Abstract
Background
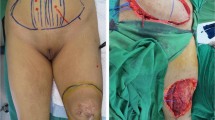
Dermatofibrosarcoma protuberans (DFSP) is a dermal sarcoma often diagnosed by excision biopsy, and is often incompletely excised, with high recurrence rates. Traditional wide excision involves resection margins of 2–4 cm, often resulting in morbid procedures requiring surgical reconstruction. An alternative is conservative re-excision (CRE), which results in narrower margins and less-frequent reconstruction. The aim of this study is to assess the effectiveness of CRE in providing local control.
Patients and Methods
A retrospective review of patients treated for DFSP at a tertiary sarcoma centre over a 10-year period.
Results
Ninety-eight patients were analysed. Median follow-up was 53 months. Fifty-four patients had microscopically incompletely excised DFSP, and of these, 41 underwent CRE of DFSP scar. Seven (17.1%) patients required more than one CRE to achieve negative margins. The mean width of CRE was 15.4 mm. Fifty-four patients had resection of intact tumours, with 19 (35.2%) requiring surgical reconstruction. One patient (1%) developed local recurrence, and one patient (1%) distant recurrence—both of these patients had high-grade fibrosarcomatous DFSP. No patient with classical DFSP who had clear margins sustained recurrence, regardless of whether their surgery was CRE of scar or wide excision of tumour.
Conclusions
CRE is a safe and acceptable alternative to traditional wide excision, with no patients developing local recurrence (LR). CRE results in low rates of surgical reconstruction, and hence lower morbidity; this is partially offset by the higher rates of inadequate excision requiring further surgery. However, the lesser rate of inadequate excision compared with rates of reconstruction makes CRE an attractive option.
Similar content being viewed by others
References
Lemm D, Mugge LO, Mentzel T, Hoffken K. Current treatment options in dermatofibrosarcoma protuberans. J Cancer Res Clin Oncol. 2009;135(5):653–65.
Huis In ‘t Veld EA, van Coevorden F, Grunhagen DJ, et al. Outcome after surgical treatment of dermatofibrosarcoma protuberans: is clinical follow-up always indicated? Cancer. 2019;125(5):735–41.
Mentzel T, Beham A, Katenkamp D, Dei Tos AP, Fletcher CD. Fibrosarcomatous (“high-grade”) dermatofibrosarcoma protuberans: clinicopathologic and immunohistochemical study of a series of 41 cases with emphasis on prognostic significance. Am J Surg Pathol. 1998;22(5):576–87.
Hayakawa K, Matsumoto S, Ae K, et al. Risk factors for distant metastasis of dermatofibrosarcoma protuberans. J Orthop Traumatol. 2016;17(3):261–6.
Bogucki B, Neuhaus I, Hurst EA. Dermatofibrosarcoma protuberans: a review of the literature. Dermatol Surg. 2012;38(4):537–51.
Dangoor A, Seddon B, Gerrand C, Grimer R, Whelan J, Judson I. UK guidelines for the management of soft tissue sarcomas. Clin Sarcoma Res. 2016;6:20.
Bichakjian CK ea. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®), Dermatofibrosarcoma Protuberans, Version 1.2019—August 31, 2018. https://www.nccn.org/professionals/physician_gls/pdf/dfsp.pdf.
Lindner NJ, Scarborough MT, Powell GJ, Spanier S, Enneking WF. Revision surgery in dermatofibrosarcoma protuberans of the trunk and extremities. Eur J Surg Oncol. 1999;25(4):392–7.
Khatri VP, Galante JM, Bold RJ, Schneider PD, Ramsamooj R, Goodnight JE, Jr. Dermatofibrosarcoma protuberans: reappraisal of wide local excision and impact of inadequate initial treatment. Ann Surg Oncol. 2003;10(9):1118–22.
Fiore M, Miceli R, Mussi C, et al. Dermatofibrosarcoma protuberans treated at a single institution: a surgical disease with a high cure rate. J Clin Oncol. 2005;23(30):7669–75.
Monnier D, Vidal C, Martin L, et al. Dermatofibrosarcoma protuberans: a population-based cancer registry descriptive study of 66 consecutive cases diagnosed between 1982 and 2002. J Eur Acad Dermatol Venereol. 2006;20(10):1237–42.
Farma JM, Ammori JB, Zager JS, et al. Dermatofibrosarcoma protuberans: How wide should we resect? Ann Surg Oncol. 2010;17(8):2112–8.
Paradisi A, Abeni D, Rusciani A, et al. Dermatofibrosarcoma protuberans: wide local excision vs. Mohs micrographic surgery. Cancer Treat Rev. 2008;34(8):728–36.
Meguerditchian AN, Wang J, Lema B, Kraybill WG, Zeitouni NC, Kane JM, 3rd. Wide excision or Mohs micrographic surgery for the treatment of primary dermatofibrosarcoma protuberans. Am J Clin Oncol. 2010;33(3):300–3.
Foroozan M, Sei JF, Amini M, Beauchet A, Saiag P. Efficacy of Mohs micrographic surgery for the treatment of dermatofibrosarcoma protuberans: systematic review. Arch Dermatol. 2012;148(9):1055–63.
Loghdey MS, Varma S, Rajpara SM, Al-Rawi H, Perks G, Perkins W. Mohs micrographic surgery for dermatofibrosarcoma protuberans (DFSP): a single-centre series of 76 patients treated by frozen-section Mohs micrographic surgery with a review of the literature. J Plast Reconstr Aesthet Surg. 2014;67(10):1315–21.
Smith-Zagone MJ, Schwartz MR. Frozen section of skin specimens. Arch Pathol Lab Med. 2005;129(12):1536–43.
Bowne WB, Antonescu CR, Leung DH, et al. Dermatofibrosarcoma protuberans: a clinicopathologic analysis of patients treated and followed at a single institution. Cancer. 2000;88(12):2711–20.
Hoesly PM, Lowe GC, Lohse CM, Brewer JD, Lehman JS. Prognostic impact of fibrosarcomatous transformation in dermatofibrosarcoma protuberans: a cohort study. J Am Acad Dermatol. 2015;72(3):419–25.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors have no disclosures to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Snow, H., Davies, E., Strauss, D.C. et al. Conservative Re-excision is a Safe and Simple Alternative to Radical Resection in Revision Surgery for Dermatofibrosarcoma Protuberans. Ann Surg Oncol 27, 919–923 (2020). https://doi.org/10.1245/s10434-019-08011-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-08011-5