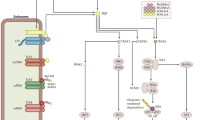
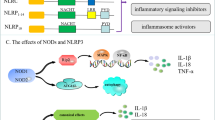
Abstract
Diabetic nephropathy (DN) is currently the most common complication of diabetes. It is considered to be one of the leading causes of end-stage renal disease (ESRD) and affects many diabetic patients. The pathogenesis of DN is extremely complex and has not yet been clarified; however, in recent years, increasing evidence has shown the important role of innate immunity in DN pathogenesis. Pattern recognition receptors (PRRs) are important components of the innate immune system and have a significant impact on the occurrence and development of DN. In this review, we classify PRRs into secretory, endocytic, and signal transduction PRRs according to the relationship between the PRRs and subcellular compartments. PRRs can recognize related pathogen-associated molecular patterns (PAMPs) and danger-associated molecular patterns (DAMPs), thus triggering a series of inflammatory responses, promoting renal fibrosis, and finally causing renal impairment. In this review, we describe the proposed role of each type of PRRs in the development and progression of DN.
摘要
目的
阐述不同类型的模式识别受体(PRR)和糖尿病肾病(DN)之间的关系,以提供治疗DN 的新方 法和策略.
摘要
DN 是目前糖尿病最常见的并发症.它被认为是 终末期肾病(ESRD)的主要原因之一,并影响 许多糖尿病患者.DN 的发病机制极为复杂,且 尚未阐明,但近年来,越来越多的证据表明先天 免疫在DN 发病机制中起到了重要作用.PRR 是 先天免疫系统的重要组成部分,对DN 的发生和 发展具有重要影响.在本综述中,我们根据PRR 与亚细胞区室之间的关系将PRR 分为分泌型、内 吞型和信号转导型三种类型.PRR 可以识别相关 的病原体相关分子模式(PAMP)和危险相关分 子模式(DAMP),从而触发一系列炎症反应, 促进肾纤维化,最终导致肾功能损害.此外,我 们还描述了各种类型的PRR在DN的发生和发展 中的作用.
Similar content being viewed by others
References
Abbas SA, Raza ST, Mir SS, et al., 2018. Role of variants rs5030717 and rs5030718 of TLR4 in the risk prediction of nephropathy, hypertension and dyslipidaemia in type 2 diabetes mellitus. Br J Biomed Sci, 75(4):163–168. https://doi.org/10.1080/09674845.2018.1477033
Awad AS, You HN, Gao T, et al., 2015. Macrophage-derived tumor necrosis factor-α mediates diabetic renal injury. Kidney Int, 88(4):722–733. https://doi.org/10.1038/ki.2015.162
Axelgaard E, Østergaard JA, Thiel S, et al., 2017a. Diabetes is associated with increased autoreactivity of mannan-binding lectin. J Diabetes Res, 2017:6368780. https://doi.org/10.1155/2017/6368780
Axelgaard E, Østergaard JA, Haxha S, et al., 2017b. Global autorecognition and activation of complement by mannan-binding lectin in a mouse model of type 1 diabetes. Mediators Inflamm, 2017:9403754. https://doi.org/10.1155/2017/9403754
Bidula S, Sexton DW, Schelenz S, 2019. Ficolins and the recognition of pathogenic microorganisms: an overview of the innate immune response and contribution of single nucleotide polymorphisms. J Immunol Res, 2019:3205072. https://doi.org/10.1155/2019/3205072
Bijkerk R, van der Pol P, Khairoun M, et al., 2016. Simultaneous pancreas-kidney transplantation in patients with type 1 diabetes reverses elevated MBL levels in association with MBL2 genotype and VEGF expression. Dia-betologia, 59(4):853–858. https://doi.org/10.1007/s00125-015-3858-3
Chen KH, Zhang JG, Zhang WW, et al., 2013. ATP-P2X4 signaling mediates NLRP3 inflammasome activation: a novel pathway of diabetic nephropathy. Int J Biochem Cell Biol, 45(5):932–943. https://doi.org/10.1016/j.biocel.2013.02.009
