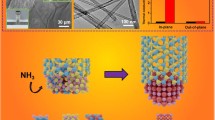
Abstract
Catalytic processes are critical steps in numerous industrial processes. The discovery of high reactivity of defects in metal-free two-dimensional materials has bolstered their emergence as catalysts. Here we consider the effect of defect-inducing methods in hexagonal boron nitride (h-BN) on their performance for olefin and CO2 hydrogenation. We compare the changes introduced by ball milling and heat treatment in h-BN and show how varying the treatment conditions can impact the properties. We provide some evidence of the reactivity of the powders. Our results highlight how characterization can be exploited to assess the potential catalytic activity of h-BN for heterogeneous catalysis.






Similar content being viewed by others
References
V. Solozhenko, A. Lazarenko, J.-P. Petitet, and A. Kanaev: Bandgap energy of graphite-like hexagonal boron nitride. J. Phys. Chem. Solids 62, 1331 (2001).
J. Tian, J. Lin, M. Xu, S. Wan, J. Lin, and Y. Wang: Hexagonal boron nitride catalyst in a fixed-bed reactor for exothermic propane oxidation dehydrogenation. Chem. Eng. Sci. 186, 142 (2018).
D.J. Nash, D.T. Restrepo, N.S. Parra, K.E. Giesler, R.A. Penabade, M. Aminpour, D. Le, Z. Li, O.K. Farha, and J.K. Harper: Heterogeneous metal-free hydrogenation over defect-laden hexagonal boron nitride. ACS Omega 1, 1343 (2016).
L. Li, Y. Liu, X. Yang, X. Yu, Y. Fang, Q. Li, P. Jin, and C. Tang: Ambient carbon dioxide capture using boron-rich porous boron nitride: a theoretical study. ACS Appl. Mater. Interfaces 9, 15399 (2017).
J.T. Grant, C.A. Carrero, F. Goeltl, J. Venegas, P. Mueller, S.P. Burt, S.E. Specht, W.P. McDermott, A. Chieregato, and I. Hermans: Selective oxidative dehydrogenation of propane to propene using boron nitride catalysts. Science 354, 1570 (2016).
J.M. Venegas, J.T. Grant, W.P. McDermott, S.P. Burt, J. Micka, C.A. Carrero, and I. Hermans: Selective oxidation of n-butane and isobutane catalyzed by boron nitride. ChemCatChem. 9, 2118 (2017).
J.T. Grant, W.P. McDermott, J.M. Venegas, S.P. Burt, J. Micka, S.P. Phivilay, C.A. Carrero, and I. Hermans: Boron and boron-containing catalysts for the oxidative dehydrogenation of propane. ChemCatChem. 9, 3623 (2017).
V. Fomichev, A. Rudnev, and S. Nemnonov: X-Ray emission bands of transition metals of the first long period. Soviet Physics Solid State 13, 1031 (1971).
L. Carpenter, and P. Kirby: The electrical resistivity of boron nitride over the temperature range 700 degrees C to 1400 degrees C. J. Phys. D: Appl. Phys. 15, 1143 (1982).
D. Akinwande, N. Petrone, and J. Hone: Two-dimensional flexible nanoelectronics. Nat. Commun. 5, 5678 (2014).
K. Watanabe, T. Taniguchi, T. Niiyama, K. Miya, and M. Taniguchi: Far-ultraviolet plane-emission handheld device based on hexagonal boron nitride. Nat. Photonics 3, 591 (2009).
L. Zhong, R.C. Bruno, K. Ethan, L. Ruitao, R. Rahul, T. Humberto, A.P. Marcos, and T. Mauricio: Defect engineering of two-dimensional transition metal dichalcogenides. 2D Materials 3, 022002 (2016).
Q. Peng, J. Crean, A.K. Dearden, C. Huang, X. Wen, S. Bordas and S. De: Defect Engineering of 2D monoatomic-layer materials. Mod. Phys. Lett. B 27, 1330017 (2013).
A. Sajid, J.R. Reimers, and M.J. Ford: Defect states in hexagonal boron nitride: assignments of observed properties and prediction of properties relevant to quantum computation. Phys Rev B. 97, 064101 (2018).
N.L. McDougall, J.G. Partridge, R.J. Nicholls, S.P. Russo, and D.G. McCulloch: Influence of point defects on the near edge structure of hexagonal boron nitride. Phys Rev B. 96, 144106 (2017).
H. Henck, D. Pierucci, Z.B. Aziza, M.G. Silly, B. Gil, F. Sirotti, G. Cassabois, and A. Ouerghi: Stacking fault and defects in single domain multilayered hexagonal boron nitride. Appl. Phys. Lett. 110, 023101 (2017).
B. Berzina, V. Korsaks, L. Trinkler, A. Sarakovskis, J. Grube, and S. Bellucci: Defect-induced blue luminescence of hexagonal boron nitride. Diamond Relat. Mater. 68, 131 (2016).
J. Kroes, A. Fasolino, and M. Katsnelson: Energetics, barriers and vibrational spectra of partially and fully hydrogenated hexagonal boron nitride. Phys. Chem. Chem. Phys. 18, 19359 (2016).
S. Immohr, M. Felderhoff, C. Weidenthaler, and F. Schüth: An orders-of-magnitude increase in the rate of the solid-catalyzed CO oxidation by in situ ball milling. Angew. Chem., Int. Ed. 52, 12688 (2013).
B. Rodriguez, A. Bruckmann, T. Rantanen, and C. Bolm: Solvent-free carbon-carbon bond formations in ball mills. Adv. Synth. Catal. 349, 2213 (2007).
