et al., 1999. Cytolytic T lymphocyte-associated antigen-4 and the TCRζ/CD3 complex, but not CD28, interact with clathrin adaptor complexes AP-1 and AP-2. J Immunol, 163(4):1868–1879.
Schreiber RD, Old LJ, Smyth MJ, 2011. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science, 331(6024):1565–1570. https://doi.org/10.1126/science.1203486
Shao LL, Hou WZ, Scharping NE, et al., 2019. IRF1 inhibits antitumor immunity through the upregulation of PD-L1 in the tumor cell. Cancer Immunol Res, 7(8):1258–1266. https://doi.org/10.1158/2326-6066.Cir-18-0711
Sharon E, Streicher H, Goncalves P, et al., 2014. Immune checkpoints in cancer clinical trials. Chin J Cancer, 33(9): 434–444. https://doi.org/10.5732/cjc.014.10122
Shen DD, Pang JR, Bi YP, et al., 2022. LSD1 deletion decreases exosomal PD-L1 and restores T-cell response in gastric cancer. Mol Cancer, 21:75. https://doi.org/10.1186/s12943-022-01557-1
Shen J, Li SY, Medeiros LJ, et al., 2020. PD-L1 expression is associated with ALK positivity and STAT3 activation, but not outcome in patients with systemic anaplastic large cell lymphoma. Mod Pathol, 33(3):324–333. https://doi.org/10.1038/s41379-019-0336-3
Shen MJ, Xu LJ, Yang L, et al., 2017. Radiation alters PD-L1/NKG2D ligand levels in lung cancer cells and leads to immune escape from NK cell cytotoxicity via IL-6-MEK/Erk signaling pathway. Oncotarget, 8(46):80506–80520. https://doi.org/10.18632/oncotarget.19193
Shi HB, Ji M, Wu J, et al., 2014. Serum B7-H4 expression is a significant prognostic indicator for patients with gastric cancer. World J Surg Oncol, 12:188. https://doi.org/10.1186/1477-7819-12-188
Shin DS, Zaretsky JM, Escuin-Ordinas H, et al., 2017. Primary resistance to PD-1 blockade mediated by JAK1/2 mutations. Cancer Discov, 7(2):188–201. https://doi.org/10.1158/2159-8290.Cd-16-1223
Shiratori T, Miyatake S, Ohno H, et al., 1997. Tyrosine phosphorylation controls internalization of CTLA-4 by regulating its interaction with clathrin-associated adaptor complex AP-2. Immunity, 6(5):583–589. https://doi.org/10.1016/s1074-7613(00)80346-5
Sica GL, Choi IH, Zhu GF, et al., 2003. B7-H4, a molecule of the B7 family, negatively regulates T cell immunity. Immunity, 18(6):849–861. https://doi.org/10.1016/s1074-7613(03)00152-3
Skafi N, Fayyad-Kazan M, Badran B, 2020. Immunomodulatory role for microRNAs: regulation of PD-1/PD-L1 and CTLA-4 immune checkpoints expression. Gene, 754: 144888. https://doi.org/10.1016/j.gene.2020.144888
Starke A, Wüthrich RP, Waeckerle-Men Y, 2007. TGF-beta treatment modulates PD-L1 and CD40 expression in proximal renal tubular epithelial cells and enhances CD8+ cytotoxic T-cell responses. Nephron Exp Nephrol, 107: E22–E29. https://doi.org/10.1159/000106506
Stutvoet TS, Kol A, de Vries EGE, et al., 2019. MAPK pathway activity plays a key role in PD-L1 expression of lung adenocarcinoma cells. J Pathol, 249(1):52–64. https://doi.org/10.1002/path.5280
Suh WK, Gajewska BU, Okada H, et al., 2003. The B7 family member B7-H3 preferentially down-regulates T helper type 1-mediated immune responses. Nat Immunol, 4(9): 899–906. https://doi.org/10.1038/ni967
Sun C, Lan PX, Han QJ, et al., 2018. Oncofetal gene SALL4 reactivation by hepatitis B virus counteracts miR-200c in PD-L1-induced T cell exhaustion. Nat Commun, 9:1241. https://doi.org/10.1038/s41467-018-03584-3
Sun JR, Zhang X, Zhang Y, 2019. MiR-214 prevents the progression of diffuse large B-cell lymphoma by targeting PD-L1. Cell Mol Biol Lett, 24:68. https://doi.org/10.1186/s11658-019-0190-9