Chen XH, Luo J, Wu MM, et al., 2018. Study on association of pentraxin 3 and diabetic nephropathy in a rat model. J Diabetes Res, 2018:8968573. https://doi.org/10.1155/2018/8968573
Collins AJ, Foley RN, Chavers B, et al., 2012. United States Renal Data System 2011 Annual Data Report: atlas of chronic kidney disease & end-stage renal disease in the United States. Am J Kidney Dis, 59(1 Suppl 1):A7. https://doi.org/10.1053/j.ajkd.2011.11.015
D’addio F, Trevisani A, Nasr MB, et al., 2014. Harnessing the immunological properties of stem cells as a therapeutic option for diabetic nephropathy. Acta Diabetol, 51(6):897–904. https://doi.org/10.1007/s00592-014-0603-1
Ding T, Chen W, Li J, et al., 2017. High glucose induces mouse mesangial cell overproliferation via inhibition of hydrogen sulfide synthesis in a TLR-4-dependent manner. Cell Physiol Biochem, 41(3):1035–1043. https://doi.org/10.1159/000461483
Dobö J, Harmat V, Beinrohr L, et al., 2009. MASP-1, a promiscuous complement protease: structure of its catalytic region reveals the basis of its broad specificity. J Immunol, 183(2):1207–1214. https://doi.org/10.4049/jimmunol.0901141
Dowling JK, O’Neill LA, 2012. Biochemical regulation of the inflammasome. Crit Rev Biochem Mol Biol, 47(5):424–443. https://doi.org/10.3109/10409238.2012.694844
Feng L, Gu CW, Li YX, et al., 2017. High glucose promotes CD36 expression by upregulating peroxisome proliferator-activated receptor γ levels to exacerbate lipid deposition in renal tubular cells. BioMed Res Int, 2017:1414070. https://doi.org/10.1155/2017/1414070
Feng YY, Yang SL, Ma YX, et al., 2015. Role of Toll-like receptors in diabetic renal lesions in a miniature pig model. Sci Adv, 1(5):e1400183. https://doi.org/10.1126/sciadv.1400183
Fernandes-Alnemri T, Yu JW, Datta P, et al., 2009. AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature, 458(7237):509–513. https://doi.org/10.1038/nature07710
Flyvbjerg A, 2017. The role of the complement system in diabetic nephropathy. Nat Rev Nephrol, 13(5):311–318. https://doi.org/10.1038/nrneph.2017.31
Guo CC, Zhang LJ, Nie LH, et al., 2016. Association of polymorphisms in the MyD88, IRAK4 and TRAF6 genes and susceptibility to type 2 diabetes mellitus and diabetic nephropathy in a southern Han Chinese population. Mol Cell Endocrinol, 429:114–119. https://doi.org/10.1016/j.mce.2016.04.003
Guo XX, Wang Y, Wang K, et al., 2018. Stability of a type 2 diabetes rat model induced by high-fat diet feeding with low-dose streptozotocin injection. J Zhejiang Univ Sci-B (Biomed & Biotechnol), 19(7):559–569. https://doi.org/10.1631/jzus.B1700254
Gutwein P, Abdel-Bakky MS, Doberstein K, et al., 2009. CXCL16 and oxLDL are induced in the onset of diabetic nephropathy. J Cell Mol Med, 13(9b):3809–3825. https://doi.org/10.1111/j.1582-4934.2009.00761.x
Gysler SM, Mulla MJ, Guerra M, et al., 2016. Antiphospho-lipid antibody-induced miR-146a-3p drives trophoblast interleukin-8 secretion through activation of Toll-like receptor 8. Mol Hum Reprod, 22(7):465–474. https://doi.org/10.1093/molehr/gaw027
