Abstract
Background
Breast cancer (BC) is the most prevalent cancer, with a higher mortality rate in women worldwide. We aimed to investigate the association of the insulinemic potential of diet and lifestyle with the odds of BC using empirical indices, including the empirical dietary index for hyperinsulinemia (EDIH), empirical lifestyle index for hyperinsulinemia (ELIH), the empirical dietary index for insulin resistance (EDIR), and empirical lifestyle index for insulin resistance (ELIR).
Methods
This hospital-based case-control study was conducted among Tehranian adult women aged≥30 years. The final analysis was performed on 134 women newly diagnosed with histologically confirmed BC as a case and 267 healthy women of the same age as control. A 168-food item food frequency questionnaire was used for assessing dietary intakes at baseline. The odds ratios (ORs) and 95% confidence intervals (CIs) of BC across tertiles of EDIH, ELIH, EDIR, and ELIR were determined using multivariable-adjusted logistic regression.
Results
The mean ± SD of age and BMI of participants were 47.9±10.3 years and 29.4±5.5 kg/m2, respectively. EDIH score was related to the higher risk of BC based on fully adjusted models (OR:2.24;95%CI:1.21–4.12, Ptrend=0.016). Furthermore, subgroup analysis showed a higher BC risk with increasing EDIH score in postmenopausal women (OR:1.74, 95%CI:1.13-2.69) and those without a history of the oral contraceptive pill (OCP) use (OR:1.44;95%CI:1.02–2.04). Moreover, ELIH scores were positively associated with an increased risk of BC in postmenopausal women (OR; 1.98; 95% CI: 1.35 – 2.89), those with a family history of cancer (OR:1.94;95%CI:1.10–3.42), and in individuals who did not use OCP (OR:1.46; 95% CI:1.00–2.12).
Conclusion
Our results showed a possible link between EDIH and higher BC risk. Also, higher EDIH and ELIH scores were strongly associated with a higher risk of BC in postmenopausal women, those with a family history of BC, and those who do not use OCP.
Similar content being viewed by others
Background
Breast cancer (BC) is the most prevalent cancer, accounting for 25% of all female-related cancers worldwide, and second cancer with the highest mortality rate in women. The disease affects about 1.4 million people annually [1]. The well-established risk factors for BC, including age, genetic mutations, premature menstruation, late pregnancy, late menopause, hormone therapy, oral contraceptives, and cancer family history, are mostly unmodifiable [2]. However, unhealthy lifestyles, including physical inactivity, obesity, and inappropriate diet, are modifiable risk factors that play an important role in cancer pathogenesis [3]. In 2018, The World Cancer Research Center reported that choosing a healthier lifestyle and changing eating habits could prevent 4 million new people from developing cancer [2].
One of the predisposing biological agents for cancer incidence and its development is insulin-related disorders, particularly hyperinsulinemia, that play a crucial role in tumor development through insulin-like growth factor-1 (IGF-1) [4]. Obese and inactive subjects are more prone to imbalanced insulin homeostasis, and unhealthy dietary patterns can lead to hyperinsulinemia and insulin resistance (IR) [5, 6].
Recently, Tabung et al. have proposed dietary and lifestyle indices to predict hyperinsulinemia and IR using serum connecting peptide (C-peptide) and Triglyceride (TGs) to high-density lipoprotein-cholesterol (HDL-C) ratio, respectively. The empirical dietary index for hyperinsulinemia and IR (EDIH and EDIR) includes only food groups related to insulin biomarker responses. In contrast, the empirical lifestyle index for hyperinsulinemia and IR (ELIH and ELIR) is composed of physical activity (PA) and body mass index (BMI), and also food groups correlated with insulin biomarkers.