Sun MY, Richards S, Prasad DVR, et al., 2002. Characterization of mouse and human B7-H3 genes. J Immunol, 168(12):6294–6297. https://doi.org/10.4049/jimmunol.168.12.6294
Sun W, Zhang Q, Wang RK, et al., 2021. Targeting DNA damage repair for immune checkpoint inhibition: mechanisms and potential clinical applications. Front Oncol, 11:648687. https://doi.org/10.3389/fonc.2021.648687
Tan SZ, Li DP, Zhu X, 2020. Cancer immunotherapy: pros, cons and beyond. Biomed Pharmacother, 124:109821. https://doi.org/10.1016/j.biopha.2020.109821
Thibult ML, Mamessier E, Gertner-Dardenne J, et al., 2013. PD-1 is a novel regulator of human B-cell activation. Int Immunol, 25(2):129–137. https://doi.org/10.1093/intimm/dxs098
Topalian SL, Taube JM, Anders RA, et al., 2016. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer, 16(5):275–287. https://doi.org/10.1038/nrc.2016.36
Topalian SL, Taube JM, Pardoll DM, 2020. Neoadjuvant checkpoint blockade for cancer immunotherapy. Science, 367(6477):eaax0182. https://doi.org/10.1126/science.aax0182
van Rensburg HJJ, Azad T, Ling M, et al., 2018. The Hippo pathway component TAZ promotes immune evasion in human cancer through PD-L1. Cancer Res, 78(6):1457–1470. https://doi.org/10.1158/0008-5472.Can-17-3139
Wang B, Ran ZJ, Liu MM, et al., 2019. Prognostic significance of potential immune checkpoint member HHLA2 in human tumors: a comprehensive analysis. Front Immunol, 10:1573. https://doi.org/10.3389/fimmu.2019.01573
Wang CG, Feng HR, Cheng X, et al., 2020. Potential therapeutic targets of B7 family in colorectal cancer. Front Immunol, 11:681. https://doi.org/10.3389/fimmu.2020.00681
Wang J, Jia Y, Zhao S, et al., 2017. BIN1 reverses PD-L1-mediated immune escape by inactivating the c-MYC and EGFR/MAPK signaling pathways in non-small cell lung cancer. Oncogene, 36(45):6235–6243. https://doi.org/10.1038/onc.2017.217
Wang J, Sanmamed MF, Datar I, et al., 2019. Fibrinogen-like protein 1 is a major immune inhibitory ligand of LAG-3. Cell, 176(1–2):334–347.e12. https://doi.org/10.1016/j.cell.2018.11.010
Wang J, Zhang RG, Lin ZY, et al., 2020. CDK7 inhibitor THZ1 enhances antiPD-1 therapy efficacy via the p38α/MYC/PD-L1 signaling in non-small cell lung cancer. J Hematol Oncol, 13:99. https://doi.org/10.1186/s13045-020-00926-x
Wang JH, Wu GP, Manick B, et al., 2019. VSIG-3 as a ligand of VISTA inhibits human T-cell function. Immunology, 156(1):74–85. https://doi.org/10.1111/imm.13001
Wang JY, Wang WP, 2020. B7-H4, a promising target for immunotherapy. Cell Immunol, 347:104008. https://doi.org/10.1016/j.cellimm.2019.104008
Wang MM, Yu WP, Gao J, et al., 2020. MicroRNA-487a-3p functions as a new tumor suppressor in prostate cancer by targeting CCND1. J Cell Physiol, 235(2):1588–1600. https://doi.org/10.1002/jcp.29078
Wang X, Li JG, Dong K, et al., 2015. Tumor suppressor miR-34a targets PD-L1 and functions as a potential immunotherapeutic target in acute myeloid leukemia. Cell Signal, 27(3):443–452. https://doi.org/10.1016/j.cellsig.2014.12.003
Wei SC, Duffy CR, Allison JP, 2018. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov, 8(9):1069–1086. https://doi.org/10.1158/2159-8290.Cd-18-0367
Woo SR, Turnis ME, Goldberg MV, et al., 2012. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res, 72(4):917–927. https://doi.org/10.1158/0008-5472.Can-11-1620
Wu Q, Jiang L, Li SC, et al., 2021. Small molecule inhibitors targeting the PD-1/PD-L1 signaling pathway. Acta Pharmacol Sin, 42(1):1–9. https://doi.org/10.1038/s41401-020-0366-x
Wu YQ, Borde M, Heissmeyer V, et al., 2006. FOXP3 controls regulatory T cell function through cooperation with NFAT. Cell, 126(2):375–387. https://doi.org/10.1016/j.cell.2006.05.042