Han F, Gao Y, Ding CG, et al., 2018. Knockdown of NLRC5 attenuates renal I/R injury in vitro through the activation of PI3K/Akt signaling pathway. Biomed Pharmacother, 103:222–227. https://doi.org/10.1016/j.biopha.2018.04.040
Han YH, Xu XX, Tang CY, et al., 2018. Reactive oxygen species promote tubular injury in diabetic nephropathy: the role of the mitochondrial ROS-TXNIP-NLRP3 biological axis. Redox Biol, 16:32–46. https://doi.org/10.1016/j.redox.2018.02.013
Henriksen ML, Brandt J, Andrieu JP, et al., 2013. Heteromeric complexes of native collectin kidney 1 and collectin liver 1 are found in the circulation with MASPs and activate the complement system. J Immunol, 191(12):6117–6127. https://doi.org/10.4049/jimmunol.1302121
Hisano S, Matsushita M, Fujita T, et al., 2007. Activation of the lectin complement pathway in post-streptococcal acute glomerulonephritis. Pathol Int, 57(6):351–357. https://doi.org/10.1111/j.1440-1827.2007.02107.x
Horiuchi S, Unno Y, Usui H, et al., 2005. Pathological roles of advanced glycation end product receptors SR-A and CD36. Ann N Y Acad Sci, 1043(1):671–675. https://doi.org/10.1196/annals.1333.076
Hou YJ, Wu M, Wei JY, et al., 2015. CD36 is involved in high glucose-induced epithelial to mesenchymal transition in renal tubular epithelial cells. Biochem Biophys Res Commun, 468(1–2):281–286. https://doi.org/10.1016/j.bbrc.2015.10.112
Hu ZB, Ma KL, Zhang Y, et al., 2018. Inflammation-activated CXCL16 pathway contributes to tubulointerstitial injury in mouse diabetic nephropathy. Acta Pharmacol Sin, 39(6):1022–1033. https://doi.org/10.1038/aps.2017.177
Huang W, Gou F, Long Y, et al., 2016. High glucose and lipopolysaccharide activate NOD1-RICK-NF-kB inflammatory signaling in mesangial cells. Exp Clin Endocrinol Diabetes, 124(8):512–517. https://doi.org/10.1055/s-0042-105641
Huang YQ, Gou R, Diao YS, et al., 2014. Charlson comorbidity index helps predict the risk of mortality for patients with type 2 diabetic nephropathy. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 15(1):58–66. https://doi.org/10.1631/jzus.B1300109
Janowski AM, Kolb R, Zhang WZ, et al., 2013. Beneficial and detrimental roles of NLRs in carcinogenesis. Front Immunol, 4:370. https://doi.org/10.3389/fimmu.2013.00370
Jheng HF, Tsai PJ, Chuang YL, et al., 2015. Albumin stimulates renal tubular inflammation through an HSP70-TLR4 axis in mice with early diabetic nephropathy. Dis Model Mech, 8(10):1311–1321. https://doi.org/10.1242/dmm.019398
Komada T, Chung H, Lau A, et al., 2018. Macrophage uptake of necrotic cell DNA activates the AIM2 inflammasome to regulate a proinflammatory phenotype in CKD. J Am Soc Nephrol, 29(4):1165–1181. https://doi.org/10.1681/asn.2017080863
Landis RC, Quimby KR, Greenidge AR, 2018. M1/M2 macrophages in diabetic nephropathy: Nrf2/HO-1 as therapeutic targets. Curr Pharm Des, 24(20):2241–2249. https://doi.org/10.2174/1381612824666180716163845
Lin M, Tang SCW, 2014. Toll-like receptors: sensing and reacting to diabetic injury in the kidney. Nephrol Dial Transplant, 29(4):746–754. https://doi.org/10.1093/ndt/gft446
Liu Q, Jiang CY, Chen BX, et al., 2015. The association between high-sensitivity C-reactive protein concentration and diabetic nephropathy: a meta-analysis. Eur Rev Med Pharmacol Sci, 19(23):4558–4568.