Previous studies investigated the link between these insulinemic dietary and lifestyle indices and the risk of several cancer types [7,8,9,10,11,12,13,14] regarding the well-established link between insulin disorders and cancer. A recent study showed that all EDIH, EDIR, ELIH, and ELIR are positively associated with the risk of hepatocellular carcinoma (HCC) [7]. Also, two studies demonstrated that a higher EDIH score increases prostate cancer risk [8, 9]. The Wang et al. study revealed that interventions to reduce the insulinemic potential of diet and lifestyle have protective effects against digestive system cancer [10]. Furthermore, there is a significant relationship between EDIH and the higher incidence of colorectal cancer (CRC) and poorer survival in patients with CRC [11, 12]. However, Lee et al. did not observe any significant relationship between EDIH and EDIR and multiple myeloma (MM) [13].
Accordingly, most previous studies observed a positive relationship between the EDIH and various types of cancer. However, despite the benefits of ELIH and ELIR as lifestyle scores indicating collective effects of diet, PA, and BMI, few studies have examined their association with cancer risk. Also, to our knowledge, there is no study exploring these indices’ relationship with the BC risk. So, we aimed to perform a case-control study to investigate the possible association of dietary and lifestyle indices for hyperinsulinemia (EDIH, ELIH) and insulin resistance (EDIR, ELIR) with BC risk in a sample of Iranian adult women.
Materials and method
Study design and sample
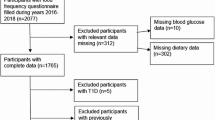
In this hospital-based, case-control study, we recruited 136 women ≥ 30 years old and newly (<6 months) diagnosed with histologically confirmed BC at Imam Hossain and Shohada hospitals, Tehran (Iran) between September 2015 and February 2016. The control group consisted of 272 women of similar age who were admitted to the same hospital for a broad spectrum of non-neoplastic diseases unrelated to smoking, alcohol consumption, and long-term diet modification. Conditions among controls included traumas and orthopedic disorders, disk disorders, acute surgical conditions, eye, nose, ear, or skin disorders. Less than 8% of subjects approached for the interview refused to participate. Seven participants were excluded from the final analysis because their reported energy intakes were outside the ±3 standard deviation (SD) of the mean energy intakes of the population (n=5 controls, 2 cases). Finally, 134 cases and 267 controls remained in the final analysis.
All participants signed the informed consent, and all procedures were according to the Helsinki Declaration’s ethical standards. The ethics research committee approved the study’s protocol of the Student Research Committee, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Dietary assessment
Participants’ dietary intake during the year before diagnosis for cases or interviews for controls was assessed in a personal interview using a valid and reliable semi-quantitative 168 food item food frequency questionnaire (FFQ) [14]. Participants were asked to specify their consumption frequency for each food item on a daily, weekly, monthly, or yearly basis. Questions on spices, including turmeric, saffron, black pepper, ginger, rosemary, and thyme, were added to the present questionnaire. Intakes were then converted to daily frequencies, and a manual for household measures was used to convert intake frequencies to daily grams of food intake [15]. The energy and nutrient content of foods was calculated by the United States Department of Agriculture (USDA) food composition table. The Iranian food composition table was used for some traditional Iranian food items that are not included in the USDA database (e.g., traditional bread). Due to Iranian regional beliefs, alcohol consumption was not asked and was unavailable for the analysis.
Calculation Of insulinemic indices
EDIH is calculated based on two groups of food components including positive (including red and processed meat, margarine, poultry, high-energy beverages, butter, French fries, low-fat dairy, tomatoes, and eggs) and negative (coffee, high-fat dairy, green and leafy vegetables, and whole fruits.) determinants. Each of the mentioned food groups is multiplied by a particular weight previously calculated in the study conducted by Tabung and his colleague [16] and then all food scores were summed as EDIH score.
Similarly, the ELIH is determined based on a set of direct (including BMI, margarine, butter, red meat, and fruit juice) and inverse (including coffee, whole fruits, physical activity, high-fat dairy products, snacks, and salad dressing) components. Like the EDIH, the ELIH score was calculated [16].
