Abstract
Background
MicroRNAs have roles in the regulation of the epithelial–mesenchymal transition (EMT). Findings have shown that miR-506 inhibits the expression of SNAI2 and that low expression of miR-506 is associated with poor prognoses in ovarian and breast cancers. This study investigated the role of miR-506 in survival and the EMT in patients with gastric cancer.
Methods
In this study, miR-506 and SNAI2 mRNA levels were measured in 141 cases of gastric cancer by quantitative reverse transcription polymerase chain reaction, and the protein expressions of SNAI2 and E-cadherin in 39 cases were validated by immunohistochemical analysis. Next, the associations between their expression levels and clinicopathologic factors were evaluated. In addition, cell proliferation, migration, and luciferase activity of the 3′ untranslated region (UTR) of SNAI2 were analyzed using pre-miR-506 precursor in two human gastric cancer cell lines.
Results
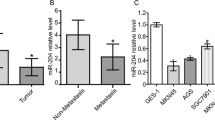
Low expression of miR-506 was significantly correlated with poor overall survival in both the univariate analysis (P = 0.016) and the multivariate analysis (P < 0.05). Low miR-506 expression was significantly correlated with high SNAI2 expression (P = 0.009) and poorly differentiated type (P = 0.015). In vitro, miR-506 suppressed SNAI2 expression by binding to its 3′UTR, resulting in increased expression of E-cadherin (P < 0.05), verified by immunohistochemical analysis. Pre-miR-506 transfected cells showed significantly suppressed cell proliferation and migration (P < 0.05) compared with the control cells.
Conclusions
The EMT was directly suppressed by miR-506, and its low expression was an independent prognostic factor in gastric cancer patients. The data indicated that miR-506 may act as a tumor suppressor and could be a novel therapeutic agent.




Similar content being viewed by others
References
IARC. GLOBOCAN 2012: Estimated cancer incidence, mortality and prevalence worldwide in 2012. Retrieved 10 January 2014 at http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx.
Hohenberger P, Gretschel S. Gastic cancer. Lancet. 2003; 362:305–15.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial–mesenchymal transitions in development and disease. Cell. 2009;139:871–90.
Thiery JP. Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442–54.
Katoh M. Epithelial–mesenchymal transition in gastric cancer (review). Int J Oncol. 2005;27:1677–83.
Murai T, Yamada S, Fuchs BC, et al. Epithelial-to-mesenchymal transition predicts prognosis in clinical gastric cancer. J Surg Oncol. 2014;109:684–9.
Kaurah P, MacMillan A, Boyd N, et al. Founder and recurrent CDH1 mutations in families with hereditary diffuse gastric cancer. J Am Med Assoc. 2007;297:2360–72.
Bhayani MK, Calin GA, Lai SY. Functional relevance of miRNA sequences in human disease. Mutat Res. 2012;731:14–9.
Alvarez-Garcia I, Miska EA. MicroRNA functions in animal development and human disease. Development. 2005;132:4653–62.
Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857–66.
Bouyssou JM, Manier S, Huynh D, Issa S, Roccaro AM, Ghobrial IM. Regulation of microRNAs in cancer metastasis. Biochim Biophys Acta. 2014;1845:255–65.
Song F, Yang D, Liu B, et al. Integrated microRNA network analyses identify a poor prognosis subtype of gastric cancer characterized by the miR-200 family. Clin Cancer Res. 2014;20:878–89.
Duarte I, Llanos O. Patterns of metastases in intestinal and diffuse types of carcinoma of the stomach. Hum Pathol. 1981;12:237–42.
Maehara Y, Moriguchi S, Kakeji Y, et al. Pertinent risk factors and gastric carcinoma with synchronous peritoneal dissemination or liver metastasis. Surgery. 1991;110:820–3.
Okugawa Y, Inoue Y, Tanaka K, et al. Smad interacting protein 1 (SIP1) is associated with peritoneal carcinomatosis in intestinal type gastric cancer. Clin Exp Metastasis. 2013;30:417–29.
Okugawa Y, Toiyama Y, Tanaka K, et al. Clinical significance of zinc finger E-box binding homeobox 1 (ZEB1) in human gastric cancer. J Surg Oncol. 2012; 106:280–5.
