Abstract
Background
The bladder cancer (BC) pathology is caused by both exogenous environmental and endogenous molecular factors. Several genes have been implicated, but the molecular pathogenesis of BC and its subtypes remains debatable. The bioinformatic analysis evaluates high numbers of proteins in a single study, increasing the opportunity to identify possible biomarkers for disorders.
Methods
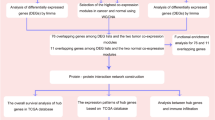
The aim of this study is to identify biomarkers for the identification of BC using several bioinformatic analytical tools and methods. BC and normal samples were compared for each probeset with T test in GSE13507 and GSE37817 datasets, and statistical probesets were verified with GSE52519 and E-MTAB-1940 datasets. Differential gene expression, hierarchical clustering, gene ontology enrichment analysis, and heuristic online phenotype prediction algorithm methods were utilized. Statistically significant proteins were assessed in the Human Protein Atlas database. GSE13507 (6271 probesets) and GSE37817 (3267 probesets) data were significant after the extraction of probesets without gene annotation information. Common probesets in both datasets (2888) were further narrowed by analyzing the first 100 upregulated and downregulated probesets in BC samples.
Results
Among the total 400 probesets, 68 were significant for both datasets with similar fold-change values (Pearson r: 0.995). Protein-protein interaction networks demonstrated strong interactions between CCNB1, BUB1B, and AURKB. The HPA database revealed similar protein expression levels for CKAP2L, AURKB, APIP, and LGALS3 both for BC and control samples.
Conclusion
This study disclosed six candidate biomarkers for the early diagnosis of BC. It is suggested that these candidate proteins be investigated in a wet lab to identify their functions in BC pathology and possible treatment approaches.
Similar content being viewed by others
Background
Bladder cancer (BC) is a widespread malignant tumor of the genitourinary system. It has a high mortality and recurrence risk, thus posing a significant health risk worldwide. The incidence rate of BC is proportional to the developmental stage of the region, but also depends on the developmental level of the population. In both situations, its incidence rises as the developmental stage increases [1].
The prevalence of BC is multidimensional; furthermore, the causative processes in its occurrence and progression are complex. Its pathology is caused by both exogenous environmental and endogenous molecular factors. Smoking and tobacco usage is one of the leading factors [2], which can be preventable. However, BC is still highly prevalent in men and women in all continents [1]. Last but the least, the Human Developmental Index (HDI) levels have been clearly implicated in several databases to be proportional to BC incidence [1, 3, 4]. Several genes have been implicated in the pathogenesis of BC [5, 6]. The treatment options for BC include conventional chemotherapy and immune checkpoint inhibitors, but fail to treat every individual [7]. Hence, personalized approaches and precise biomarker discoveries remain important.
Currently, the most common source of large-scale molecular data is transcriptome data for malignancies. High-throughput approaches such as microarray technology generate a large amount of genomic and expression data which are publicly available through various online repositories. The generation of big data necessitates the employment of high-throughput computer approaches and several methodologies can be utilized for statistical analysis. Bioinformatics, which is the intersection of biology, information science, and computation, was once an emerging component of cancer research [8], but now is one of the most common initial approaches prior to wet lab studies.
Methods
The aim of this research is to identify biomarkers for the early diagnosis and treatment of BC by using a variety of bioinformatic analytical tools and methods.
BC gene expression data sources
Four different datasets were downloaded from the gene expression omnibus (GEO) database (geneontology.org), including GSE13507 [Illumina] [9], GSE37817 [Illumina] [10], GSE21519 [Illumina] [11], and E-MTAB-1940 [Affymetrix] [12]. The latter two were used for validation studies. The datasets were downloaded from the ArrayExpress database in a normalized arrangement, and Affymetrix gene IDs were obtained accordingly (ebi.ac.uk/arrayexpress/).
Data processing and statistical tests
R 3.4.0 programming language was utilized for the statistical tests [13]. Student’s T test was used to compare gene expression levels in different groups.
Experimental design
BC and normal samples were compared for each probeset by T test in the GSE13507 and GSE37817 datasets with Venn diagrams using the online tool Venny 2.1 [14], and volcano plots were generated. The analysis was restricted to the first 100 upregulated and downregulated probesets. The median values of common probesets in the BC and normal samples were calculated. Then, “Pearson r” values were calculated for the gene expression values. With this approach, tumor and normal “r values” were obtained for all samples. Hierarchical clustering analysis was performed with Cluster 3.0 using Euclidean distance and complete linkage parameters [15]. The Heuristic Online Phenotype Prediction (HOPP) algorithm was applied as previously reported [16].
