Abstract
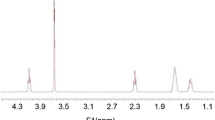
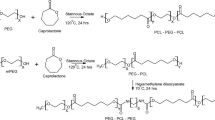
Supramolecular hydrogels formed by cyclodextrins and polymers have been widely investigated as a biocompatible, biodegradable and controllable drug delivery system. In this study, a supramolecular hydrogel based on biodegradable poly(caprolactone)–poly(ethylene glycol)–poly(caprolactone) (PCL-PEG-PCL) triblock copolymers and γ-cyclodextrin (γ-CD) was prepared through inclusion complexation as an injectable, sustained-release vehicle for insulin. The triblock copolymer PCL-PEG-PCL was synthesised by the ring-opening polymerisation method, using microwave irradiation. The polymerisation reaction and the copolymer structures were evaluated by nuclear magnetic resonance (NMR) and gel permeation chromatography (GPC). The supramolecular hydrogel was prepared in aqueous solution by blending an aqueous γ-CD solution with an aqueous solution of PCL-PEG-PCL triblock copolymer at room temperature. In vitro insulin release through the hydrogel system was studied. The relative surface hydrophobicity of standard and released insulin from the SMGel was estimated using 8-anilino-1-naphthalene sulfonic acid (ANS). Results of 1HNMR and gel permeation chromatography revealed that microwave irradiation is a simple and reliable method for synthesis of PCL-PEG-PCL copolymer. Gelation occurred within a minute. The supramolecular hydrogel obtained by mixing 10.54% (w/v) γ-CD and 2.5% (w/v) copolymer had an excellent syringeability. Insulin was released up to 80% over a period of 20 days. Insulin kept its initial folding after formulating and releasing from SMGel. A supramolecular hydrogel based on complexation of triblock PCL-PEG-PCL copolymer with γ-cyclodextrin is a suitable system for providing sustained release of therapeutic proteins, with desirable flow behaviour.









Similar content being viewed by others
References
Li J. Self-assembled supramolecular hydrogels based on polymer–cyclodextrin inclusion complexes for drug delivery. NPG Asia Mater. 2010;2(3):112–8.
Bromberg LE, Ron ES. Temperature-responsive gels and thermogelling polymer matrices for protein and peptide delivery. Adv Drug Deliv Rev. 1998;31(3):197–221.
Bender ML, Komiyama M. Cyclodextrin chemistry. Berlin: Springer; 1978.
Harada A, Li J, Kamachi M. Double-stranded inclusion complexes of cyclodextrin threaded on poly (ethylene glycol). Nature. 1994;370(6485):126–8.
Li J, Harada A, Kamachi M. Sol–gel transition during inclusion complex formation between α-cyclodextrin and high molecular weight poly (ethylene glycol) s in aqueous solution. Polym J. 1994;26(9):1019–26.
Li J, Ni X, Leong KW. Injectable drug-delivery systems based on supramolecular hydrogels formed by poly (ethylene oxide) s and α-cyclodextrin. J Biomed Mater Res A. 2003;65(2):196–202.
Harada A, Li J, Kamachi M. The molecular necklace: a rotaxane containing many threaded α-cyclodextrins. Nature. 1992;356(6367):325–7.
Li J, Li X, Toh KC, Ni X, Zhou Z, Leong KW. Inclusion complexation and formation of polypseudorotaxanes between poly [(ethylene oxide)-r an-(propylene oxide)] and cyclodextrins. Macromolecules. 2001;34(26):8829–31.
Huh KM, Cho YW, Chung H, Kwon IC, Jeong SY, Ooya T, et al. Supramolecular hydrogel formation based on inclusion complexation between poly (ethylene glycol)-modified chitosan and α-cyclodextrin. Macromol Biosci. 2004;4(2):92–9.
Higashi T, Hirayama F, Misumi S, Arima H, Uekama K. Design and evaluation of polypseudorotaxanes of pegylated insulin with cyclodextrins as sustained release system. Biomaterials. 2008;29(28):3866–71.
Ma D, Zhang H-B, Chen D-H, Zhang L-M. Novel supramolecular gelation route to in situ entrapment and sustained delivery of plasmid DNA. J Colloid Interface Sci. 2011;364(2):566–73.
Prabaharan M, Mano J. Chitosan derivatives bearing cyclodextrin cavitiesas novel adsorbent matrices. Carbohydr Polym. 2006;63(2):153–66.
Ganta S, Devalapally H, Shahiwala A, Amiji M. A review of stimuli-responsive nanocarriers for drug and gene delivery. J Control Release. 2008;126(3):187–204.
Li J, Li X, Ni X, Wang X, Li H, Leong KW. Self-assembled supramolecular hydrogels formed by biodegradable PEO–PHB–PEO triblock copolymers and α-cyclodextrin for controlled drug delivery. Biomaterials. 2006;27(22):4132–40.
Vyas A, Saraf S, Saraf S. Cyclodextrin based novel drug delivery systems. J Incl Phenom Macrocycl Chem. 2008;62(1–2):23–42.
Yu J, Fan H, Huang J, Chen J. Fabrication and evaluation of reduction-sensitive supramolecular hydrogel based on cyclodextrin/polymer inclusion for injectable drug-carrier application. Soft Matter. 2011;7(16):7386–94.
Harries M, Smith I. The development and clinical use of trastuzumab (Herceptin). Endocr-Relat Cancer. 2002;9(2):75–85.
Kerbel R, Folkman J. Clinical translation of angiogenesis inhibitors. Nat Rev Cancer. 2002;2(10):727–39.
Marshall H. Anti-CD20 antibody therapy is highly effective in the treatment of follicular lymphoma. Trends Immunol. 2001;22(4):183–4.
