Abstract
Background
Reduction in energy expenditure during fasting and chronic food restriction is well known and represents a mechanism of energy conservation, sparing energy, and preventing excessive weight and energy loss. The present study aimed to investigate if obesity can alter the reduction in energy expenditure that occurs in response to chronic food restriction, using two different experimental models that promote distinct types of obesity.
Methods
Female Wistar rats were submitted to two models of obesity: hypercaloric diet ingestion (HD) and bilateral lesion of ventromedial hypothalamus (VMHL). Thirty days after the beginning of each treatment (HD and VMHL), the control and obese animals were subdivided in two groups that ingested for 30 days ad libitum diet or 50% of food ingested by the respective ad libitum group (control or obese). At the end of this period, the animals were euthanized, and the energy balance and body composition were determined.
Results
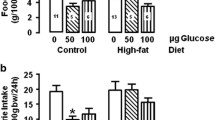
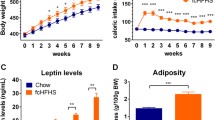
Obese animals (HD and VMHL) showed increase on their body fat, confirming the effectiveness of the models in obesity development. When submitted to food restriction, control and obese animals showed a significant reduction in body weight and energy, metabolizable energy intake, energy expenditure, gross food efficiency, and body fat content, when compared with their respective ad libitum groups. Energy expenditure reduction shown by obese food-restricted animals for both models was significantly lower than the one shown by non-obese food-restricted groups.
Conclusion
Obesity reduces the “sparing of energy” in food-restricted animals, suggesting that fat availability is one of the modulators of this mechanism; moreover, this physiologic adaptation should be considered in clinical nutrition protocols.


Similar content being viewed by others
Abbreviations
- HD:
-
Hypercaloric diet ingestion
- VMHL:
-
Bilateral lesion of ventromedial hypothalamus
- MSG:
-
Monosodium glutamate
- VMH:
-
Ventromedial hypothalamic nucleus
- UNIFESP:
-
Universidade Federal de São Paulo
- CEDEME:
-
Center for Development of Experimental Models
- NORMO:
-
Normocaloric group
- HYPER:
-
Hypercaloric group
- RES:
-
Food restriction
- CON:
-
Control
- OB:
-
Obese
- CAPES:
-
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
References
Olney JW. Brain lesions, obesity and other disturbances in mice treated with monosodium glutamate. Science. 1969;164:719–21.
Magarinos AM, Estivariz F, Morado MI, DeNicola AF. Regulation of central nervous system-pituitary-adrenal axis in rats after neonatal treatment with monosodium glutamate. Neuroendocrinology. 1988;48:105–11.
King BM. The rise, fall, and resurrection of the ventromedial hypothalamus in the regulation of feeding behavior and body weight. Physiol Behav. 2006;87:221–44.
Nascimento AF, Sugizaki MM, Leopoldo AS, Lima-Leopoldo AP, Luvizotto RAM, Nogueira CR. A hypercaloric pellet-diet cycle induces obesity and co-morbidities in Wistar rats. Arq Bras Endocrinol Metab. 2008;52(6):968–74.
Shiraev T, Chen J, Morris MJ. Differential effects of restricted versus unlimited high-fat feeding in rats on fat mass, plasma hormones and brain appetite regulators. J Neuroendocrinol. 2009;21:602–9.
Furnes MW, Zhao CM, Chen D. Development of obesity is associated with increased calories per meal rather than per day. A study of high-fat diet-induced obesity in young rats. Obes Surg. 2009;19:1430–8.
Woods SC, Seeley RJ, Rushing PA, D’Alessio D, Tso P. A controlled high-fat diet induces an obese syndrome in rats. J Nutr. 2003;133:1081–7.
Luz J, Pasin VP, Silva DJM, Zemdegs JC, Amaral LS, Affonso-Silva SM. Effect of food restriction on energy expenditure of monosodium glutamate-induced obese rats. Ann Nutr Metab. 2010;56:31–5.
Estadella D, Oyama LM, Bueno AA, Habitante CA, Souza GI, Ribeiro EB, et al. A palatable hyperlipidic diet causes obesity and affects brain glucose metabolism in rats. Lipids Health Dis. 2011;10:168–77.
Westerterp KR. Control of energy expenditure in humans. Eur J Clin Nutr. 2017;71:340–4.
Blum JW, Schnyder W, Kunz PL, Blom AK, Bickel H, Schurch A. Reduced and compensatory growth: endocrine and metabolic changes during food restriction and refeeding in steers. J Nutr. 1985;115:417–24.
Muller MJ, Bosy-Westphal A. Adaptative thermogenesis with weight loss in humans. Obesity (Silver Spring). 2013;21(2):218–28.
