Abstract
An ultrasonic-assisted technique was employed to extract crude polysaccharide from Tricholoma matsutake fruiting bodies. Single-factor tests and orthogonal experimental design (L 9(33)) were used to obtain the optimal extraction conditions. Results showed that the optimal parameters were as follows: ultrasonic temperature, 40 °C; ultrasonic time, 50 min; water to raw material ratio, 25 ml/g; ultrasonic frequency, 45 kHz; and ultrasonic power, 100 W. Three novel T. matsutake polysaccharide (TMP) fractions (TMP30, TMP60, and TMP80) were isolated and purified from TMP by stepwise alcohol precipitation. Their preliminary structural features were determined by high-performance anion-exchange chromatography with pulsed-amperometric detection (HPAEC-PAD) and Fourier transform infrared spectrophotometer (FT-IR) analyses. Furthermore, their in vitro antioxidant activity was investigated in terms of a reducing power assay and the scavenging rates of 2,2-diphenyl-1-picrylhydrazyl (DPPH) and hydroxyl radicals. The order of the various fractions based on their antioxidant activity was TMP80>TMP>TMP60>TMP30. These findings suggested that novel polysaccharide fractions from T. matsutake, especially TMP80, could be promising active macromolecules for biomedical use.
摘要
目 的
对超声提取松茸多糖(TMP)的工艺条件进行优化, 对TMP 进一步分离纯化, 并对纯化多糖(TMP30、TMP60和TMP80)的结构和抗氧化活性进行评估。
创新点
首次对超声辅助制备的TMP 进行了乙醇分级, 对其进行了结构和抗氧化活性评估, 并筛选出了抗氧化活性最强的部位(TMP80)。
方 法
采用单因素试验和正交试验(L 9(33))对超声提取TMP 的工艺条件进行了优化; 采用不同的乙醇终浓度对TMP 分离纯化为3种多糖TMP30、TMP60和TMP80; 采用高效阴离子色谱(HPAEC-PAD)和傅里叶红外(FT-IR)等技术对3种多糖的理化性质进行了结构表征; 利用还原力测定、二苯三硝基苯肼(DPPH)和羟自由基的清除作用对它们的抗氧化活性进行了评估。
结 论
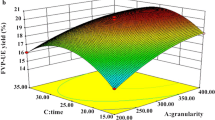
正交试验结果表明, 超声提取TMP 的最优工艺条件为: 超声温度40 °C, 超声时间50 min, 水原料比例25 ml/g, 超声波频率45 kHz 和超声波功率100 W(表1和表2)。在此条件下的TMP 得率为8.06%。采用乙醇分级法对TMP进一步分离纯化, 制备得到3种多糖TMP30、TMP60和TMP80。HPAEC-PAD 和FT-IR 的结果表明, TMP30、TMP60和TMP80均主要由岩藻糖(l-Fuc)、半乳糖(d-Gal)、葡萄糖(d-Glc)、木糖(d-Xyl)和甘露糖(d-Man)组成, 它们具有相同的单糖组成, 但含有不同的摩尔比, 且均有β-构型(图2、图3和表3)。抗氧化活性结果表明, 它们的能力高低顺序为: TMP80>TMP>TMP60>TMP30(图4和表4), TMP80抗氧化活性最高, 为松茸多糖的进一步开发利用提供重要的科学依据。
Similar content being viewed by others

References
Achat, S., Tomao, V., Madani, K., et al., 2012. Direct enrichment of olive oil in oleuropein by ultrasound-assisted maceration at laboratory and pilot plant scale. Ultrason. Sonochem., 19(4): 777–786. http://dx.doi.org/10.1016/j.ultsonch.2011.12.006
Afshari, K., Samavati, V., Shahidi, S.A., 2015. Ultrasonicassisted extraction and in-vitro antioxidant activity of polysaccharide from Hibiscus leaf. Int. J. Biol. Macromol., 74: 558–567. http://dx.doi.org/10.1016/j.ijbiomac.2014.07.023
Ardestani, A., Yazdanparast, R., 2007. Antioxidant and free radical scavenging potential of Achillea santolina extracts. Food Chem., 104(1): 21–29. http://dx.doi.org/10.1016/j.foodchem.2006.10.066
Bohn, J.A., BeMiller, J.N., 1995. (1→3)-β-d-Glucans as biological response modifiers: a review of structure-functional activity relationships. Carbohydr. Polym., 28(1): 3–14. http://dx.doi.org/10.1016/0144-8617(95)00076-3
Brand-Williams, W., Cuvelier, M.E., Berset, C., 1995. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol., 28(1): 25–30. http://dx.doi.org/10.1016/S0023-6438(95)80008-5
Chemat, F., Rombaut, N., Sicaire, A., et al., 2017. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem., 34: 540–560. http://dx.doi.org/10.1016/j.ultsonch.2016.06.035
Devasagayam, T.P.A., Tilak, J.C., Boloor, K.K., et al., 2004. Free radicals and antioxidants in human health: current status and future prospects. J. Assoc. Phys. India, 52: 794–804.
