Abstract
Listeria monocytogenes (Lm) is a facultative, intracellular Gram-positive pathogenic bacterium that causes sepsis, a condition characterized by persistent excessive inflammation and organ dysfunction. However, the pathogenesis of Lm-induced sepsis is unknown. In this research, we discovered that TRIM32 is required for innate immune regulation during Lm infection. Trim32 deficiency remarkably reduced bacteremia and proinflammatory cytokine secretion in mice with severe Lm infection, preventing sepsis. Trim32−/− mice had a lower bacterial burden after Lm infection and survived significantly longer than wild-type (WT) mice, as well as lower serum levels of inflammatory cytokines TNF-α, IL-6, IL-18, IL-12p70, IFN-β, and IFN-γ at 1 day post infection (dpi) compared to WT mice. On the other hand, the chemokines CXCL1, CCL2, CCL7, and CCL5 were enhanced at 3 dpi in Trim32−/− mice than WT mice, reflecting increased recruitment of neutrophils and macrophages. Furthermore, Trim32−/− mice had higher levels of macrophage-associated iNOS to kill Lm. Collectively, our findings suggest that TRIM32 reduces innate immune cells recruitment and Lm killing capabilities via iNOS production.
Graphical Abstract

Similar content being viewed by others
Introduction
Listeria monocytogenes (Lm) is a Gram-positive pathogenic bacterium that can cause a variety of clinical syndromes such as sepsis, meningitis, and rhombencephalitis [1]. The infection of mice with this pathogen in the laboratory provides a wealth of information about the roles of immune cells and cytokines in host immunity against this pathogen. Over the last 20 years, the use of Lm as an intracellular pathogen model to investigate the host immune responses has established a paradigm in infection biology and basic microbiology [2, 3].
The activation of the host’s innate immune system and inflammatory responses are required for effective Lm infection elimination [4]. Lm infection causes the release of pro-inflammatory cytokines [5], IFN-β and IFN-γ [6]. Mice investigations have demonstrated that in the absence of IFN-β signaling, animals cannot be effectively infected by Lm, and mice with greater IFN-β levels have a higher bacterial load [7, 8]. One of the key mechanisms for controlling and eliminating Lm infection at the early stages is the recruitment of myeloid cells such as neutrophils, macrophages, and dendritic cells (DCs) to the infected sites [9, 10]. Macrophages produce inducible nitric oxide synthase (iNOS), which is primarily regulated by the NF-κB and JAK-STAT1 signaling pathways, with the goal of eradicating Lm proliferation [11,12,13].
TRIM32 is a member of the tripartite motif family, which is characterized by the presence of a RING domain that confers E3 ubiquitin ligase activity and functions as an important immune-regulatory protein in response to pathogen infection [14]. TRIM32 has been shown to promote IFN-β secretion during herpes simplex virus infection via the STING-IRF3 signaling pathway in order to eliminate infection [15]. Furthermore, it also increased IFN-γ production in response to Streptococcus suis infection, influencing blood–brain barrier permeability and aiding bacterial clearance in the brain [16]. However, the roles of TRIM32 in immunoregulation in Gram-positive intracellular bacterial infections such as Lm, on the other hand, are unknown.
In this study, we discovered that TRIM32 deficiency may promote the synthesis of iNOS and increase the secretion of chemokines helpful for neutrophil and macrophage recruitment while decreasing the production of inflammatory cytokines to prevent organ dysfunction to kill Lm. The information presented here will provide further insights into the regulation of antimicrobial innate immunity.
Materials and methods
Bacterial strains, growth medium, and cell culture
The strain Lm 10403S used in this study was originally isolated from skin lesions in Bozeman, Montana, USA, and has previously been used in intracellular pathology experimental models [7, 17]. Single colonies were inoculated into 5 ml of BHI (brain–heart infusion) medium and shaking vigorously overnight at 37 °C. Working cultures were created by inoculating 1% dilutions of overnight cultures into new BHI medium and culturing for 3 h at 37 °C to mid-log phase (about 5 × 108 CFU/ml).
Thioglycolate-elicited peritoneal macrophages (PMs) from C57BL/6 mice were prepared and cultured in endotoxin-free RPMI-1640 medium with 10% (vol/vol) FBS (Invitrogen). Bone marrow-derived macrophages (BMDMs) from C57BL/6 mice were cultured in endotoxin-free DMEM (Gibico) supplemented with 10% FBS after being prepared and generated in M-CSF (30 ng/ml) for 7 days (Peprotech). THP-1 cells were cultured in endotoxin-free RPMI-1640 medium with 10% (vol/vol) FBS (Invitrogen).
Ethics statement
All animals used in this study were housed at the Academy of Military Medical Sciences (AMMS) animal center in Beijing, China. Animals were cared for in accordance with the approved principles of laboratory animal care in China. All experimental procedures were approved by the Institutional Animal Care and Use Committee of the AMMS (IACUC-DWZX-2021-023).
Trim32-deficient mice
Trim32-deficient C57BL/6 mice were generated as previously described [16] using transcription activator-like effector nuclease (TALEN). Briefly, exon 2 of the trim32 gene (Ensembl accession no. ENSMUSG00000051675) was chosen as a TALEN target site to create trim32-deficient C57BL/6 mice. TALEN mRNAs generated by in vitro transcription were injected into fertilized eggs to produce knockout mice. The founders were genotyped using PCR and DNA sequencing, and the positive testers were bred to the next generation.