The EDIR encompasses two groups of food components, including positive and negative determinants [16]. The positive determinants included margarine, red meat, refined grains, processed meats, tomatoes, other vegetables, fish, and fruit juice. Negative determinants included coffee, green leafy vegetables, high-fat dairy products, dark yellow vegetables, and nuts. Similarly, the ELIR is determined based on positive and negative components [16]. The positive components included were BMI, refined grains, red meat, margarine, tomatoes, fruit juice, potatoes, processed meat, other vegetables, and tea. Negative ingredients were coffee, high-fat dairy products, PA, and green leafy vegetables.
Each of the mentioned components was multiplied by a particular weight, and all weighted values were summed to form the overall scores.
Assessment of non-dietary exposures
Trained dietitians administered all other questionnaires and measurements during the same interview. Participants’ socio-demographic, lifestyle, and clinical information collected by general questionnaires, including age (years), age at menarche (years), age at first pregnancy (years), abortion history during lifetime (yes, no), number of live births (number), breastfeeding history during lifetime (month), menopausal status at this time (pre-menopause, post-menopause), education (illiterate, less than a high school diploma, high school diploma and more), history of hormone replacement therapy during lifetime (yes, no), oral contraceptive pills(OCP) consumption history during lifetime (month), benign breast diseases history (yes, no), cancer family history (yes, no), breast cancer family history (yes, no), bra wearing (day (yes, no), night (yes, no)), marital status (single, married, divorced, widowed), smoking during lifetime (yes, no), supplement intakes in last year (including calcium, iron, zinc, selenium, B complex, Vitamin C, folic acid, vitamin A vitamin C, β carotene, vitamin E, vitamin D, multivitamins-minerals, omega-3 fatty acids, and probiotics) (yes, no; If yes, the complementary information on dose and frequency), and anti-inflammatory drug use (yes, no). Also, data on physical activity during the last year was assessed with a valid and reliable questionnaire [17].
Weight was measured to the nearest 0.5 kg using a digital scale (Seca, Germany), with the participant wearing lightweight clothing and no shoes. Height was measured to the nearest 0.5 cm using by tape meter fixed to a wall. Body mass index (BMI) was calculated by dividing weight (kg) by the square of height (meter). Furthermore, waist circumference (at the level midway between the lowest rib margin and the iliac) and hip circumference (at the widest point over the buttocks) were measured to the nearest 0.5 cm using a non-stretchable tape measure. Subsequently, the waist-hip ratio (WHR) was calculated.
Statistical analysis
The normal distribution of variables between case and control groups was assessed using a histogram chart and the Kolmogorov-Smirnoff test. The mean values of continuous and categorical variables were compared using the independent sample t-test or Mann-Whitney U (for non-normal variables) and the chi-square test.
The correlation coefficient (r) between different insulin indices was calculated using a partial correlation test. Each insulin indices were categorized as tertiles based on the three equal categories among controls. The odds ratios (ORs) and 95% confidence intervals (95% CIs) of breast cancer across tertiles of insulin indices were calculated by logistic regression analysis adjusted for various potential confounders in different models.
For selecting the confounding variables, we conducted a univariate test for the list of variables discussed in previous studies and selected those with P-values lower than 0.2. So the final model was adjusted for age, age at first pregnancy, family history of cancer, menopausal status, anti-inflammatory drugs, vitamin D supplement, BMI (for EDIH and EDIR), and physical activity (for EDIH and EDIR).
After testing for interaction, analyses were stratified by menopausal status, cancer family history, and OCP use (P-interaction<0.05). Statistical tests were performed using SPSS software (v.16.0). P-values < 0.05 were considered to be statistically significant.
Results
The baseline characteristics of participants, including demographic and lifestyle variables, medical history, and dietary intakes, are indicated in Table 1. Participants’ mean ± SD of age and BMI were 47.9 ± 10.3 years and 29.4 ± 5.5 kg/m2, respectively. Individuals in the case group had higher age, first pregnancy age, postmenopausal women percent, and cancer family history, whereas they had lower anti-inflammatory drug consumption and vitamin D supplement intake than the control group (P<0.05). There were no significant differences between cases and controls in insulinemic indices, including EDIH, EDIR, ELIH, EDIH, and other variables.