Du, X.J., Yang, Y., Ye, L.B., et al., 2009. Structural elucidation and immuno-stimulating property of an acidic heteropolysaccharide (TAPA1) from Tremella aurantialba. Carbohydr. Res., 344(5): 672–678. http://dx.doi.org/10.1016/j.carres.2009.01.021
Du, X.J., Mu, H.M., Zhou, S., et al., 2013. Chemical analysis and antioxidant activity of polysaccharides extracted from Inonotus obliquus sclerotia. Int. J. Biol. Macromol., 62(11): 691–696. http://dx.doi.org/10.1016/j.ijbiomac.2013.10.016
Hoshi, H., Iijima, B., Ishihara, Y., et al., 2008. Absorption and tissue distribution of an immunomodulatory α-d-glucan after oral administration of Tricholoma matsutake. J. Agric. Food Chem., 56(17): 7715–7720. http://dx.doi.org/10.1021/jf801123k
Hu, J., Jia, X.J., Fang, X.B., et al., 2016. Ultrasonic extraction, antioxidant and anticancer activities of nove lpolysaccharides from Chuanxiong rhizome. Int. J. Biol. Macromol., 85: 277–284. http://dx.doi.org/10.1016/j.ijbiomac.2015.12.046
Huang, S.Q., Ning, Z.X., 2010. Extraction of polysaccharide from Ganoderma lucidum and its immune enhancement activity. Int. J. Biol. Macromol., 47(3): 336–341. http://dx.doi.org/10.1016/j.ijbiomac.2010.03.019
Kim, S.S., Lee, J.S., Cho, J.Y., et al., 2010a. Effects of C/N ratio and trace elements on mycelial growth and exopolysaccharide production of Tricholoma matsutake. Biotechnol. Bioproc. E, 15(2): 293–298. http://dx.doi.org/10.1007/s12257-008-0226-x
Kim, S.S., Lee, J.S., Cho, J.Y., et al., 2010b. Process development for mycelial growth and polysaccharide production in Tricholoma matsutake liquid culture. J. Biosci. Bioeng., 109(4): 351–355. http://dx.doi.org/10.1016/j.jbiosc.2009.10.010
Li, Y., Fabiano-Tixier, A.S., Tomao, V., et al., 2013. Green ultrasound-assisted extraction of carotenoids based on the bio-refinery concept using sunflower oil as an alternative solvent. Ultrason. Sonochem., 20(1): 12–18. http://dx.doi.org/10.1016/j.ultsonch.2012.07.005
Luo, Q., Zhang, J., Yan, L., et al., 2011. Composition and antioxidant activity of water-soluble polysaccharides from Tuber indicum. J. Med. Food., 14(12): 1609–1616. http://dx.doi.org/10.1089/jmf.2011.1659
Lv, Y., Yang, X.B., Zhao, Y., et al., 2009. Separation and quantification of component monosaccharides of the tea polysaccharides from Gynostemma pentaphyllum by HPLC with indirect UV detection. Food Chem., 112(3): 742–746. http://dx.doi.org/10.1016/j.foodchem.2008.06.042
Murphey, J.M., Spayd, S.E., Powers, J.R., 1989. Effect of grape maturation on soluble protein characteristics of Gewurztraminer and White Riesling juice and wine. Am. J. Enol. Viticult., 40(3): 199–207.