Liu YW, Hao YC, Chen YJ, et al., 2018. Protective effects of sarsasapogenin against early stage of diabetic nephropathy in rats. Phytother Res, 32(8):1574–1582. https://doi.org/10.1002/ptr.6088
Lu L, Peng WH, Wang W, et al., 2011. Effects of atorvastatin on progression of diabetic nephropathy and local RAGE and soluble RAGE expressions in rats. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 12(8):652–659. https://doi.org/10.1631/jzus.B1101004
Luan PP, Zhuang JH, Zou J, et al., 2018. NLRC5 deficiency ameliorates diabetic nephropathy through alleviating inflammation. FASEB J, 32(2):1070–1084. https://doi.org/10.1096/fj.201700511RR
Masters SL, 2013. Specific inflammasomes in complex diseases. Clin Immunol, 147(3):223–228. https://doi.org/10.1016/j.clim.2012.12.006
McKnight AJ, Duffy S, Maxwell AP, 2015. Genetics of diabetic nephropathy: a long road of discovery. Curr Diab Rep, 15(7):41. https://doi.org/10.1007/s11892-015-0610-9
Medzhitov R, Janeway CA Jr., 1997. Innate immunity: impact on the adaptive immune response. Curr Opin Immunol, 9(1):4–9. https://doi.org/10.1016/S0952-7915(97)80152-5
Niewczas MA, Pavkov ME, Skupien J, et al., 2019. A signature of circulating inflammatory proteins and development of end-stage renal disease in diabetes. Nat Med, 25(5):805–813. https://doi.org/10.1038/s41591-019-0415-5
Nosadini R, Tonolo G, 2011. Role of oxidized low density lipoproteins and free fatty acids in the pathogenesis of glomerulopathy and tubulointerstitial lesions in type 2 diabetes. Nutr Metab Cardiovasc Dis, 21(2):79–85. https://doi.org/10.1016/j.numecd.2010.10.002
Osborn O, Olefsky JM, 2012. The cellular and signaling networks linking the immune system and metabolism in disease. Nat Med, 18(3):363–374. https://doi.org/10.1038/nm.2627
Østergaard JA, Bjerre M, Ramachandrarao SP, et al., 2012. Mannan-binding lectin in diabetic kidney disease: the impact of mouse genetics in a type 1 diabetes model. Exp Diabetes Res, 2012:678381. https://doi.org/10.1155/2012/678381
Østergaard JA, Bjerre M, Dagnaes-Hansen F, et al., 2013. Diabetes-induced changes in mannan-binding lectin levels and complement activation in a mouse model of type 1 diabetes. Scand J Immunol, 77(3):187–194. https://doi.org/10.1111/sji.12027
Østergaard JA, Thiel S, Hovind P, et al., 2014. Association of the pattern recognition molecule H-ficolin with incident microalbuminuria in an inception cohort of newly diagnosed type 1 diabetic patients: an 18 year follow-up study. Diabetologia, 57(10):2201–2207. https://doi.org/10.1007/s00125-014-3332-7
Østergaard JA, Thiel S, Hoffmann-Petersen IT, et al., 2017. Incident microalbuminuria and complement factor mannan-binding lectin-associated protein 19 in people with newly diagnosed type 1 diabetes. Diabetes Metab Res Rev, 33(5):e2895. https://doi.org/10.1002/dmrr.2895
Overgaard AJ, McGuire JN, Hovind P, et al., 2013. Serum amyloid A and C-reactive protein levels may predict microalbuminuria and macroalbuminuria in newly diagnosed type 1 diabetic patients. J Diabetes Complications, 27(1):59–63. https://doi.org/10.1016/j.jdiacomp.2012.06.016
Pombinho R, Sousa S, Cabanes D, 2018. Scavenger receptors: promiscuous players during microbial pathogenesis. Crit Rev Microbiol, 44(6):685–700. https://doi.org/10.1080/1040841x.2018.1493716