Yang D, Sun Y, Hu L, et al. Integrated analyses identify a master microRNA regulatory network for the mesenchymal subtype in serous ovarian cancer. Cancer Cell. 2013;23:186–99.
Arora H, Qureshi R, Park WY. miR-506 regulates epithelial mesenchymal transition in breast cancer cell lines. PloS One. 2013;8:e64273.
Mori M, Mimori K, Yoshikawa Y, et al. Analysis of the gene-expression profile regarding the progression of human gastric carcinoma. Surgery. 2002;131:S39–47.
Ieta K, Ojima E, Tanaka F, et al. Identification of overexpressed genes in hepatocellular carcinoma, with special reference to ubiquitin-conjugating enzyme E2C gene expression. Int J Cancer. 2007;121:33–8.
Mizuno H, Kitada K, Nakai K, Sarai A. PrognoScan: a new database for meta-analysis of the prognostic value of genes. BMC Med Genomics. 2009;2:18.
Akagawa S, Ohuchida K, Torata N, et al. Peritoneal myofibroblasts at metastatic foci promote dissemination of pancreatic cancer. Int J Oncol. 2014;45:113–20.
Miyake S, Kitajima Y, Nakamura J, et al. HIF-1alpha is a crucial factor in the development of peritoneal dissemination via natural metastatic routes in scirrhous gastric cancer. Int J Oncol. 2013;43:1431–40.
Berx G, Van Roy F. The E-cadherin/catenin complex: an important gatekeeper in breast cancer tumorigenesis and malignant progression. Breast Cancer Res. 2001;3:289–93.
Paredes J, Figueiredo J, Albergaria A, et al. Epithelial E- and P-cadherins: role and clinical significance in cancer. Biochim Biophys Acta. 2012;1826:297–311.
Yin M, Ren X, Zhang X, et al. Selective killing of lung cancer cells by miRNA-506 molecule through inhibiting NF-kappaB p65 to evoke reactive oxygen species generation and p53 activation. Oncogene. 2014. doi: 10.1038/onc.2013.597.
Wen SY, Lin Y, Yu YQ, et al. miR-506 acts as a tumor suppressor by directly targeting the hedgehog pathway transcription factor Gli3 in human cervical cancer. Oncogene. 2014. doi:10.1038/onc.2014.9.
Liu G, Sun Y, Ji P, et al. MiR-506 suppresses proliferation and induces senescence by directly targeting the CDK4/6-FOXM1 axis in ovarian cancer. J Pathol. 2014;233:308–18.
Guo F, Parker Kerrigan BC, Yang D, et al. Posttranscriptional regulatory network of epithelial-to-mesenchymal and mesenchymal-to-epithelial transitions. J Hematol Oncol. 2014;7:19.
Liu Z, Chen L, Zhang X, et al. RUNX3 regulates vimentin expression via miR-30a during epithelial–mesenchymal transition in gastric cancer cells. J Cell Mol Med. 2014;18:610–23.
Maier HJ, Schmidt-Strassburger U, Huber MA, Wiedemann EM, Beug H, Wirth T. NF-kappaB promotes epithelial–mesenchymal transition, migration, and invasion of pancreatic carcinoma cells. Cancer Lett. 2010;295:214–28.
Takeshita F, Patrawala L, Osaki M, et al. Systemic delivery of synthetic microRNA-16 inhibits the growth of metastatic prostate tumors via downregulation of multiple cell-cycle genes. Mol Ther. 2010;18:181–7.
Lanford RE, Hildebrandt-Eriksen ES, Petri A, et al. Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science. 2010; 327:198–201.
Acknowledgment
We thank K. Oda, M. Kasagi, S. Kohno, T. Kawano, and M. Aoyagi for their technical assistance. This work was supported in part by Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (Grant numbers 21591644, 21791295, 21791297, 215921014, and 21679006).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sakimura, S., Sugimachi, K., Kurashige, J. et al. The miR-506-Induced Epithelial–Mesenchymal Transition is Involved in Poor Prognosis for Patients with Gastric Cancer. Ann Surg Oncol 22 (Suppl 3), 1436–1443 (2015). https://doi.org/10.1245/s10434-015-4418-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4418-2