Table 2 shows the correlation coefficient of insulin indices. There was a significant correlation between EDIH and EDIR(r=0.263), EDIH and ELIH(r=0.215), and EDIR and ELIR(r=0.851).
The association of insulin indices with the risk of BC is presented in Table 3. A significant positive association was observed between higher EDIH score and risk of BC in the highest compared to the lowest tertiles in the age and age first pregnancy model (OR: 2.85; 95% CI: 1.05 – 3.23, P for trend=0.059). After adjusting for confounding factors in the final model, participants with the highest EDIH score had higher odds of BC than those with the lowest EDIH score (OR: 2.24; 95% CI: 1.21 – 4.12, P for trend=0.016). However, based on all logistic regression models, there is no significant association between ELIH, EDIR, and ELIR and the risk of BC.
Table 4 showed the adjusted OR (95% CI) of BC per one SD increment of insulin indices in subgroup analysis based on three variables, including menopausal status, cancer family history, and OCP use. Each SD increase in the EDIH score was associated with an increased risk of BC among postmenopausal women (OR: 1.74, 95% CI: 1.13–2.69) and those who do not use OCP (OR: 1.44;95% CI:1.02 – 2.04). Also, each SD increment of the ELIH score was related to a higher OR of BC based on postmenopausal status (OR; 1.98; 95% CI: 1.35 – 2.89), family history of cancer (OR: 1.94; 95% CI: 1.10 – 3.42), and no use of OCP (OR: 1.46; 95% CI: 1.00 – 2.12). The dietary or lifestyle indices for insulin resistance (EDIR and ELIR) showed no association with BC odds among subgroups.
Discussion
Current research provides the first evidence about the association between EDIH, EDIR, ELIH, and ELIR and the risk of BC. Based on our findings, EDIH was related to the higher risk of BC based on fully adjusted models. Furthermore, elevated EDIH level was associated with higher BC risk based on postmenopausal and non-using OCP based on subgroup analysis. Moreover, ELIH increment in postmenopausal women, those with a family history of cancer, and not using OCP was associated with an increased risk of BC.
Our findings are consistent with some previous studies investigating the possible association of the insulinemic potential of diet and lifestyle with the risk of various cancers. There is evidence that the higher score of EDIH was associated with a 26% increased colorectal cancer risk in men and women [18]. Similarly, Yang et al. have observed a positive association between higher scores of EDIR and increased risk of hepatocellular carcinoma [19]. Another study has reported that EDIH and ELIH were significantly associated with digestive tract cancers [20]. Furthermore, two studies claimed that participants with hyperinsulinemic diets had a greater risk of advanced and fatal prostate cancer [9, 21]. Although EDIR was associated with increased multiple myeloma risk in another study, EDIH did not show any significant relationship with the risk of multiple myeloma [13]. Although Cheng et al. declared that a higher score of EDIH is potentially related to colon cancer risk, no significant association was observed between a potential insulinemic diet and the risk of colon cancer recurrence, survival, or mortality in patients with late-stage (III) colon cancer [22]. EDIH, EDIR, ELIH, and ELIR are indicators that have been implicated in the pathogenesis of various cancer and complications [23, 24]. Insulin is a key regulator hormone in cell growth and energy metabolism. Up-regulation of insulin secretion and insulin resistance results in elevated IGF-1 production and bioavailability, resulting in cell proliferation and tumor growth [25]. It should be noted that dietary insulin indices are related to insulin resistance [26]. Moreover, some evidence shows that the dietary hyperinsulinemia index potentially affects cancer progression, especially in those with a low level of PA [18].
The subgroup analyses have revealed a positive association between EDIH and ELIH scores and the risk of BC in women who did not use OCP. Previous studies demonstrated that OCP use might increase BC’s risk [27]; however, this increased risk is slight and highly depends on different underlying factors like age, genetics, duration of use, and formulation of pills [28,29,30,31]. Besides, most of those studies included women who used OCP in the 1980s or earlier, when the pills tended to have a higher hormone content than they do now [32]. Furthermore, according to previous studies, OCP consumption increases insulin resistance risk [33, 34]. It is possible that in patients without a history of OCP use, due to fewer metabolic disorders than individuals with this history, the role of hyperinsulinemic diet and lifestyle in the development of metabolic disorders and increased cancer risk for each SD increase in scores is more prominent and therefore the overall relationship is significant.
