Abstract
Introduction
Malignant phyllodes tumors are rare fibroepithelial breast neoplasms. Appropriate surgical management remains a subject of debate. The purpose of our study was to define optimal surgical treatment and to identify factors associated with outcome.
Methods
After confirmatory pathology review, we identified 67 patients with borderline (n = 15) and malignant (n = 52) phyllodes tumors treated at our institution between 1971 and 2008. We used Cox proportional hazards models to evaluate associations between treatment, patient and tumor characteristics, and disease-free (DFS) and cancer-specific survival (CSS).
Results
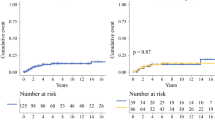
Median patient age was 47 years. For 32 patients, definitive surgical treatment was wide local excision (WLE): 27 with margins ≥1 cm and 5 with margins <1 cm. Thirty-five underwent mastectomy. Two patients received radiotherapy after WLE and two after mastectomy with microscopically positive margins. After 10 years median follow-up, 16 patients (24 %) recurred locally (8 postmastectomy and 8 after WLE). Treatment type and margin extent did not impact local recurrence. Fifteen patients (22 %) developed distant disease. Overall 5-year DFS was 67.9 % and CSS 80.1 %. Tumor size >5 cm, mitotic rate ≥10/10 HPF, stromal overgrowth and cellularity (all p < 0.05) predicted DFS, whereas CSS was associated with the latter three variables. CSS was diminished for mastectomy patients who were significantly more likely to harbor tumors with adverse features.
Conclusions
With long-term follow-up, extent of surgical resection did not affect DFS for patients with borderline and malignant phyllodes tumors. Tumor features, most notably stromal overgrowth, were predictive of recurrence and survival, suggesting these high-risk patients may benefit from additional therapeutic strategies.

Similar content being viewed by others
References
Kapiris I, Nasiri N, A’Hern R, Healy V, Gui GP. Outcome and predictive factors of local recurrence and distant metastases following primary surgical treatment of high-grade malignant phyllodes tumours of the breast. Eur J Surg Oncol. 2001;27:723–730.
Asoglu O, Ugurlu MM, Blanchard K, Grant CS, Reynolds C, Cha SS, Donohue JH. Risk factors for recurrence and death after primary surgical treatment of malignant phyllodes tumors. Ann Surg Oncol. 2004;11:1011–1017.
Reinfuss M, Mitus J, Duda K, Stelmach A, Rys J, Smolak K. The treatment and prognosis of patients with phyllodes tumor of the breast: an analysis of 170 cases. Cancer. 1996;77:910–916.
Belkacemi Y, Bousquet G, Marsiglia H, et al. Phyllodes tumor of the breast. Int J Radiat Oncol Biol Phys. 2008;70:492–500.
Barth RJ Jr, Wells WA, Mitchell SE, Cole BF. A prospective, multi-institutional study of adjuvant radiotherapy after resection of malignant phyllodes tumors. Ann Surg Oncol. 2009;16:2288–2294.
Tan PH, Thike AA, Tan WJ, et al. Predicting clinical behaviour of breast phyllodes tumours: a nomogram based on histological criteria and surgical margins. J Clin Pathol. 2012;65:69–76.
Barth RJ Jr. Histologic features predict local recurrence after breast conserving therapy of phyllodes tumors. Breast Cancer Res Treat. 1999;57:291–295.
International Agency for Research on Cancer. In: Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ (eds.) WHO Classification of Tumours of the Breast, 4th ed. Geneva: World Health Organization; 2012.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–381.
Fou A, Schnabel FR, Hamele-Bena D, et al. Long-term outcomes of malignant phyllodes tumors patients: an institutional experience. Am J Surg. 2006;192:492–495.
National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Breast Cancer Version 3 2014. http://www.nccn.org/professionals/physician_gls/pdf/breast.pdf (2014). Accessed 4 April 2014.
Macdonald OK, Lee CM, Tward JD, Chappel CD, Gaffney DK. Malignant phyllodes tumor of the female breast: association of primary therapy with cause-specific survival from the Surveillance, Epidemiology, and End Results (SEER) program. Cancer. 2006;107:2127–2133.
Chaney AW, Pollack A, McNeese MD, Zagars GK, Pisters PW, Pollock RE, Hunt KK. Primary treatment of cystosarcoma phyllodes of the breast. Cancer. 2000;89:1502–1511.
Mangi AA, Smith BL, Gadd MA, Tanabe KK, Ott MJ, Souba WW. Surgical management of phyllodes tumors. Arch Surg. 1999;134:487–492; discussion 492–493.
Barrio AV, Clark BD, Goldberg JI, et al. Clinicopathologic features and long-term outcomes of 293 phyllodes tumors of the breast. Ann Surg Oncol. 2007;14:2961–2970.
Spitaleri G, Toesca A, Botteri E, et al. Breast phyllodes tumor: a review of literature and a single center retrospective series analysis. Crit Rev Oncol/Hematol. 2013;88:427–436.
Pandey M, Mathew A, Kattoor J, Abraham EK, Mathew BS, Rajan B, Nair KM. Malignant phyllodes tumor. Breast J. 2001;7:411–416.
August DA, Kearney T. Cystosarcoma phyllodes: mastectomy, lumpectomy, or lumpectomy plus irradiation. Surg Oncol. 2000;9:49–52.
Salvadori, B, Cusumano F, Del Bo R, et al. Surgical treatment of phyllodes tumors of the breast. Cancer. 1989; 63:2532–2536.
Gnerlich JL, Williams RT, Yao K, Jaskowiak N, Kulkami SA. Utilization of radiotherapy for malignant phyllodes tumors: analysis of the national cancer data base, 1998-2009. Ann Surg Oncol. 2014;21(4):1222–1230.
Disclosure
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Onkendi, E.O., Jimenez, R.E., Spears, G.M. et al. Surgical Treatment of Borderline and Malignant Phyllodes Tumors: The Effect of the Extent of Resection and Tumor Characteristics on Patient Outcome. Ann Surg Oncol 21, 3304–3309 (2014). https://doi.org/10.1245/s10434-014-3909-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3909-x