Functional enrichment analysis
Gene ontology (GO) enrichment analyses were performed using the PANTHER Classification Systems database [17]. Gene sets at p<0.05 were considered to be significant.
Candidate hub gene identification
A network analysis was performed using the Search Tool for the Retrieval of Interaction Genes 10.5 (STRING) database [18]. Protein-protein interaction (PPI) networks for differentially expressed genes (DEGs) were built on high confidence (0.700) interaction scores.
Results
Following the subtraction of probesets lacking gene annotation information, the comparison provided 6271 and 3267 significant probesets in GSE13507 and GSE37817 datasets, respectively (Fig. 1a). These two datasets showed significant overlapping for 2888 probesets (43.4%). To narrow the significant probesets, the first 100 upregulated and downregulated probesets in the tumor were selected with respect to their order of significance. For this purpose, a total of 200 probesets were compared from both datasets (GSE13507, GSE37817). Among these 400 probesets, 68 (20.5%) were significantly overlapping (Fig. 1b) and these mutual probesets showed similar fold-change values in the same direction (Pearson r: 0.995) (Fig. 1c; Table S1). All probesets were visualized in Volcano plots (Fig. 1d).
Common probesets between GSE13507 and GSE37817 datasets are determined using Venny diagrams a without restriction [common 2888 probesets, 34.4%] and b with restriction to the first 100 upregulated and downregulated in both datasets [common 68 probesets, 20.5%], c mutual probesets expressed similar fold-change values in the same direction (Pearson r: 0.995), d all probesets in both datasets were represented by volcano plots where each point represent a single probeset with fold change values on the x axis and logarithmic t test p value on the y axis [red and blue (first 100) dots indicating significant probesets, gray dots indicating non-significant probesets]
GO enrichment analysis
Among the 68 mutually significant probesets, a GO enrichment analysis was performed for the 42 upregulated (Fig. 2a) and 26 downregulated genes (Fig. 2b). The genes were enriched for biological processes, cellular components, and molecular functions. Some statistically significant pathways included, but were not limited to, GO:0051301: Cell division (fold enrichment: 23.72, p=7.1E−22); GO:0000280: Nuclear division (fold change: 28.69, p= 1.4E−14); GO:0140014: Mitotic nuclear division (fold change: 46.42, p= 2.2E−13); GO:0000070: Mitotic sister chromatid segregation (fold change: 52.08, p=4.2E−11); GO:1902099: Regulation of metaphase/anaphase transition of cell cycle (fold change: 78.35, p=4.2E−08); GO:0031577: Spindle checkpoint (fold change: 91.49, p= 1.1E−03); GO:0006260: DNA replication (fold change: 14.68, p= 2.8E−02). The PPI analysis indicates a strong interaction between all 42 upregulated proteins. Particularly, strong interactions between CCNB1, BUB1B, AURKB, CCNB2, CDC20, CDCA5, CENPF, and KNTC1 proteins were noticeable.
Validation
For the mutual 68 genes, two validation studies were performed using the GSE52519 and E-MTAB-1940 datasets. Out of the 68 probesets, 11 were missing in the GSE52519 dataset; hence, the validation was performed with 57 probesets (excluded probesets: ILMN_1792494, ILMN_1768291, ILMN_1808347, ILMN_1733950, ILMN_1663332, ILMN_1745594, ILMN_1747118, ILMN_1750347, ILMN_171251667, ILMN_1712574). The GSE52519 dataset included 9 BC samples and 3 healthy bladder samples. Normal samples were distinguished from BC samples with a hierarchical clustering analysis (Fig. 3a). Where BC samples were observed to form three clusters, subtypes 1, 2, and 3 (ST1, ST2, ST3). Two tumor samples showed closer proximity to normal samples, and this was observed with the HOPP (Fig. 3b).