McCarthy AA. New approaches to diabetes disease control, insulin delivery, and monitoring. Chem Biol. 2004;11(12):1597–8.
Banting F, Best CH, Collip JB, Campbell WR, Fletcher AA. Pancreatic extracts in the treatment of diabetes mellitus. Can Med Assoc J. 1922;12(3):141.
Onuki Y, Morishita M, Takayama K. Formulation optimization of water-in-oil-water multiple emulsion for intestinal insulin delivery. J Control Release. 2004;97(1):91–9.
Takenaga M, Yamaguchi Y, Kitagawa A, Ogawa Y, Kawai S, Mizushima Y, et al. Optimum formulation for sustained-release insulin. Int J Pharm. 2004;271(1):85–94.
Takenaga M, Yamaguchi Y, Kitagawa A, Ogawa Y, Mizushima Y, Igarashi R. A novel sustained-release formulation of insulin with dramatic reduction in initial rapid release. J Control Release. 2002;79(1):81–91.
Hoare T, Pelton R. Charge-switching, amphoteric glucose-responsive microgels with physiological swelling activity. Biomacromolecules. 2008;9(2):733–40.
Lin C-C, Metters AT. Hydrogels in controlled release formulations: network design and mathematical modeling. Adv Drug Deliv Rev. 2006;58(12):1379–408.
van Dijk‐Wolthuis W, Van Steenbergen M, Underberg W, Hennink W. Degradation kinetics of methacrylated dextrans in aqueous solution. J Pharm Sci. 1997;86(4):413–7.
Irie T, Uekama K. Pharmaceutical applications of cyclodextrins. III. Toxicological issues and safety evaluation. J Pharm Sci. 1997;86(2):147–62.
Khodaverdi A, Akbari A, Tekie FSM, Mohajeri SA, Zohuri G, Hadizadeh F. Sustained delivery of amphotericin B and vancomycin hydrochloride by an injectable thermogelling tri-block copolymer. PDA J Pharm Sci Technol. 2013. doi:10.5731/pdajpst.2013.00908.
Jeong B, Choi Y, Bae Y, Zentner G, Kim S. New biodegradable polymers for injectable drug delivery systems. J Control Release. 1999;62(1):109–14.
Abu Hashim II, Higashi T, Anno T, Motoyama K, Abd-ElGawad A-EH, El-Shabouri MH, et al. Potential use of γ-cyclodextrin polypseudorotaxane hydrogels as an injectable sustained release system for insulin. Int J Pharm. 2010;392(1):83–91.
Khodaverdi E, Rajabi O, Farhadi F, Jalali A, Mirzazadeh TF. Preparation and investigation of poly (N-isopropylacrylamide-acrylamide) membranes in temperature responsive drug delivery. Iran J Basic Med Sci. 2010;13(3):102–10.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72(1):248–54.
Li X, Li J. Supramolecular hydrogels based on inclusion complexation between poly (ethylene oxide)‐b‐poly (ε‐caprolactone) diblock copolymer and α-cyclodextrin and their controlled release property. J Biomed Mater Res A. 2008;86(4):1055–61.
Masaro L, Zhu X. Physical models of diffusion for polymer solutions, gels and solids. Prog Polym Sci. 1999;24(5):731–75.
Möckel JE, Lippold BC. Zero-order drug release from hydrocolloid matrices. Pharm Res. 1993;10(7):1066–70.
Sarkar P, Bharill S, Gryczynski I, Gryczynski Z, Nair MP, Lacko AG. Binding of 8-anilino-1-naphthalenesulfonate to lecithin: cholesterol acyltransferase studied by fluorescence techniques. J Photochem Photobiol B Biol. 2008;92(1):19–23.
Hoogenboom R, Schubert US. Microwave-assisted polymer synthesis: recent developments in a rapidly expanding field of research. Macromol Rapid Commun. 2007;28(4):368–86.
Sosnik A, Gotelli G, Abraham GA. Microwave-assisted polymer synthesis (MAPS) as a tool in biomaterials science: how new and how powerful. Prog Polym Sci. 2011;36(8):1050–78.
Zhao S-P, Zhang L-M, Ma D. Supramolecular hydrogels induced rapidly by inclusion complexation of poly(epsilon-caprolactone)-poly(ethylene glycol)-poly(epsilon-caprolactone) block copolymers with alpha-cyclodextrin in aqueous solutions. J Phys Chem B. 2006;110(25):12225–9.
Ni X, Cheng A, Li J. Supramolecular hydrogels based on self-assembly between PEO-PPO-PEO triblock copolymers and α-cyclodextrin. J Biomed Mater Res A. 2009;88(4):1031–6.
Kopecek J. Hydrogels: from soft contact lenses and implants to self-assembled nanomaterials. J Polym Sci A Polym Chem. 2009;47(22):5929–46.
Khodaverdi E, Tekie FSM, Mohajeri SA, Ganji F, Zohuri G, Hadizadeh F. Preparation and investigation of sustained drug delivery systems using an injectable, thermosensitive, in situ forming hydrogel composed of PLGA–PEG–PLGA. AAPS PharmSciTech. 2012;13(2):590–600.
Acknowledgments
This project was supported by a grant from Vice Chancellor of Research, Mashhad University of Medical Sciences (MUMS), Mashhad, Iran. The results described in this paper were part of a Pharm D student thesis proposal related to Zinat Heidari.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Khodaverdi, E., Heidari, Z., Tabassi, S.A.S. et al. Injectable Supramolecular Hydrogel from Insulin-Loaded Triblock PCL-PEG-PCL Copolymer and γ-Cyclodextrin with Sustained-Release Property. AAPS PharmSciTech 16, 140–149 (2015). https://doi.org/10.1208/s12249-014-0198-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-014-0198-4