Qiao YC, Tian XX, Men L, et al., 2018. Spleen tyrosine kinase promotes NLR family pyrin domain containing 3 inflammasome-mediated IL-1β secretion via c-Jun N-terminal kinase activation and cell apoptosis during diabetic nephropathy. Mol Med Rep, 18(2):1995–2008. https://doi.org/10.3892/mmr.2018.9164
Rao V, Tan SH, Candasamy M, et al., 2019. Diabetic nephropathy: an update on pathogenesis and drug development. Diabetes Metab Syndr, 13(1):754–762. https://doi.org/10.1016/j.dsx.2018.11.054
Shaheer AK, Tharayil JK, Krishna PW, 2017. A comparative study of high sensitivity C-reactive protein and metabolic variables in type 2 diabetes mellitus with and without nephropathy. J Clin Diagn Res, 11(9):BC01–BC04. https://doi.org/10.7860/jcdr/2017/30272.10528
Shang J, Wan Q, Wang XJ, et al., 2015. Identification of NOD2 as a novel target of RNA-binding protein HuR: evidence from NADPH oxidase-mediated HuR signaling in diabetic nephropathy. Free Radic Biol Med, 79:217–227. https://doi.org/10.1016/j.freeradbiomed.2014.12.013
Shang J, Zhang Y, Jiang YM, et al., 2017. NOD2 promotes endothelial-to-mesenchymal transition of glomerular endothelial cells via MEK/ERK signaling pathway in diabetic nephropathy. Biochem Biophys Res Commun, 484(2):435–441. https://doi.org/10.1016/j.bbrc.2017.01.155
Sifuentes-Franco S, Padilla-Tejeda DE, Carrillo-Ibarra S, et al., 2018. Oxidative stress, apoptosis, and mitochondrial function in diabetic nephropathy. Int J Endocrinol, 2018:1875870. https://doi.org/10.1155/2018/1875870
Soares JLS, Fernandes FP, Patente TA, et al., 2018. Gain-of-function variants in NLRP1 protect against the development of diabetic kidney disease: NLRP1 inflammasome role in metabolic stress sensing? Clin Immunol, 187:46–49. https://doi.org/10.1016/j.clim.2017.10.003
Subhash V, Monika S, Richa S, et al., 2018. Distribution of single nucleotide polymorphisms and protein domain architecture of Toll-like receptor-2 in Pahari cattle (Indian non-descript indigenous breed). Res Vet Sci, 117:144–149. https://doi.org/10.1016/j.rvsc.2017.12.003
Sun HB, Tian J, Xian WH, et al., 2015. Pentraxin-3 attenuates renal damage in diabetic nephropathy by promoting M2 macrophage differentiation. Inflammation, 38(5):1739–1747. https://doi.org/10.1007/s10753-015-0151-z
Sun L, Wen JH, Sun HL, et al., 2012. Perindopril attenuates renal tubulointerstitium injury by inhibiting scavenger receptor A over-expression in diabetic rats. J Endocrinol Invest, 35(5):511–515. https://doi.org/10.3275/7867
Tang Y, Fung E, Xu AP, et al., 2017. C-reactive protein and ageing. Clin Exp Pharmacol Physiol, 44(S1):9–14. https://doi.org/10.1111/1440-1681.12758
Thakur V, Nargis S, Gonzalez M, et al., 2017. Role of glycyrrhizin in the reduction of inflammation in diabetic kidney disease. Nephron, 137(2):137–147. https://doi.org/10.1159/000477820
Tsun JGS, Shiu SWM, Wong Y, et al., 2013. Impact of serum amyloid A on cellular cholesterol efflux to serum in type 2 diabetes mellitus. Atherosclerosis, 231(2):405–410. https://doi.org/10.1016/j.atherosclerosis.2013.10.008
Tsun JGS, Yung S, Chau MKM, et al., 2014. Cellular cholesterol transport proteins in diabetic nephropathy. PLoS ONE, 9(9):e105787. https://doi.org/10.1371/journal.pone.0105787
Usui HK, Shikata K, Sasaki M, et al., 2007. Macrophage scavenger receptor-A-deficient mice are resistant against diabetic nephropathy through amelioration of microinflammation. Diabetes, 56(2):363–372. https://doi.org/10.2337/db06-0359