A higher EDIH and ELIH score also increased the risk of BC in postmenopausal women. Increased endogenous estrogen status [35] is associated with postmenopausal breast cancer risk [36]. Although insufficient evidence is available on the possible effect of pre-and post-menopausal cancer, the risk of uterine, ovarian, and breast cancers increases with aging, especially after age 55 years old [37]. Lifestyle changes, anthropometric changes, including elevated BMI and adiposity, low physical activity, and hormone replacement therapy, increase by getting older and in those experiencing a postmenopausal period, can enhance BC risk [38, 39]. For instance, Toklu and Nogay indicate that unhealthy dietary patterns and eating deep-fried red meat, a sedentary lifestyle, and a high BMI, especially during the postmenopausal period, are risk factors for BC [40].
In addition, according to subgroup analysis results, a significant direct association of ELIH with the risk of BC was observed in subjects with a history of cancer. There is some evidence on the relationship between family history and the risk of BC, which expresses that first-degree family history can promote invasive BC risk [41]. For instance, Reiner et al. reported that having a first-degree relative with BC is associated with higher BC risk [42]. Also, Ahern et al. demonstrated that first- and second-degree relatives can estimate BC occurrence [43]. As well as hereditary has an important role in breast cancer incidence [44], family members may have a similar unhealthy lifestyle and dietary behaviors.
Strengths of the current study include the novelty of the investigation and data gathering from well-documented research centers. Furthermore, we have used valid and reliable questionnaires for dietary intake and physical activity evaluation. In addition, multiple logistic regression models, considering various confounders, were used in our study. However, we acknowledge some limitations.
The main limitation of the current study was the case-control study design which cannot provide a causal relationship. Also, our trained dietitians were not masked about cases and controls; however, during training, focus was given to minimize possible information bias to collect the data without mental background about participants’ cancer status. Since the study is conducted in Tehran, Iran, we cannot generalize the results to the other women population. Also, recall bias is possible due to using FFQ for dietary assessment. Furthermore, conducting stratified analyses based on three variables may increase the possibility of showing chance findings. However, the findings about EDIH, EDIR, and ELIR were repeated. Only ELIH showed a positive association with cancer among postmenopausal women, those with the cancer family history, and those with no OCP consumption. So, these findings should be tested among mentioned subgroups in other studies for a better perception of the ELIH-cancer relationship.
Conclusion
In conclusion, our results showed a possible link between EDIH and higher BC risk. Also, higher EDIH and ELIH scores were associated with a higher risk of BC in women in the postmenopausal period, having a family history of BC, and those who do not use OCP.
Availability of data and materials
The data analyzed in the present study are available by the corresponding author on a reasonable request.
Abbreviations
- BC:
-
Breast cancer
- EDIH:
-
Empirical dietary index for hyperinsulinemia
- ELIH:
-
Empirical lifestyle index for hyperinsulinemia
- EDIR:
-
Empirical dietary index for insulin resistance
- ELIR:
-
Empirical lifestyle index for insulin resistance
- OR:
-
Odds ratio
- CI:
-
Confidence interval
- SD:
-
Standard deviation
- OCP:
-
Oral contraceptive pill
- IGF-1:
-
Insulin-like growth factor-1
- IR:
-
Insulin resistance
- C-peptide:
-
Connecting peptide
- TG:
-
Triglyceride
- HDL-C:
-
High-density lipoprotein-cholesterol
- PA:
-
Physical activity
- BMI:
-
Body mass index
- HCC:
-
Hepatocellular carcinoma
- CRC:
-
Colorectal cancer
- MM:
-
Multiple myeloma
- FFQ:
-
Food frequency questionnaire
- USDA:
-
United States Department of Agriculture
- SPSS:
-
Statistical Package for the Social Sciences
- Kg:
-
Kilogram
References
DeSantis CE, Ma J, Goding Sauer A, Newman LA, Jemal A. Breast cancer statistics, 2017, racial disparity in mortality by state. CA Cancer J Clin. 2017;67(6):439–48.