Pingret, D., Durand, G., Fabiano-Tixier, A., et al., 2012. Degradation of edible oil during food processing by ultrasound: electron paramagnetic resonance, physicochemical, and sensory appreciation. J. Agric. Food Chem., 60(31): 7761–7768. http://dx.doi.org/10.1021/jf301286f
Smironff, N., Cumbes, Q.J., 1989. Hyroxyl radical scavenging activity of compatible solutes. Phytochemistry, 28(4): 1051–1060. http://dx.doi.org/10.1016/0031-9422(89)80182-7
Tursun, K., Zhan, R., Zhang, H., et al., 2010. Study on antioxidant activity of Acroptilon repens. Lett. Biol., 21(3): 406–411 (in Chinese). http://dx.doi.org/10.3969/j.issn.1009-0002.2010.03.025
Wang, N.N., Zhang, Y., Wang, X.P., et al., 2016. Antioxidant property of water-soluble polysaccharides from Poria cocos Wolf using different extraction methods. Int. J. Biol. Macromol., 83: 103–110. http://dx.doi.org/10.1016/j.ijbiomac.2015.11.032
Wang, Y.F., Yang, Z.W., Wei, X.L., 2012. Antioxidant activities potential of tea polysaccharide fractions obtained by ultrafiltration. Int. J. Biol. Macromol., 50(3): 558–564. http://dx.doi.org/10.1016/j.ijbiomac.2011.12.028
Xu, F., Jabasini, M., Baba, Y., 2005. Screening of mixed poly (ethylene oxide) solutions for microchip separation of double-stranded DNA using an orthogonal design approach. Electrophoresis, 26(15): 3013–3020. http://dx.doi.org/10.1002/elps.200410434
Yang, S., Ren, X.D., Sheng, J.X., et al., 2010. Preparation and the antitumor activity in vitro of polysaccharides from Tricholoma matsutake. W. J. Microbiol. Biotechnol., 26(3): 497–503. http://dx.doi.org/10.1007/s11274-009-0196-y
Ye, C., Han, N., Teng, F.K., et al., 2013. Extraction optimization of polysaccharides of Schisandrae Fructus and evaluation of their analgesic activity. Int. J. Biol. Macromol., 57(6): 291–296. http://dx.doi.org/10.1016/j.ijbiomac.2013.03.025
Ye, L.B., Zhang, J.S., Yang, Y., et al., 2009. Structural characterisation of a heteropolysaccharide by NMR spectra. Food Chem., 112(4): 962–966. http://dx.doi.org/10.1016/j.foodchem.2008.07.017
Yin, X.L., You, Q.H., Jiang, Z.H., 2009. Extraction and purification of Tricholoma matsutake polysaccharides. China Brewing, 211(10): 171–173.
Yin, X.L., You, Q.H., Jiang, Z.H., 2011. Optimization of enzyme assisted extraction of polysaccharides from Tricholoma matsutake by response surface methodology. Carbohydr. Polym., 86(3): 1358–1364. http://dx.doi.org/10.1016/j.carbpol.2011.06.053
You, L.J., Gao, Q., Feng, M.Y., et al., 2013. Structural characterisation of polysaccharides from Tricholoma matsutake and their antioxidant and antitumour activities. Food Chem., 138(4): 2242–2249. http://dx.doi.org/10.1016/j.foodchem.2012.11.140
You, Q.H., Yin, X.L., Ji, C.W., 2014. Pulsed counter-current ultrasound-assisted extraction and characterization of polysaccharides from Boletus edulis. Carbohydr. Polym., 101(1): 379–385. http://dx.doi.org/10.1016/j.carbpol.2013.09.031
Zhang, W.J., 1999. Biochemical Research Technology on Glycoconjugates, 2nd Ed. Zhejiang University Press, Hangzhou (in Chinese).
Zhao, Z., Xu, X., Ye, Q., et al., 2013. Ultrasound extraction optimization of Acanthopanax senticosus polysaccharides and its antioxidant activity. Int. J. Biol. Macromol., 59(4): 290–294. http://dx.doi.org/10.1016/j.ijbiomac.2013.04.067
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Natural Science Foundation of Shandong Province of China (No. ZR2015CL002) and the Doctoral Research Startup Foundation of Liaocheng University (No. 31805), China
Rights and permissions
About this article
Cite this article
Chen, Y., Du, Xj., Zhang, Y. et al. Ultrasound extraction optimization, structural features, and antioxidant activity of polysaccharides from Tricholoma matsutake . J. Zhejiang Univ. Sci. B 18, 674–684 (2017). https://doi.org/10.1631/jzus.B1600239
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1600239
Key words
- Tricholoma matsutake polysaccharide
- Orthogonal test design
- Ultrasound-assisted extraction
- Monosaccharide composition
- Antioxidant activity