R. Eckert, M. Felderhoff, and F. Schüth: Preferential carbon monoxide oxidation over copper-based catalysts under in situ ball milling. Angew. Chem. 129, 2485 (2017).
V. Molchanov, R. Byanov, and V. Goidin: Use of mechanochemical methods in preparation of supported catalysts. Kinet. Catal. 39, 434 (1998).
V. Zazhigalov, J. Haber, J. Stoch, L. Bogutskaya, and I. Bacherikova: Mechanochemistry as activation method of the VPO catalysts for n-butane partial oxidation. Appl. Catal., A 135, 155 (1996).
J. Ghosh, S. Mazumdar, M. Das, S. Ghatak, and A. Basu: Microstructural characterization of amorphous and nanocrystalline boron nitride prepared by high-energy ball milling. Mater. Res. Bull. 43, 1023 (2008).
J. Huang, X. Jia, H. Yasuda, and H. Mori: Stacking disordering in hexagonal BN induced by shearing under ball milling. Philos. Mag. Lett. 79, 217 (1999).
D. Graf, F. Molitor, K. Ensslin, C. Stampfer, A. Jungen, C. Hierold, and L. Wirtz: Spatially resolved Raman spectroscopy of single-and few-layer graphene. Nano Lett. 7, 238 (2007).
S. Mignuzzi, A.J. Pollard, N. Bonini, B. Brennan, I.S. Gilmore, M.A. Pimenta, D. Richards, and D. Roy: Effect of disorder on Raman scattering of single-layer MoS2. Phys Rev B. 91, 195411 (2015).
N. Kang, H.P. Paudel, M.N. Leuenberger, L. Tetard, and S.I. Khondaker: Photoluminescence quenching in single-layer MoS2 via oxygen plasma treatment. J. Phys. Chem. C. 118, 21258 (2014).
L.H. Li and Y. Chen: Atomically thin boron nitride: unique properties and applications. Adv. Funct. Mater. 26, 2594 (2016).
Y. Kobayashi, K.-I. Fukui, T. Enoki, K. Kusakabe, and Y. Kaburagi: Observation of zigzag and armchair edges of graphite using scanning tunneling microscopy and spectroscopy. Phys Rev B. 71, 193406 (2005).
A. Hashimoto, K. Suenaga, A. Gloter, K. Urita, and S. Iijima: Direct evidence for atomic defects in graphene layers. Nature 430, 870 (2004).
S. Torii, K. Jimura, S. Hayashi, R. Kikuchi, and A. Takagaki: Utilization of hexagonal boron nitride as a solid acid–base bifunctional catalyst. J. Catal. 355, 176 (2017).
B. Fakrach, A. Rahmani, H. Chadli, K. Sbai, M. Bentaleb, J.-L. Bantignies, and J.-L. Sauvajol: Infrared spectrum of single-walled boron nitride nanotubes. Phys Rev B. 85, 115437 (2012).
E. Aradi, S. Naidoo, D. Billing, D. Wamwangi, I. Motochi, and T.E. Derry: Ion beam modification of the structure and properties of hexagonal boron nitride: an infrared and x-ray diffraction study. Nucl. Instrum. Methods Phys. Res., Sect. B 331, 140 (2014).
J.S.M. Nithya and A. Pandurangan: Efficient mixed metal oxide routed synthesis of boron nitride nanotubes. RSC Adv. 4, 26697 (2014).
M.I. Baraton, T. Merle, P. Quintard, and V. Lorenzelli: Surface activity of a boron nitride powder: a vibrational study. Langmuir 9, 1486 (1993).
J. Li, X. Xiao, X. Xu, J. Lin, Y. Huang, Y. Xue, P. Jin, J. Zou, and C. Tang: Activated boron nitride as an effective adsorbent for metal ions and organic pollutants. Sci. Rep. 3, 3208 (2013).
C. Tang, Y. Bando, Y. Huang, C. Zhi, and D. Golberg: Synthetic routes and formation mechanisms of spherical boron nitride nanoparticles. Adv. Funct. Mater. 18, 3653 (2008).
L.H. Li, J. Cervenka, K. Watanabe, T. Taniguchi, and Y. Chen: Strong oxidation resistance of atomically thin boron nitride nanosheets. ACS Nano 8, 1457 (2014).
Y. Liao, K. Tu, X. Han, L. Hu, J.W. Connell, Z. Chen, and Y. Lin: Oxidative etching of hexagonal boron nitride toward nanosheets with defined edges and holes. Sci. Rep. 5, 14510 (2015).
Q. Yang, J. Sha, L. Wang, Y. Zou, J. Niu, C. Cui, and D. Yang: Crystalline boron oxide nanowires on silicon substrate. Physica E 27, 319 (2005).
ACKNOWLEDGMENTS
The authors would like to thank Saint-Gobain Ceramic Materials for the generous donation of hexagonal boron nitride powders used in this study. We acknowledge the NSF support for CHE-1465105 entitled “SusChEM: Defect-laden 2D Catalysts for Carbon Sequestration and Safer Hydrogenation”.
Author information
Authors and Affiliations
Corresponding author
Supplementary materials
Supplementary materials
The supplementary material for this article can be found at https://doi.org/10.1557/mrc.2018.113.
Rights and permissions
About this article
Cite this article
Ding, Y., Torres-Davila, F., Khater, A. et al. Defect engineering in Boron Nitride for catalysis. MRS Communications 8, 1236–1243 (2018). https://doi.org/10.1557/mrc.2018.113
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/mrc.2018.113