The first validation study conducted with the GSE52519 dataset a revealed four clusters for the 57 mutual probesets, the rightmost group in the heat map represents the normal samples (N), two BC samples were located in the same cluster (ST1), four BC samples were clustered in the third group (ST2), and the fourth cluster included three BC samples that clustered separately (ST3); this validation included the CKAP2L and AURKB genes [red arrows] that were upregulated in ST2 and ST3 [BC and control samples are represented on the vertical axis, probes (genes) on the horizontal axis; red, black, and green colors represent high, medium, and low gene expression levels, respectively]. b The Heuristic Online Phenotype Prediction (HOPP), a gene expression analysis algorithm that employs PEARSON (r) correlations, grouped both (normal and BC) phenotypes and predicted two BC samples along with the normal sample
The second validation performed with the E-MTAB-1940 dataset was a 96 Affymetrix platform. Hence, the Affymetrix probesets corresponding to the genes of interest were determined, and out of the 68 genes, 52 were identified. The hierarchical clustering analysis performed with these probesets showed some separation between normal and tumor samples. Although, many tumor samples were clustered in the same group with normal samples, and one normal sample was clustered with tumor samples (Fig. S1).
Protein and tumor database analysis
The investigation of the common 68 proteins in The Human Protein Atlas database ([19]; proteinatlas.org) revealed a similar association between tumor and normal tissues at the protein level for CKAP2L, AURKB, CDC25A, APIP, and LGALS3.
Discussion
In this study, we performed bioinformatic analysis using microarray analysis to identify biomarkers related to BC which hold the potential to substantially sort between protein patterns for BC and normal tissue. Four datasets were used herein to categorize DEGs and validate results. The fact that the common probes were excessively high, we opted to select the first 100 upregulated and downregulated probes in tumor samples. With this approach, the high number of significant probesets (2888), which is an indication that the probability of false positives may be low, was narrowed to 68 common significant probesets. The comparable fold-change values of the shared probesets in both datasets behaved in the same manner. That is, the common proteins were upregulated and downregulated in both datasets. This is indicative that the effect of these common probesets on the formation of tumorigenesis can be determined independently in different experiment settings, thus possessing strong differentiating properties between cancer and healthy tissue.
GEO and STRING analysis
The GO enrichment analysis conducted with the 68 common probes demonstrates that most of these genes have functions related to cell division and mitosis; additionally, they were mostly found to encode proteins found in the “cell cycle-related pathways.” These included “regulation of cell cycle checkpoints” and “separation of sister chromatids,” which are key pathways of cellular and nuclear division, and are proposed to lead to cancer development [20]. The lack of commonly downregulated genes in BC samples indicates that these genes may have false-positive findings. Nevertheless, GEO data and STRING analysis results suggest that the trigger for BC development may be by accelerating biological mechanisms and pathways rather than inhibiting them. This could contribute to the selection of drugs and treatment approaches that suppress the accelerated pathways.
Specifically, the interaction between eight DEGs (CCNB1, CCNB2, CDC20, CDCA5, CENPF, KNTC1, BUB1B, and AURKB) was particularly apparent. CCNB1 has previously been reported in several studies on BC [21,22,23]. It encodes a key regulatory protein involved in the replication of nuclear matter by creating a complex with CDC2 (cell division cycle) also known as p34. Together, they control the mitosis at the G2/M-specific checkpoint [24]. Although CDC2 was not detected in this study, other regulatory genes responsible for the cell division cycle were enriched in the GEO and STRING analysis. This is likely to imbalance in the mitotic activity in BC samples. The CCNB1 gene is located on chromosome 5q13.2 and is composed of 9 exons; it encodes a 2029 bp mRNA and its longest transcript encodes a protein of 433 aa. Some SNPs on this gene have been previously reported in relation to cancer development; however, these SNPs have not been assessed in BC yet.
The spindle checkpoint kinase BUB1B (BUB1 mitotic checkpoint serine/threonine kinase beta) is involved in mitotic checkpoint and has recently been reported in a study as a hub candidate gene for BC [24]. It is thought to be localized to the kinetochore and delays the anaphase-promoting complex/cyclosome, enabling chromosomes to properly segregate [25]. Located on chromosome 15q15.1, it encodes an mRNA of 3669 bp and a protein of 1050 aa, and its impaired activity has been implicated in the formation of breast cancer [26].
Furthermore, similar oncogenes, including CCNB1, CDC20, and AURKA from the aurora kinase group were reported as hub genes by Zhang et al. [27], recently. Our study also supports their Go and KEGG enrichment analyses of DEGs, which implies the importance of mitotic checkpoints.
