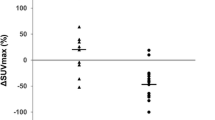
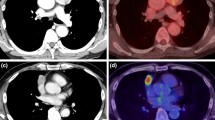
Abstract
Background
Preoperative fluorine-18-fluorodeoxyglucose positron emission tomography (18F-FDG PET) of thymic epithelial tumors (TETs) is well known for identifying malignant-grade TETs; however, its predictive power for determining locally advanced tumors, lymph node (LN) metastasis, and prognosis remains unknown.
Patients and Methods
We retrospectively evaluated patients with resectable TETs who were preoperatively assessed using 18F-FDG PET from January 2012 to January 2023. The receiver operating characteristic curve was used to evaluate the cutoff value of the maximum standardized uptake value (SUVmax) to predict advanced-stage disease. Recurrence/progression-free survival (RFS/PFS) was analyzed using the Kaplan–Meier method. The staging was classified according to the tumor–node–metastasis system.
Results
Our study included 177 patients; 145 (81.9%) had pathological early-stage TET (stage I or II), and 32 (19.1%) had advanced stage (stage III or IV). The area under the curve value for predicting the advanced stage was 0.903, and the cutoff value was 5.6 (sensitivity 81.3%, specificity 84.8%). SUVmax > 5.6 was associated with worse prognosis for RFS/PFS. LN metastasis was preoperatively detected by FDG uptake in 30.8% of patients with pathological LN positivity, whereas LN metastasis was not pathologically detected in patients with SUVmax < 5.9. In patients with advanced-stage TETs, LN recurrence was more frequent in patients who were preoperatively detected by 18F-FDG PET than those who were not (75.0% versus 7.1%).
Conclusions
18F-FDG PET is a potentially valuable tool for predicting advanced stage and poor prognosis of recurrence in patients with TETs. SUVmax can help thoracic surgeons to guide them in selecting appropriate therapeutic strategies for TETs.


Similar content being viewed by others
References
National Comprehensive Cancer Network. NCCN guidelines for treatment of cancer by site: Thymomas and Thymic Carcinomas; version 1.2023. 2022.https://www.nccn.org/professionals/physician_gls/pdf/thymic.pdf (2023. 3 March 2023, date last accessed).
Zhang Y, Li Z, Chen Y, et al. Induction strategy for locally advanced thymoma. Front Oncol. 2021;11:704220.
Kuriyama S, Imai K, Ishiyama K, et al. Using CT to evaluate mediastinal great vein invasion by thymic epithelial tumors: measurement of the interface between the tumor and neighboring structures. Eur Radiol. 2022;32(3):1891–901.
Ruffini E, Rami-Porta R, Huang J, et al. The International Association for the Study of Lung Cancer Thymic Epithelial Tumor Staging Project: unresolved issues to be addressed for the next ninth edition of the TNM classification of malignant tumors. J Thorac Oncol. 2022;17(6):838–51.
Kim K, Jeong JH, Kim SJ. Diagnostic test accuracy of 18F-FDG PET or PET/CT for characterization of histologic type of thymic epithelial tumor: a meta-analysis. Clin Nucl Med. 2022;47(1):36–42.
Nakagawa K, Takahashi S, Endo M, Ohde Y, Kurihara H, Terauchi T. Can (18)F-FDG PET predict the grade of malignancy in thymic epithelial tumors? An evaluation of only resected tumors. Cancer Manag Res. 2017;9:761–8.
Sugawara H, Ito K, Watanabe H, et al. Clinical usefulness of PET/MRI in differentiating anterior mediastinal masses. Nucl Med Commun. 2022;43(1):92–9.
Akamine T, Nakagawa K, Ito K, et al. Role of 18F-FDG PET in selecting candidates for a minimally invasive approach for thymic epithelial tumour resection. Interdiscip CardioVasc Thorac Surg. 2023;36(5):ivad082.
Han S, Kim YI, Oh JS, et al. Diagnostic and prognostic values of 2-[(18)F]FDG PET/CT in resectable thymic epithelial tumour. Eur Radiol. 2022;32(2):1173–83.
Detterbeck FC, Stratton K, Giroux D, et al. The IASLC/ITMIG Thymic Epithelial Tumors Staging Project: proposal for an evidence-based stage classification system for the forthcoming (8th) edition of the TNM classification of malignant tumors. J Thorac Oncol. 2014;9(9 Suppl 2):S65-72.
Okumura M, Yoshino I, Yano M, et al. Tumour size determines both recurrence-free survival and disease-specific survival after surgical treatment for thymoma. Eur J Cardiothorac Surg. 2019;56(1):174–81.
Okumura M, Ohta M, Tateyama H, et al. The World Health Organization histologic classification system reflects the oncologic behavior of thymoma. Cancer. 2002;94(3):624–32.
Araujo-Filho JAB, Mayoral M, Zheng J, et al. CT radiomic features for predicting resectability and TNM staging in thymic epithelial tumors. Ann Thorac Surg. 2022;113(3):957–65.
Fukui T, Fukumoto K, Okasaka T, et al. Prognostic impact of tumour size in completely resected thymic epithelial tumours. Eur J Cardiothorac Surg. 2016;50(6):1068–74.
Wang ZM, Li F, Sarigul L, et al. A predictive model of lymph node metastasis for thymic epithelial tumours. Eur J Cardiothorac Surg. 2022;62(5):ezac210.
Hwang Y, Park IK, Park S, Kim ER, Kang CH, Kim YT. Lymph node dissection in thymic malignancies: implication of the ITMIG lymph node map, TNM stage classification, and recommendations. J Thorac Oncol. 2016;11(1):108–14.
Acknowledgement
We thank Editage (www.editage.cn) for English language editing.
Funding
This work was not supported by any funding sources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosue
Masahiko Kusumoto received honoraria for lecture fees from AstraZeneca K.K., Daiichi-Sankyo Inc., and Daiichi-Sankyo Co., Ltd. and a research grant from Canon medical systems corporation. The other authors declare no conflicts of interest associated with this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10434_2023_14328_MOESM2_ESM.tif
Supplementary FIG. 2. Sankey diagram of the clinical and pathological TNM staging. Twelve patients were pathologically upstaged to an advanced stage from clinical stage I. Nine patients with cN0 disease had pathological lymph node metastasis
10434_2023_14328_MOESM3_ESM.tif
Supplementary FIG. 3. Kaplan–Meier curves of recurrence/progression-free survival according to SUVmax in patients with pathologically early-stage (A) and advanced-stage (B) thymic epithelial tumors
10434_2023_14328_MOESM4_ESM.tif
Supplementary FIG. 4. Kaplan–Meier curves of recurrence/progression-free survival according to pathological tumor-node-metastasis staging
10434_2023_14328_MOESM5_ESM.tif
Supplementary FIG. 5. Kaplan–Meier curves of recurrence/progression-free survival in patients with pN positive according to cN0 and cN1 determined by 18F-FDG PET
10434_2023_14328_MOESM6_ESM.tif
Supplementary FIG. 6. Receiver operating characteristic curve of SUVmax in predicting advanced-stage disease in PET/CT and PET/MRI
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akamine, T., Nakagawa, K., Ito, K. et al. Impact of 18F-FDG PET on TNM Staging and Prognosis in Thymic Epithelial Tumors. Ann Surg Oncol 31, 192–200 (2024). https://doi.org/10.1245/s10434-023-14328-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-14328-z