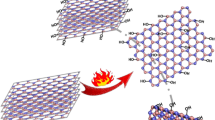
Abstract
The catalytic activity of hexagonal boron nitride (hBN) is not yet well understood, but it seems to be related to hBN hydroxylated sites. A good understanding of those sites and how they are produced is a key step to reveal the true nature of hBN catalytic activity. Here, we report a set of thermal treatments to produce a diversity of hydroxylated sites, as well as a method to remove the boric acid produced by hBN decomposition. We found that some of the boric acid dehydrates on the surface of hBN to produce a borated hBN (hBNO) characterized by an FTIR peat at 1090 cm−1 and an XRD shift due to the variation in the planar distance. We also showed that when characterizing the thermal treated hBN, the storage of the sample changed the borate’s hydration degree and modified both the XRD pattern and the FTIR spectrum. When stored in a humid environment, boric acid peaks are visible, but they are not present if samples are stored in a dry place. The methods established here are a solid basis for the manufacture, purification and characterization of hBN by thermal treatments, showing how the process can be designed to generate various functional groups on its surface.
Graphical abstract











Similar content being viewed by others
References
Shi L, Yan B, Shao D et al (2017) Selective oxidative dehydrogenation of ethane to ethylene over a hydroxylated boron nitride catalyst. Chin J Catal 38:389–396. https://doi.org/10.1016/S1872-2067(17)62786-4
Zhou Y, Lin J, Li L et al (2018) Enhanced performance of boron nitride catalysts with induction period for the oxidative dehydrogenation of ethane to ethylene. J Catal 365:14–24. https://doi.org/10.1016/j.jcat.2018.05.023
Loiland JA, Zhao Z, Patel A, Hazin P (2019) Boron-containing catalysts for the oxidative dehydrogenation of ethane/propane mixtures. Ind Eng Chem Res 58:2170–2181. https://doi.org/10.1021/acs.iecr.8b04906
Huang R, Zhang B, Wang J et al (2017) Direct insight into ethane oxidative dehydrogenation over boron nitrides. ChemCatChem 9:3293–3298. https://doi.org/10.1002/cctc.201700725
Pakdel A, Bando Y, Golberg D (2014) Nano boron nitride flatland. Chem Soc Rev ChemSocRev 43:934–960. https://doi.org/10.1039/c3cs60260e
Shi Z, Wang J, Qiao G, Jin Z (2008) Effects of weak boundary phases (WBP) on the microstructure and mechanical properties of pressureless sintered Al 2O 3/h-BN machinable composites. Mater Sci Eng A 492:29–35. https://doi.org/10.1016/j.msea.2008.03.004
Golberg D, Bando Y, Huang Y et al (2010) Boron nitride nanotubes and nanosheets. Acs Nano ACS Nano 4:2979–2994. https://doi.org/10.1021/nn1006495
Jin H, Li Y, Li X et al (2016) Functionalization of hexagonal boron nitride in large scale by a low-temperature oxidation route. Mater Lett 175:244–248. https://doi.org/10.1016/j.matlet.2016.04.008
Feng Xiao SN, Casillas G, Khan MH, Katkus T, Jiang L, Liu H, Li H, Huang Z (2015) Edge-hydroxylated boron nitride nanosheets as an effective additive to improve the thermal response of hydrogels. Adv Mater 27:7196–7204
Cui Z, Oyer AJ, Glover AJ et al (2014) Large scale thermal exfoliation and functionalization of boron nitride. Small 10:2352–2356. https://doi.org/10.1002/smll.201303236
Nazarov AS, Demin VN, Grayfer ED et al (2012) Functionalization and Dispersion of Hexagonal Boron Nitride (h-BN) Nanosheets Treated with Inorganic Reagents. Chem Asian J. https://doi.org/10.1002/asia.201100710
Bhimanapati GR, Kozuch D, Robinson JA (2014) Large-scale synthesis and functionalization of hexagonal boron nitride nanosheets. Nanoscale Nanoscale 6:11671–11676. https://doi.org/10.1039/c4nr01816h
Weng Q, Wang B, Wang X et al (2014) Highly water-soluble, porous, and biocompatible boron nitrides for anticancer drug delivery. ACS Nano 8:6123–6131. https://doi.org/10.1021/nn5014808
Venegas JM, Mcdermott WP, Hermans I (2018) Serendipity in catalysis research: boron-based materials for alkane oxidative dehydrogenation. Acc Chem Res 51:2556. https://doi.org/10.1021/acs.accounts.8b00330
Fu L, Wang T, Yu J et al (2017) An ultrathin high-performance heat spreader fabricated with hydroxylated boron nitride nanosheets. 2D Mater. https://doi.org/10.1088/2053-1583/aa636e
Venegas JM, Grant JT, McDermott WP et al (2017) Selective oxidation of n-butane and isobutane catalyzed by boron nitride. ChemCatChem 9:2118–2128. https://doi.org/10.1002/cctc.201601686
Liao Y, Tu K, Han X et al (2015) Oxidative etching of hexagonal boron nitride toward nanosheets with defined edges and holes. Sci Rep 5:14510. https://doi.org/10.1038/srep14510
Zhan Y, Yan J, Wu M et al (2017) Boron nitride nanosheets as a platform for fluorescence sensing. Talanta 174:365–372. https://doi.org/10.1016/j.talanta.2017.06.032
M. Sudeep P, Vinod S, Ozden S, et al (2015) Functionalized boron nitride porous solids. RSC Adv 5:93964–93969. https://doi.org/10.1039/C5RA19091F
Lei Z, Xu S, Wan J, Wu P (2015) Facile preparation and multifunctional applications of boron nitride quantum dots. Nanoscale 7:18902–18908. https://doi.org/10.1039/C5NR05960G
Yu Z, Hou X, Chen Z, Chou K-C (2013) Effect of water-vapor content on reaction rate of hexagonal BN powder at 1273 K. High Temp Mater Process 32:275–281. https://doi.org/10.1515/htmp-2012-0138
Aghili S, Panjepour M, Meratian M (2018) Kinetic analysis of formation of boron trioxide from thermal decomposition of boric acid under non-isothermal conditions. J Therm Anal Calorim 131:2443–2456. https://doi.org/10.1007/s10973-017-6740-3
Gautam C, Yadav AK, Singh AK (2012) A Review on infrared spectroscopy of borate glasses with effects of different additives. ISRN Ceram. https://doi.org/10.5402/2012/428497
Shi L, Wang Y, Yan B et al (2018) Progress in selective oxidative dehydrogenation of light alkanes to olefins promoted by boron nitride catalysts. Chem Commun 54:10936–10947. https://doi.org/10.1039/c8cc04604b
Sibeijn M, Vanveen J, Bliek A, Moulijn J (1994) On the nature and formation of the active-sites in re2o7 metathesis catalysts supported on borated alumina. J Catal 145:416–429. https://doi.org/10.1006/jcat.1994.1052
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Yaroslava Yingling.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Garro Mena, L., Hohn, K.L. Modification of hexagonal boron nitride by thermal treatment. J Mater Sci 56, 7298–7307 (2021). https://doi.org/10.1007/s10853-020-05719-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-020-05719-8