Validations studies
Two validation studies were conducted. The first one showed clear differentiation between tumor and normal samples. Almost none of the upregulated genes in tumor samples had high expression in normal samples. However, several upregulated genes in normal samples were also upregulated in some of the BC samples. Thus, genes with increased expression in the BC showed an enhanced discrimination power. This initial validation clustered the probes into four distinct groups. Two BC samples (ST1) were clustered with healthy tissues (N) and did not express the upregulated genes in BC, but rather expressed genes that were downregulated in tumor samples. The ST2 cluster containing 4 tumor samples showed increased expression in genes that were upregulated in the tumors. They behaved as expected from tumor samples. The final cluster (ST3) included 3 BC samples and expressed slightly less the upregulated genes in tumor samples. This indicates that based on the common 57 gene expressions, BC samples revealed subgroups that are comparable to or distinct from healthy tissue samples. Due to the fact that the datasets lacked information on tumor subtypes, a clustering of probes based on BC subtypes could not be implemented in this study. Nevertheless, provided that the dataset includes the BC subtype information, this can be accomplished and might offer alternative approaches and specific genes for personalized treatments.
The second validation dataset, although acquired from a different platform, was also capable of distinguishing between subtypes based on the common 52 genes out of 68, but to a lower extent. This dataset was composed of 86 samples with 4 normal samples and separated the BC samples into 3 subtypes. The distinction was not as apparent as it was in the first validation, due to the fact that there were multiple probesets corresponding to the same gene. Moreover, some probesets had no contribution to the separation, but nevertheless, a separation between BC subtypes was still noticeable.
Thus, based on the validation studies, the 68 featured proteins have been produced in two different platforms and distinguished not just BC from normal tissue but also differentiated between subtypes of BC.
Protein database
Protein database investigations led to the prominence of four of these 68 proteins (CKAP2L, AURKB, APIP, LGALS3) which were found consistent with the results of statistical protein levels presented herein. CKAP2L (cytoskeleton-associated protein 2-like) is involved in spindle organization and cell cycle progression from prometaphase to telophase [28]. AURKB (aurora kinase B), a member of the kinase family, is thought to have a role in the control of chromosomal alignment and segregation during mitosis by interacting with microtubules. A group of small-molecule inhibitors of AURKB, with ongoing or completed Phase I and II trials, have recently been proposed as potential drugs for cancer treatment [29]. Given that the expression levels of CKAP2L and AURKB statistically increased in BC samples, this could be a promising approach to investigate. APIP (APAF-1 interacting protein) is a protein found mostly in the cytosol and interacts with Apaf-1 (apoptotic protease activating factor-1), which holds a central role in the initiation to form the apoptosome complex and downstream pathway to intrinsic apoptosis [30]. APIP is thought to block the intrinsic mitochondrial apoptosis pathway via two routes, one Apaf-1-dependent [31] and the other Apaf-1-independent [32]. The downregulation of APIP expression in BC tissue samples could, however, be indicative of the absence of a regulatory protein. Similarly, mRNA and protein expressions of APIP were reported downregulated in non-small cell lung carcinoma [33]. LGALS3 (galectin 3), a member of the galectin family of carbohydrate-binding proteins, has previously been reported to induce apoptosis in human breast cancer cell lines through TRAIL signals that were dependent on increased PTEN activation and decreased PI3K/AKT survival pathway [34]. Downregulation of LGALS3 in the BC tissue most likely intervenes with TRAIL-induced apoptotic pathways. However, these findings contradict Oka et al. [35], who reported that overexpression of LGALS3 protects J82 human bladder cancer cells against TRAIL-induced apoptosis [35]. This contradiction can most likely be explained by the diverse apoptotic molecular pathways in different cell lines [34, 35].
BC is one of the most studied cancer types in the bioinformatic analysis due to its prevalence in humanity [27, 36, 37]. However, the use of different datasets as well as various types of statistical approaches, which improve constantly diverse biomarkers is constantly being predicted for BC [27, 36, 37], thus underlying the multiple factorial nature of BC and increasing the value of bioinformatic studies.