Uzun S, Ozari M, Gursu M, et al., 2016. Changes in the inflammatory markers with advancing stages of diabetic nephropathy and the role of pentraxin-3. Ren Fail, 38(8):1193–1198. https://doi.org/10.1080/0886022x.2016.1209031
Wada J, Makino H, 2016. Innate immunity in diabetes and diabetic nephropathy. Nat Rev Nephrol, 12(1):13–26. https://doi.org/10.1038/nrneph.2015.175
Wang C, Hou XX, Rui HL, et al., 2018. Artificially cultivated Ophiocordyceps sinensis alleviates diabetic nephropathy and its podocyte injury via inhibiting P2X7R expression and NLRP3 inflammasome activation. J Diabetes Res, 2018:1390418. https://doi.org/10.1155/2018/1390418
Wang HQ, Wang SS, Chiufai K, et al., 2019. Umbelliferone ameliorates renal function in diabetic nephropathy rats through regulating inflammation and TLR/NF-κB pathway. Chin J Nat Med, 17(5):346–354. https://doi.org/10.1016/s1875-5364(19)30040-8
Wang SF, Li YB, Fan JJ, et al., 2017. Interleukin-22 ameliorated renal injury and fibrosis in diabetic nephropathy through inhibition of NLRP3 inflammasome activation. Cell Death Dis, 8(7):e2937. https://doi.org/10.1038/cddis.2017.292
Wilhelm I, Nyúl-Tóth A, Kozma M, et al., 2017. Role of pattern recognition receptors of the neurovascular unit in inflamm-aging. Am J Physiol Heart Circ Physiol, 313(5):H1000–H1012. https://doi.org/10.1152/ajpheart.00106.2017
Wu XH, Huang SM, Fan WX, et al., 2011. Influence of high glucose and mannose binding lectin complement pathway activation to IL-6 and TNF-alpha’s expression by human renal glomerular endothelial cells. J Sichuan Univ (Med Sci Ed), 42(1):90–94 (in Chinese).
Yang WF, Han F, Zhang XH, et al., 2013. Extra-pulmonary tuberculosis infection in the dialysis patients with end stage renal diseases: case reports and literature review. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 14(1):76–82. https://doi.org/10.1631/jzus.B1200244
Yang YL, Lin SH, Chuang LY, et al., 2007. CD36 is a novel and potential anti-fibrogenic target in albumin-induced renal proximal tubule fibrosis. J Cell Biochem, 101(3):735–744. https://doi.org/10.1002/jcb.21236
Yang YX, Huang SM, Yan XY, et al., 2011. Relationship between activation of mannan-binding lectin complement and NF-κB in diabetic nephropathy. J Sichuan Univ (Med Sci Ed), 42(4):490–493 (in Chinese).
Ye YN, Chen QZ, Li JM, et al., 2017. CXCL16 deficiency attenuates diabetic nephropathy through decreasing oxidative stress and inflammation. Biochem Biophys Res Commun, 491(3):848–854. https://doi.org/10.1016/j.bbrc.2017.05.013
Yilmaz MI, Axelsson J, Sonmez A, et al., 2009. Effect of renin angiotensin system blockade on pentraxin 3 levels in type-2 diabetic patients with proteinuria. Clin J Am Soc Nephrol, 4(3):535–541. https://doi.org/10.2215/cjn.04330808
Yilmaz MI, Carrero JJ, Martin-Ventura JL, et al., 2010. Combined therapy with renin-angiotensin system and calcium channel blockers in type 2 diabetic hypertensive patients with proteinuria: effects on soluble TWEAK, PTX3, and flow-mediated dilation. Clin J Am Soc Nephrol, 5(7):1174–1181. https://doi.org/10.2215/cjn.01110210
You YK, Huang XR, Chen HY, et al., 2016. C-reactive protein promotes diabetic kidney disease in db/db mice via the CD32b-Smad3-mTOR signaling pathway. Sci Rep, 6(1):26740. https://doi.org/10.1038/srep26740
Yuan F, Kolb R, Pandey G, et al., 2016. Involvement of the NLRC4-inflammasome in diabetic nephropathy. PLoS ONE, 11(10):e0164135. https://doi.org/10.1371/journal.pone.0164135