Laudisio D, Barrea L, Muscogiuri G, Annunziata G, Colao A, Savastano S. Breast cancer prevention in premenopausal women: Role of the Mediterranean diet and its components. Nutrition research reviews. 2020;33(1):19-32.
Hashemi S, Karimi S, Mahboobi H. Lifestyle changes for prevention of breast cancer. Electron Physician. 2014;6:894–905.
Orgel E, Mittelman SD. The links between insulin resistance, diabetes, and cancer. Curr Diab Rep. 2013;13(2):213–22.
Erion KA, Corkey BE. Hyperinsulinemia: a cause of obesity? Curr Obes Rep. 2017;6(2):178–86.
Feskens EJM, Loeber JG, Kromhout D. Diet and physical activity as determinants of hyperinsulinemia: the Zutphen elderly study. Am J Epidemiol. 1994;140(4):350–60.
Yang W, Sui J, Zhao L, Ma Y, Tabung FK, Simon TG, et al. Association of Inflammatory and Insulinemic Potential of Diet and Lifestyle with Risk of Hepatocellular Carcinoma. Cancer Epidemiol Biomarkers Prev. 2021;30(4):789–96.
Fu BC, Tabung FK, Pernar CH, Wang W, Gonzalez-Feliciano AG, Chowdhury-Paulino IM, et al. Insulinemic and inflammatory dietary patterns and risk of prostate cancer. Eur Urol. 2021;79(3):405–12.
Aroke D, Folefac E, Shi N, Jin Q, Clinton SK, Tabung FK. Inflammatory and insulinemic dietary patterns: influence on circulating biomarkers and prostate cancer risk. Cancer Prev Res (Phila). 2020;13(10):841–52.
Wang W, Fung TT, Wang M, Smith-Warner SA, Giovannucci EL, Tabung FK. Association of the insulinemic potential of diet and lifestyle with risk of digestive system cancers in men and women. JNCI Cancer Spectr. 2018;2(4):pkz080.
Tabung FK, Noonan A, Lee DH, Song M, Clinton SK, Spakowicz D, et al. Post-diagnosis dietary insulinemic potential and survival outcomes among colorectal cancer patients. BMC Cancer. 2020;20(1):817.
Tabung FK, Wang W, Fung TT, Smith-Warner SA, Keum N, Wu K, et al. Association of dietary insulinemic potential and colorectal cancer risk in men and women. Am J Clin Nutr. 2018;108(2):363–70.
Lee DH, Fung TT, Tabung FK, Colditz GA, Ghobrial IM, Rosner BA, et al. Dietary pattern and risk of multiple myeloma in two large prospective US cohort studies. JNCI Cancer Spectr. 2019;3(2):pkz025.
Asghari G, Rezazadeh A, Hosseini-Esfahani F, Mehrabi Y, Mirmiran P, Azizi F. Reliability, comparative validity and stability of dietary patterns derived from an FFQ in the Tehran Lipid and Glucose Study. Br J Nutr. 2012;108(6):1109–17.
Ghaffarpour M, Houshiar rad A, Kianfar H. The manual for household measures, cooking yields factors and edible portion of food. Tehran: Keshaverzi press; 1999. ([in persian]).
Tabung FK, Wang W, Fung TT, Hu FB, Smith-Warner SA, Chavarro JE, et al. Development and validation of empirical indices to assess the insulinaemic potential of diet and lifestyle. Br J Nutr. 2016;116(10):1787–98.
Aadahl M, Jorgensen T. Validation of a new self-report instrument for measuring physical activity. Med Sci Sports Exerc. 2003;35(7):1196–202.