Conclusions
Considering these results, it seems that these 68 proteins, validated with different datasets on different platforms can be suggested as prominent candidate proteins for further investigations for biomarker detection. Moreover, based on the supportive findings, the six featured proteins can be proposed as possible biomarkers for BC, for which our laboratory has begun cell culture investigations to confirm our results. However, their efficacy in the BC subtype differentiation yet remains unclear. The global burden of BC has been previously reported [3, 4], and most developed countries in all five continents with high to moderate HDI levels suffer from BC today. Still, an increase is predicted in the next two decades, for which the countries with the highest HDI levels are still in the lead. This fact increases the urgency to uncover accurate biomarkers for early detection, prognosis assessment, and most importantly repurposing drugs or the discovery of new target agents and different treatment approaches. As a closing remark, it is of utmost importance to observe the long-term effects of the COVID-19 pandemic, which might have a reflection on the incidence and mortality rates of BC.
Availability of data and materials
The datasets analyzed during the current study are available in the [GEO DATASETS, NCBI] repository, [https://www.ncbi.nlm.nih.gov/gds/] with GSE13507, GSE37817 and GSE5.
Abbreviations
- BC:
-
Bladder cancer
- HDI:
-
Human Developmental Index
- GEO:
-
Gene expression omnibus
- HOPP:
-
Heuristic Online Phenotype Prediction
- GO:
-
Gene ontology
- STRING:
-
Search Tool for the Retrieval of Interaction Genes 10.5 (STRING)
- PPI:
-
Protein-protein interaction
- DEG:
-
Differentially expressed gene
- CDC2:
-
Cell division cycle
- BUB1B:
-
BUB1 mitotic checkpoint serine/threonine kinase beta
- CKAP2L:
-
Cytoskeleton-associated protein 2-like
- AURKB:
-
Aurora kinase B
- APIP:
-
APAF-1 interacting protein
- Apaf-1:
-
Apoptotic protease activating factor-1
- LGALS3:
-
Galectin 3
References
Antoni S, Ferlay J, Soerjomataram I, Znaor A, Jemal A, Bray F. Bladder cancer incidence and mortality: a global overview and recent trends. Eur Urol. 2017;71(1):96–108. https://doi.org/10.1016/j.eururo.2016.06.010.
Cumberbatch MG, Rota M, Catto JW, La Vecchia C. The role of tobacco smoke in bladder and kidney carcinogenesis: a comparison of exposures and meta-analysis of incidence and mortality risks. Eur Urol. 2016;70(3):458–66. https://doi.org/10.1016/j.eururo.2015.06.042.
Fidler MM, Soerjomataram I, Bray F. A global view on cancer incidence and national levels of the human development index. Int J Cancer. 2016;139(11):2436–46. https://doi.org/10.1002/ijc.30382.
Richters A, Aben KKH, Kiemeney LALM. The global burden of urinary bladder cancer: an update. World J Urol. 2020;38(8):1895–904. https://doi.org/10.1007/s00345-019-02984-4.
Timirci-Kahraman O, Ozkan NE, Turan S, Farooqi AA, Verim L, et al. Genetic variants in the tumor necrosis factor-related apoptosis-inducing ligand and death receptor genes contribute to susceptibility to bladder cancer. Genet Test Mol Biomarkers. 2015;19(6):309–15. https://doi.org/10.1089/gtmb.2015.0050.
Choi W, Ochoa A, McConkey DJ, Aine M, Höglund M, et al. Genetic alterations in the molecular subtypes of bladder cancer: illustration in the cancer genome atlas dataset. Eur Urol. 2017;72(3):354–65. https://doi.org/10.1016/j.eururo.2017.03.010.
Massari F, Di Nunno V, Cubelli M, Santoni M, Fiorentino M, et al. Immune checkpoint inhibitors for metastatic bladder cancer. Cancer Treat Rev. 2018;64:11–20. https://doi.org/10.1016/j.ctrv.2017.12.007.
Hanauer DA, Rhodes DR, Sinha-Kumar C, Chinnaiyan AM. Bioinformatics approaches in the study of cancer. Curr Mol Med. 2007;7(1):133–41. https://doi.org/10.2174/156652407779940431.
Kim WJ, Kim EJ, Kim SK, Kim YJ, Ha YS, et al. Predictive value of progression-related gene classifier in primary non-muscle invasive bladder cancer. Mol Cancer. 2010;9:3. https://doi.org/10.1186/1476-4598-9-3.
Kim YJ, Yoon HY, Kim JS, Kang HW, Min BD, et al. HOXA9, ISL1 and ALDH1A3 methylation patterns as prognostic markers for nonmuscle invasive bladder cancer: array-based DNA methylation and expression profiling. Int J Cancer. 2013;133(5):1135–42. https://doi.org/10.1002/ijc.28121.