Wu YQ, Zhang C, Liu XL, et al., 2021. ARIH1 signaling promotes anti-tumor immunity by targeting PD-L1 for proteasomal degradation. Nat Commun, 12:2346. https://doi.org/10.1038/s41467-021-22467-8
Wu Z, Huang RT, Yuan L, 2019. Crosstalk of intracellular post-translational modifications in cancer. Arch Biochem Biophys, 676:108138. https://doi.org/10.1016/j.abb.2019.108138
Xie GC, Li W, Li RD, et al., 2017. Helicobacter pylori promote B7-H1 expression by suppressing miR-152 and miR-200b in gastric cancer cells. PLoS ONE, 12(1):e0168822. https://doi.org/10.1371/journal.pone.0168822
Xie WB, Liang LH, Wu KG, et al., 2018. MiR-140 expression regulates cell proliferation and targets PD-L1 in NSCLC. Cell Physiol Biochem, 46(2):654–663. https://doi.org/10.1159/000488634
Xiong WJ, Gao Y, Wei WY, et al., 2021. Extracellular and nuclear PD-L1 in modulating cancer immunotherapy. Trends Cancer, 7(9):837–846. https://doi.org/10.1016/j.trecan.2021.03.003
Xu LJ, Chen XD, Shen MJ, et al., 2018. Inhibition of IL-6-JAK/Stat3 signaling in castration-resistant prostate cancer cells enhances the NK cell-mediated cytotoxicity via alteration of PD-L1/NKG2D ligand levels. Molr Oncol, 12(3):269–286. https://doi.org/10.1002/1878-0261.12135
Xu SH, Tao Z, Hai B, et al., 2016. miR-424(322) reverses chemoresistance via T-cell immune response activation by blocking the PD-L1 immune checkpoint. Nat Commun, 7:11406. https://doi.org/10.1038/ncomms11406
Xu SJ, Wang XY, Yang YB, et al., 2021. LSD1 silencing contributes to enhanced efficacy of anti-CD47/PD-L1 immunotherapy in cervical cancer. Cell Death Dis, 12(4):282. https://doi.org/10.1038/s41419-021-03556-4
Xu YP, Zhu SX, Song M, et al., 2014. B7-H4 expression and its role in interleukin-2/interferon treatment of clear cell renal cell carcinoma. Oncol Lett, 7(5):1474–1478. https://doi.org/10.3892/ol.2014.1961
Yamaguchi H, Hsu JM, Yang WH, et al., 2022. Mechanisms regulating PD-L1 expression in cancers and associated opportunities for novel small-molecule therapeutics. Nat Rev Clin Oncol, 19(5):287–305. https://doi.org/10.1038/s41571-022-00601-9
Yamamoto R, Nishikori M, Tashima M, et al., 2009. B7-H1 expression is regulated by MEK/ERK signaling pathway in anaplastic large cell lymphoma and Hodgkin lymphoma. Cancer Sci, 100(11):2093–2100. https://doi.org/10.1111/j.1349-7006.2009.01302.x
Yang H, Bueso-Ramos C, Dinardo C, et al., 2014. Expression of PD-L1, PD-L2, PD-1 and CTLA4 in myelodysplastic syndromes is enhanced by treatment with hypomethylating agents. Leukemia, 28(6):1280–1288. https://doi.org/10.1038/leu.2013.355
Yang Y, Hsu JM, Sun L, et al., 2019. Palmitoylation stabilizes PD-L1 to promote breast tumor growth. Cell Res, 29(1): 83–86. https://doi.org/10.1038/s41422-018-0124-5
Yao H, Lan J, Li CS, et al., 2019. Inhibiting PD-L1 palmitoylation enhances T-cell immune responses against tumours. Nat Biomed Eng, 3(4):306–317. https://doi.org/10.1038/s41551-019-0375-6
Yao S, Jiang L, Moser EK, et al., 2015. Control of pathogenic effector T-cell activities in situ by PD-L1 expression on respiratory inflammatory dendritic cells during respiratory syncytial virus infection. Mucosal Immunol, 8(4): 746–759. https://doi.org/10.1038/mi.2014.106
Yi M, Niu MK, Xu LP, et al., 2021. Regulation of PD-L1 expression in the tumor microenvironment. J Hematol Oncol, 14:10. https://doi.org/10.1186/s13045-020-01027-5
Yuan JM, Dong XD, Yap J, et al., 2020. The MAPK and AMPK signalings: interplay and implication in targeted cancer therapy. J Hematol Oncol, 13:113. https://doi.org/10.1186/s13045-020-00949-4