Tabung FK, Wang W, Fung TT, Smith-Warner SA, Keum N, Wu K, et al. Association of dietary insulinemic potential and colorectal cancer risk in men and women. Am J Clin Nutr. 2018;108(2):363–70.
Yang W, Sui J, Zhao L, Ma Y, Tabung FK, Simon TG, et al. Association of Inflammatory and Insulinemic Potential of Diet and Lifestyle with Risk of Hepatocellular Carcinoma. Cancer Epidemiol Biomarkers Prev. 2021;30(4):789–96.
Wang W, Fung TT, Wang M, Smith-Warner SA, Giovannucci EL, Tabung FK. Association of the insulinemic potential of diet and lifestyle with risk of digestive system cancers in men and women. JNCI Cancer Spectr. 2018;2(4):pky080.
Fu BC, Tabung FK, Pernar CH, Wang W, Gonzalez-Feliciano AG, Chowdhury-Paulino IM, et al. Insulinemic and inflammatory dietary patterns and risk of prostate cancer. Eur Urol. 2021;79(3):405–12.
Cheng E, Zhang S, Ou F-S, Mullen B, Ng K, Saltz LB, et al. The diet of higher insulinemic potential is not associated with worse survival in patients with stage III colon cancer (Alliance). Cancer Epidemiol Biomarkers Prev. 2020;29(8):1692–5.
Jin Q, Shi N, Aroke D, Lee DH, Joseph JJ, Donneyong M, et al. Insulinemic and Inflammatory Dietary Patterns Show Enhanced Predictive Potential for Type 2 Diabetes Risk in Postmenopausal Women. Diabetes care. 2021;44(3):707–14.
Mazidi M, Katsiki N, Mikhailidis DP, Banach M, Panel ILE. Effect of Dietary Insulinemia on All-Cause and Cause-Specific Mortality: Results From a Cohort Study. J Am Coll Nutr. 2020;39(5):407–13.
Zhu Y, Wang T, Wu J, Huang O, Zhu L, He J, et al. Biomarkers of insulin and the insulin-like growth factor axis in relation to breast cancer risk in Chinese women. OncoTargets and therapy. 2020;13:8027.
Mirmiran P, Esfandiari S, Bahadoran Z, Tohidi M, Azizi F. Dietary insulin load and insulin index are associated with the risk of insulin resistance: a prospective approach in tehran lipid and glucose study. J Diabetes Metab Disord. 2015;15(1):1–7.
Mørch LS, Skovlund CW, Hannaford PC, Iversen L, Fielding S, Lidegaard Ø. Contemporary Hormonal Contraception and the Risk of Breast Cancer. N Engl J Med. 2017;377(23):2228–39.
Hankinson SE, Colditz GA, Manson JE, Willett WC, Hunter DJ, Stampfer MJ, et al. A prospective study of oral contraceptive use and risk of breast cancer (Nurses’ Health Study, United States). Cancer Causes Control. 1997;8(1):65–72.
Beaber EF, Buist DSM, Barlow WE, Malone KE, Reed SD, Li CI. Recent oral contraceptive use by formulation and breast cancer risk among women 20 to 49 years of age. Cancer Res. 2014;74(15):4078–89.
Iodice S, Barile M, Rotmensz N, Feroce I, Bonanni B, Radice P, et al. Oral contraceptive use and breast or ovarian cancer risk in BRCA1/2 carriers: a meta-analysis. Eur J Cancer. 2010;46(12):2275–84.
Silvera SA, Miller AB, Rohan TE. Oral contraceptive use and risk of breast cancer among women with a family history of breast cancer: a prospective cohort study. Cancer Causes Control. 2005;16(9):1059–63.
Watch HWsH. Study finds weak link between birth control and breast cancer. 2020. [Available from: https://www.health.harvard.edu/womens-health/study-finds-weak-link-between-birth-control-and-breast-cancer.
Wahidin M, Djuwita R, Adisasmita A. Oral contraceptive and breast cancer risks: a case control study in six referral hospitals in Indonesia. Asian Pac J Cancer Prev. 2018;19(8):2199.