Li MH, Fu SB, Xiao HS. Genome-wide analysis of microRNA and mRNA expression signatures in cancer. Acta Pharmacol Sin. 2015;36(10):1200–11. https://doi.org/10.1038/aps.2015.67.
Biton A, Bernard-Pierrot I, Lou Y, Krucker C, Chapeaublanc E, et al. Independent component analysis uncovers the landscape of the bladder tumor transcriptome and reveals insights into luminal and basal subtypes. Cell Rep. 2014;9(4):1235–45. https://doi.org/10.1016/j.celrep.2014.10.035.
R Core Team. R a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2020. https://www.R-project.org/. Access date: 16 Jul 2021
Oliveros, J.C. Venny. An interactive tool for comparing lists with Venn’s diagrams. 2015. http://bioinfogp.cnb.csic.es/tools/venny/index.html. Access date: 07 Jul 2021
de Hoon MJ, Imoto S, Nolan J, Miyano S. Open source clustering software. Bioinformatics. 2004;20(9):1453–4. https://doi.org/10.1093/bioinformatics/bth078.
Widmer DS, Cheng PF, Eichhoff OM, Belloni BC, Zipser MC, et al. Systematic classification of melanoma cells by phenotype-specific gene expression mapping. Pigment Cell Melanoma Res. 2012;25(3):343–53. https://doi.org/10.1111/j.1755-148X.2012.00986.x.
Mi H, Muruganujan A, Casagrande JT, Thomas PD. Large-scale gene function analysis with the PANTHER classification system. Nat Protoc. 2013;8(8):1551–66. https://doi.org/10.1038/nprot.2013.092.
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, et al. STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2014;43:D447–4452. https://doi.org/10.1093/nar/gku1003.
Uhlen M, Zhang C, Lee S, Sjöstedt E, Fagerberg L, et al. A pathology atlas of the human cancer transcriptome. Science. 2017;357(6352):eaan2507. https://doi.org/10.1126/science.aan2507.
Wenzel ES, Singh ATK. Cell-cycle checkpoints and aneuploidy on the path to cancer. In Vivo. 2018;32(1):1–5. https://doi.org/10.21873/invivo.11197.
Kim SK, Roh YG, Park K, Kang TH, Kim WJ, et al. Expression signature defined by FOXM1-CCNB1 activation predicts disease recurrence in non-muscle-invasive bladder cancer. Clin Cancer Res. 2014;20(12):3233–43. https://doi.org/10.1158/1078-0432.CCR-13-2761.
Chai N, Xie HH, Yin JP, Sa KD, Guo Y, et al. FOXM1 promotes proliferation in human hepatocellular carcinoma cells by transcriptional activation of CCNB1. Biochem Biophys Res Commun. 2018;500(4):924–9. https://doi.org/10.1016/j.bbrc.2018.04.201.
Liu A, Zeng S, Lu X, Xiong Q, Xue Y, et al. Overexpression of G2 and S phase-expressed-1 contributes to cell proliferation, migration, and invasion via regulating p53/FoxM1/CCNB1 pathway and predicts poor prognosis in bladder cancer. Int J Biol Macromol. 2019;123:322–34. https://doi.org/10.1016/j.ijbiomac.2018.11.032.
Yan X, Liu XP, Guo ZX, Liu TZ, Li S. Identification of hub genes associated with progression and prognosis in patients with bladder cancer. Front Genet. 2019;10:408. https://doi.org/10.3389/fgene.2019.00408.
Alfieri C, Zhang S, Barford D. Visualizing the complex functions and mechanisms of the anaphase promoting complex/cyclosome (APC/C). Open Biol. 2017;7(11):170204. https://doi.org/10.1098/rsob.170204.
Koyuncu D, Sharma U, Goka ET, Lippman ME. Spindle assembly checkpoint gene BUB1B is essential in breast cancer cell survival. Breast Cancer Res Treat. 2021;185(2):331–41. https://doi.org/10.1007/s10549-020-05962-2.
Zhang C, Berndt-Paetz M, Neuhaus J. Identification of key biomarkers in bladder cancer: evidence from a bioinformatics analysis. Diagnostics (Basel). 2020;10(2):66. https://doi.org/10.3390/diagnostics10020066.
Yumoto T, Nakadate K, Nakamura Y, Sugitani Y, Sugitani-Yoshida R, et al. Radmis, a novel mitotic spindle protein that functions in cell division of neural progenitors. PLoS One. 2013a;8(11):e79895. https://doi.org/10.1371/journal.pone.0079895.