Zerdes I, Matikas A, Bergh J, et al., 2018. Genetic, transcriptional and post-translational regulation of the programmed death protein ligand 1 in cancer: biology and clinical correlations. Oncogene, 37(34):4639–4661. https://doi.org/10.1038/s41388-018-0303-3
Zhang GB, Chen YJ, Shi Q, et al., 2004. Human recombinant B7-H3 expressed in E. coli enhances T lymphocyte proliferation and IL-10 secretion in vitro. Acta Biochim Biophys Sin, 36(6):430–436. https://doi.org/10.1093/abbs/36.6.430
Zhang JF, Bu X, Wang HZ, et al., 2018. Cyclin D-CDK4 kinase destabilizes PD-L1 via cullin 3-SPOP to control cancer immune surveillance. Nature, 553(7686):91–95. https://doi.org/10.1038/nature25015
Zhang WJ, Liu Y, Yan ZY, et al., 2020. IL-6 promotes PD-L1 expression in monocytes and macrophages by decreasing protein tyrosine phosphatase receptor type O expression in human hepatocellular carcinoma. J Immunother Cancer, 8(1):e000285. https://doi.org/10.1136/jitc-2019-000285
Zhang Y, Xiang C, Wang YL, et al., 2017. PD-L1 promoter methylation mediates the resistance response to anti-PD-1 therapy in NSCLC patients with EGFR-TKI resistance. Oncotarget, 8(60):101535–101544. https://doi.org/10.18632/oncotarget.21328
Zhang YY, Zhang ZM, 2020. The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol, 17(8):807–821. https://doi.org/10.1038/s41423-020-0488-6
Zhu D, Xu RD, Huang XP, et al., 2021. Deubiquitinating enzyme OTUB1 promotes cancer cell immunosuppression via preventing ER-associated degradation of immune checkpoint protein PD-L1. Cell Death Differ, 28(6):1773–1789. https://doi.org/10.1038/s41418-020-00700-z
Zhu TY, Hu ZH, Wang ZY, et al., 2020. Epigenetically silenced PD-L1 confers drug resistance to anti-PD1 therapy in gastric cardia adenocarcinoma. Int Immunopharmacol, 82: 106245. https://doi.org/10.1016/j.intimp.2020.106245
Zhu YW, Yao S, Iliopoulou BP, et al., 2013. B7-H5 costimulates human T cells via CD28H. Nat Commun, 4:2043. https://doi.org/10.1038/ncomms3043
Acknowledgments
This work was supported by the National Key Research and Development Program of China (No. 2021YFC2700903), the National Natural Science Foundation of China (Nos. 81672791 and 81872300), the Zhejiang Provincial Natural Science Fund for Distinguished Young Scholars of China (No. LR18C060002), the Huadong Medicine Joint Funds of the Zhejiang Provincial Natural Science Foundation of China (No. LHDMY22H160006), and the ZJU-QILU Joint Research Institute and Qilu Group.
Author information
Authors and Affiliations
Contributions
Aifu LIN determined the topic of the article and proposed this program. Zerui GAO, Xingyi LING, Chengyu SHI, and Ying WANG collected the literature. Zerui GAO and Xingyi LING summarized and drew a diagram of the mechanism. Zerui GAO, Xingyi LING, Chengyu SHI, and Aifu LIN wrote the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Additional information
Compliance with ethics guidelines
Zerui GAO, Xingyi LING, Chengyu SHI, Ying WANG, and Aifu LIN declare that they have no conflict of interest.
This review does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Gao, Z., Ling, X., Shi, C. et al. Tumor immune checkpoints and their associated inhibitors. J. Zhejiang Univ. Sci. B 23, 823–843 (2022). https://doi.org/10.1631/jzus.B2200195
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200195
Key words
- Immune checkpoint
- Immune checkpoint inhibitor
- Programmed cell death-ligand 1 (PD-L1)
- Cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4)
- Lymphocyte activation gene-3 (LAG-3)
- T-cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT)
- B7 family