Frempong BA, Ricks M, Sen S, Sumner AE. Effect of low-dose oral contraceptives on metabolic risk factors in African-American women. J Clin Endocrinol Metab. 2008;93(6):2097–103.
Marchand GB, Carreau A-M, Weisnagel SJ, Bergeron J, Labrie F, Lemieux S, et al. Increased body fat mass explains the positive association between circulating estradiol and insulin resistance in postmenopausal women. Am J Physiol Endocrinol Metab. 2018;314(5):E448–56.
Sampson JN, Falk RT, Schairer C, Moore SC, Fuhrman BJ, Dallal CM, et al. Association of estrogen metabolism with breast cancer risk in different cohorts of postmenopausal women. Cancer Res. 2017;77(4):918–25.
Surakasula A, Nagarjunapu GC, Raghavaiah K. A comparative study of pre-and post-menopausal breast cancer: Risk factors, presentation, characteristics and management. J Res Pharm Pract. 2014;3(1):12.
Heer E, Harper A, Escandor N, Sung H, McCormack V, Fidler-Benaoudia MM. Global burden and trends in premenopausal and postmenopausal breast cancer: a population-based study. Lancet Global Health. 2020;8(8):e1027–37.
Vogel VG. Epidemiology, genetics, and risk evaluation of postmenopausal women at risk of breast cancer. Menopause. 2008;15(4):782–9.
Toklu H, Nogay N. Effects of dietary habits and sedentary lifestyle on breast cancer among women attending the oncology day treatment center at a state university in Turkey. Niger J Clin Pract. 2018;21(12):1576–84.
Braithwaite D, Miglioretti DL, Zhu W, Demb J, Trentham-Dietz A, Sprague B, et al. Family history and breast cancer risk among older women in the breast cancer surveillance consortium cohort. JAMA Internal Medi. 2018;178(4):494–501.
Reiner AS, Sisti J, John EM, Lynch CF, Brooks JD, Mellemkjær L, et al. Breast cancer family history and contralateral breast cancer risk in young women: an update from the women’s environmental cancer and radiation epidemiology study. J Clin Oncol. 2018;36(15):1513.
Ahern TP, Sprague BL, Bissell MC, Miglioretti DL, Buist DS, Braithwaite D, et al. Family history of breast cancer, breast density, and breast cancer risk in a US breast cancer screening population. Cancer Epidemiol Biomarkers Prev. 2017;26(6):938–44.
Chavarri-Guerra Y, Blazer KR, Weitzel JN. Genetic cancer risk assessment for breast cancer in Latin America. Rev Invest Clin. 2017;69(2):94–102.
Acknowledgments
This study is related to project NO 1399/60859 from the Student Research Committee, Shahid Beheshti University of Medical Sciences, Tehran, Iran. We also appreciate the “Student Research Committee” and “Research & Technology Chancellor” at Shahid Beheshti University of Medical Sciences for their financial support of this study. The authors express their appreciation to all of the participants of this study.
Funding
This study was supported by a grant from the student research committee, project NO [1399/60859], Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Author information
Authors and Affiliations
Contributions
E.M and F.T conceptualized and designed the study. F.T and S.J analyzed and interpreted the data. E.M and S.J drafted the initial manuscript. E.M, S.J, H.F, and SA.M edited and prepared the final manuscript. GH.D contributed to the revision and editing of the revised manuscript. B.R and P.M supervised the project, and all authors approved the final version of the manuscript as submitted.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All participants filled out written informed consent. All procedures were according to the Helsinki Declaration’s ethical standards. The ethics research committee approved the study’s protocol of the Student Research Committee, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Consent for publication
Not applicable.
Competing interests
The authors declared that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Mokhtari, E., Jamshidi, S., Daftari, G. et al. The relationship between the insulinemic potential of diet and lifestyle and risk of breast cancer: a case-control study among iranian adult women. Arch Public Health 81, 4 (2023). https://doi.org/10.1186/s13690-022-01016-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13690-022-01016-9