Munch IC, Markussen NH, Oritsland NA. Resting oxygen consumption in rats during food restriction, starvation and refeeding. Acta Physiol Scand. 1993;148:335–40.
Muller MJ, Enderle J, Bosy-Westphal A. Changes in energy expenditure with weight gain and weight loss in humans. Curr Obes Rep. 2016;5(4):413–23.
Luz J, Griggio MA. Distribution of energy between food-restricted dams and offspring. Annals Nutr Metab. 1996;40:165–74.
Santos-Pinto N, Luz J, Griggio MA. Energy expenditure of rats subjected to long-term food restriction. Int J Food Sci Nutr. 2001;52:193–200.
Griggio MA, Ingram DL. Effects of long term differences in energy intake on metabolic rate and thyroid hormones. Horm Metab Res. 1985;17:67–71.
Luz J, Griggio MA. Food restriction and refeeding in growing rats. Comp Biochem Physiol. 1991;99A:477–80.
Griggio MA, Luz J, Gorgulho AA, Sucassas CM. The influence of food restriction during different periods of pregnancy. Int J Food Sci Nutr. 1997;48:129–34.
Passadore MD, Griggio MA, Nunes MT, Luz J. Effects of ageing on the energy balance of food-restricted rats. Acta Physiol Scand. 2004;181:193–8.
Luz J, Griggio MA, Natrieli RM, Aumond MD. Energy balance of rats subjected to continuous and intermittent food restriction. Braz J Med Biol Res. 1995;28:1019–23.
Hill JO, Talano CM, Nickel M, DiGirolamo M. Energy utilization in food-restricted female rats. J Nutr. 1986;116:2000–12.
Estadella D, Oyama LM, Damaso AR, Ribeiro EB, Oller do Nascimento CM (2004). Effect of palatable hyperlipidic diet on lipid metabolism of sedentary and exercised rats. Nutrition. 20: 218–224.
Vido DA, Nejm MB, Silva NR, Silva SMA, Cravo SL, Luz J. Maternal obesity and late effects on offspring metabolism. Arq Bras Endocrinol Metab. 2014;58:301–7.
Barr HG, McCracken KJ. High efficiency of energy utilization in “cafeteria”-and forced-fed rats kept at 29C. Br J Nutr. 1984;51:379–87.
Folch J, Lees M, Sloane-Stanley GHA. A simple method for isolation and purification of total lipids from animal tissue. J Biol Chem. 1957;226:497–509.
Leshner AL, Litwin VA. A simple method for carcass analysis. Physiol Behav. 1972;9:282–9.
Morris MJ, Tortelli CF, Filippis A, Proietto J. Reduced BAT function as a mechanism for obesity in the hypophagic, neuropeptide Y deficient monosodium glutamate-treated rat. Regul Pept. 1998;75-76:441–7.
Iossa S, Lionetti L, Mollica MP, Crescenzo R, Botta M, Barletta A, et al. Effect of high-fat feeding on metabolic efficiency and mitochondrial oxidative capacity in adult rats. Br J Nutr. 2003;90(5):953–60.
So M, Gaidhy MP, Maghdoori B, Ceddia RB. Analysis of time-dependent adaptations in whole-body energy balance in obesity induced by high-fat diet in rats. Lipids Health Disease. 2011;10:99–110.
Bruss DB, Khambatta CF, Ruby MA, Aggarwal I, Hellerstein MK. Calorie restriction increases fatty acid synthesis and whole body fat oxidation rates. Am J Physiol Endocrinol Metab. 2010;298:E108–16.
Dulloo AG, Schultz Y. Adaptative thermogenesis in resistance to obesity therapies: issues in quantifying thrifty energy expenditure phenotypes in humans. Curr Obes Rep. 2015;4:230–40.
Trayhurn P, Beattie JH. Physiological role of adipose tissue: white adipose tissue as an endocrine and secretory organ. Proc Nutr Soc. 2001;60:329–39.
McGown C, Birerdinc A, Younossi ZM. Adipose tissue as an endocrine organ. Clin Liver Dis. 2014;18:41–58.
Acknowledgments
The authors acknowledge Daniele de Sá Vido for technical assistance, and Patricia Cassiolato Tufanetto for English review.
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
Vanessa Reis had a graduate student scholarship from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experimental protocol was approved by the Ethical Research Committee of the Universidade Federal de São Paulo (UNIFESP; n° 1505/10) and followed the Guidelines of the Brazilian National Council for Control of Animal Experimentation, based on the Federal Law 11.794.
Rights and permissions
About this article
Cite this article
Reis, V.A.B., Silva, N.R., Silva, S.M.A. et al. Obesity can alter energetic responses to food restriction. Nutrire 45, 7 (2020). https://doi.org/10.1186/s41110-019-0110-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-019-0110-9