Zhang H, Zhang R, Chen J, et al., 2017. High mobility group box1 inhibitor glycyrrhizic acid attenuates kidney injury in streptozotocin-induced diabetic rats. Kidney Blood Press Res, 42(5):894–904. https://doi.org/10.1159/000485045
Zhang L, Shen ZY, Wang K, et al., 2019. C-reactive protein exacerbates epithelial-mesenchymal transition through Wnt/ β-catenin and ERK signaling in streptozocin-induced diabetic nephropathy. FASEB J, 33(5):6551–6563. https://doi.org/10.1096/fj.201801865RR
Zhang NN, Zhuang MQ, Ma AX, et al., 2013. Association of levels of mannose-binding lectin and the MBL2 gene with type 2 diabetes and diabetic nephropathy. PLoS ONE, 8(12):e83059. https://doi.org/10.1371/journal.pone.0083059
Zhao LP, Wu F, Jin LG, et al., 2014. Serum CXCL16 as a novel marker of renal injury in type 2 diabetes mellitus. PLoS ONE, 9(1):e87786. https://doi.org/10.1371/journal.pone.0087786
Zhao M, Han JG, 2018. Dendrobium officinale Kimura et Migo ameliorates insulin resistance in rats with diabetic nephropathy. Med Sci Monit Basic Res, 24:84–92. https://doi.org/10.12659/msmbr.909242
Zheng JM, Ren XG, Jiang ZH, et al., 2018. Lectin-induced renal local complement activation is involved in tubular interstitial injury in diabetic nephropathy. Clin Chim Acta, 482:65–73. https://doi.org/10.1016/j.cca.2018.03.033
Zheng SR, Coventry S, Cai L, et al., 2016. Renal protection by genetic deletion of the atypical chemokine receptor ACKR2 in diabetic OVE mice. J Diabetes Res, 2016:5362506. https://doi.org/10.1155/2016/5362506
Zhou HL, Tan KCB, Shiu SWM, et al., 2008. Cellular cholesterol efflux to serum is impaired in diabetic nephropathy. Diabetes Metab Res Rev, 24(8):617–623. https://doi.org/10.1002/dmrr.895
Zhou LP, Huang W, Xu YH, et al., 2018. Sweet taste receptors mediated ROS-NLRP3 inflammasome signaling activation: implications for diabetic nephropathy. J Diabetes Res, 2018:7078214. https://doi.org/10.1155/2018/7078214
Zhu H, Yu WH, Xie YY, et al., 2017. Association of pentraxin 3 gene polymorphisms with susceptibility to diabetic nephropathy. Med Sci Monit, 23:428–436. https://doi.org/10.12659/msm.902783
Zhu XG, Shi J, Li HC, 2018. Liquiritigenin attenuates high glucose-induced mesangial matrix accumulation, oxidative stress, and inflammation by suppression of the NF-κB and NLRP3 inflammasome pathways. Biomed Pharmacother, 106:976–982. https://doi.org/10.1016/j.biopha.2018.07.045
Author information
Authors and Affiliations
Contributions
Zhi-feng ZHOU and Lei JIANG set up the theme and the frame of this review, wrote and edited the manuscript. Qing ZHAO, Yu WANG, and Jing ZHOU performed the references collection and selection. Jin-lei LV and Qin-kai CHEN contributed the design and revision of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Additional information
Compliance with ethics guidelines
Zhi-feng ZHOU, Lei JIANG, Qing ZHAO, Yu WANG, Jing ZHOU, Qin-kai CHEN, and Jin-lei LV declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Project supported by the National Natural Science Foundation of China (Nos. 81060063 and 81660129)
Rights and permissions
About this article
Cite this article
Zhou, Zf., Jiang, L., Zhao, Q. et al. Roles of pattern recognition receptors in diabetic nephropathy. J. Zhejiang Univ. Sci. B 21, 192–203 (2020). https://doi.org/10.1631/jzus.B1900490
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900490