Ahmed A, Shamsi A, Mohammad T, Hasan GM, Islam A, Hassan MI. Aurora B kinase: a potential drug target for cancer therapy. J Cancer Res Clin Oncol. 2021;147(8):2187–98. https://doi.org/10.1007/s00432-021-03669-5.
Shakeri R, Kheirollahi A, Davoodi J. Apaf-1: Regulation and function in cell death. Biochimie. 2017;135:111–25. https://doi.org/10.1016/j.biochi.2017.02.001.
Cho DH, Hong YM, Lee HJ, Woo HN, Pyo JO, et al. Induced inhibition of ischemic/hypoxic injury by APIP, a novel Apaf-1-interacting protein. J Biol Chem. 2004;279(38):39942–50. https://doi.org/10.1074/jbc.M405747200.
Cho DH, Lee HJ, Kim HJ, Hong SH, Pyo JO, et al. Suppression of hypoxic cell death by APIP-induced sustained activation of AKT and ERK1/2. Oncogene. 2007;26(19):2809–14. https://doi.org/10.1038/sj.onc.1210080.
Moravcikova E, Krepela E, Prochazka J, Rousalova I, Cermak J, Benkova K. Down-regulated expression of apoptosis-associated genes APIP and UACA in non-small cell lung carcinoma. Int J Oncol. 2012;40(6):2111–21. https://doi.org/10.3892/ijo.2012.1397.
Mazurek N, Sun YJ, Liu KF, Gilcrease MZ, Schober W, et al. Phosphorylated galectin-3 mediates tumor necrosis factor-related apoptosis-inducing ligand signalling by regulating phosphatase and tensin homologue deleted on chromosome 10 in human breast carcinoma cells. J Biol Chem. 2007;282(29):21337–48. https://doi.org/10.1074/jbc.M608810200.
Oka N, Nakahara S, Takenaka Y, Fukumori T, Hogan V, Kanayama HO, et al. Galectin-3 inhibits tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis by activating Akt in human bladder carcinoma cells. Cancer Res. 2005;65(17):7546–5753. https://doi.org/10.1158/0008-5472.CAN-05-1197.
Chen X, Jin Y, Gong L, He D, Cheng Y, Xiao M, et al. Bioinformatics analysis finds immune gene markers related to the prognosis of bladder cancer. Front Genet. 2020;11:607. https://doi.org/10.3389/fgene.2020.00607.
Meng XY, Shi MJ, Zeng ZH, Chen C, Liu TZ, Wu QJ, et al. The role of col5a2 in patients with muscle-invasive bladder cancer: a bioinformatics analysis of public datasets involving 787 subjects and 29 cell lines. Front Oncol. 2019;8:659. https://doi.org/10.3389/fonc.2018.00659.
Acknowledgements
The researcher would like to acknowledge the creators of the datasets who made their findings public.
Funding
This work was supported by the Scientific Research Projects Coordination Unit of Istanbul University. Project number: 47999.
Author information
Authors and Affiliations
Contributions
GIG obtained the funds for the study, interpreted the bioinformatic results, and was a major contributor in writing the manuscript; MI, SD, and TÇ performed the bioinformatic analysis of the publicly available datasets; MI contributed to the writing of the manuscript, OTK prepared the article figures and tables, and IY and TI contributed to the interpretation of the results and in the writing of the discussion. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Istanbul Okan University Ethical Committee assembled on December 29, 2021, that the presented study does not require an ethical approval (meeting number: 147, date: December 29, 2021).
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Fig. S1.
Hierarchical clustering analysis of the second validation study with 52|common probes (genes) in the E-MTAB-1940 dataset. [BC and control samples are represented on the vertical axis, probes (genes) on the horizontal axis; red, black, and green colours represent high, medium, and low gene expression levels, respectively].
Additional file 2: Table S1.
Mutual probesets in both datasets and their corresponding genes, expression levels in BC and normal tissue, fold-change and t-test p-values.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Inal Gültekin, G., Timirci Kahraman, Ö., Işbilen, M. et al. Six potential biomarkers for bladder cancer: key proteins in cell-cycle division and apoptosis pathways. J Egypt Natl Canc Inst 34, 54 (2022). https://doi.org/10.1186/s43046-022-00153-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43046-022-00153-0