Lm infection
The bacteria were collected at the mid-logarithmic growth stage, washed and re-suspended in PBS, and counted on agar plates. To evaluate mice survival rates, 1.0 × 105 CFUs were injected intraperitoneally (i.p.). Meanwhile, mice were injected i.p. with 1.0 × 106 CFUs to assess bacterial burden in the spleen and liver by plate counting, organ lesions by hematoxylin and eosin (H&E) staining, and immune cell components by flow cytometry.
BMDMs and PMs were seeded in 24-well plates with 12 mm-coverslips over each well, followed by Lm infection at a multiplicity of infection (MOI) of 0.25. After discarding the Lm media, the mixture was incubated for 1 h with DMEM containing 50 μg/ml gentamicin. Bacterial counts were determined at 6 h post infection (hpi).
Flow cytometry
Spleens were minced through a 70 μm sieve and leukocytes were obtained after lysing red blood cells with ACK buffer. Livers were minced through a 100 μm cell strainer and digested for 30 min at 37 °C with rotation in HBSS (Ca2+, Mg2+) buffer containing 1 mg/ml (335 U/mg) collagenase IV (Sangon Biotech, Shanghai, China) and 200 μg/ml (100 Kunitz U/ml) DNase I (Sangon Biotech, Shanghai, China). Liver leukocytes were separated by percoll gradient centrifugation (GE Healthcare, Freiburg, Germany). The cells were then resuspended in RPMI-1640 at 1 × 106/ml, and surface staining was performed as previously described [16].
F4/80 and CD11b were used to stain macrophages, CD11c and CD11b for DCs, and Ly6G and CD11b for granulocytes. CD11c-FITC (N418; Biolegend), Gr1-PE (RB6-8C5; Biolegend), CD11b-PerCP-Cy5.5 (M1/70; eBioscience), F4/80-APC (BM8; Biolegend), and CD3-Pacific Blue (145-2C11; eBioscience) antibodies were utilized. As for intracellular iNOS-PE (W16030C; Biolegend) staining, the fixation and permeabilization steps were followed by Intracellular Fixation & Permeabilization Kit (eBioscience). Flow cytometric acquisition was performed with a BD FACSVerse flow cytometer, and data were analyzed using FlowJo software (TreeStar, Ashland, OR, USA).
Western blot
1–2 × 106 cells were resuspended in 150 μl RIPA lysis solution (Beyotime, Shang Hai, China) containing 1 tablet/8 ml EDTA-free complete protease inhibitor (Roche), 1 tablet/8 ml phosphatase inhibitor (Roche) and 1 mM PMSF (Sigma, Taufkirchen, Germany). After protein quantification, 5 × SDS loading buffer was added for 10 min at 95 to 100 ℃. Equal amounts of protein samples were separated by SDS-PAGE and transferred to nitrocellulose filter membrane. Blots were blocked with 5% BSA before being treated with primary antibodies, which were then followed with IRDye 800CW/680RD secondary antibodies (LI-COR Biosciences, USA). GAPDH (Sigma, Taufkirchen, Germany) and anti-TRIM32 (Abcam, Cambridge, UK) were employed. Western blot images were captured by the Odyssey SA infrared imaging system (LI-COR Biosciences, U.S.A.) and analyzed with Image Studio 5.x software (LI-COR Biosciences, America).
ELISA
Inflammatory cytokines were measured using the Cytokine & Chemokine 20-Plex Mouse ProcartaPlex™ Panel 1 (Invitrogen) kit and the Luminex 200 system (Luminex Corporation, Austin, TX, USA). Furthermore, the levels of IFN-γ/IFN-β in the supernatants of serum, spleen, and liver were determined using ELISA kit (R & D Systems). The procedures were carried out in accordance with the kit’s protocol.
Histopathology
Mice tissues were dissected and placed into 10% formalin-fixed liquid for H&E analysis. Sample sections were stained with H&E reagents according to the routine procedures. In brief, tissue samples were dehydrated in a graded series of alcohol washes, embedded in paraffin, sectioned into 4 μm slices, followed by H&E stain for a microscopic examination. Pictures were acquired using an Olympus BX53 microscope.
Statistical analysis
Unless otherwise specified, all of the data in this research are expressed as the mean ± standard error of the mean (SEM). The data analysis was based on the data’s normality and homogeneity of variance. Log-rank (Mantel-Cox) test was conducted to determine statistical significance for survival curve data, while others were analyzed using an unpaired two-tailed Student’s t-test on the results from two groups or two-way ANOVA followed by Dunnett’s multiple comparisons teston the results from three or more groups. For all tests, a P value of < 0.05 was considered the threshold for significance. All statistical analysis in this study were performed with the GraphPad Prism 8.0.1 software program (GraphPad Software Inc, San Diego, CA, USA).
Results
Trim32 deficiency improved resistance against Lm infection
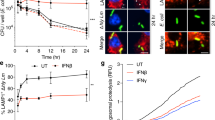
We used PMA-differentiated THP-1 cells to examine the host trim32 gene’s response to Lm infection (Fig. 1A). TRIM32 steadily elevated from 0 to 12 hpi and maintained it at 24 hpi. To determine if TRIM32 plays an antibacterial and immunological function in Lm infection, we infected wild-type (WT) and Trim32−/− mice (Additional file 1: Fig S1) with Lm and followed their survival rate over time (Fig. 1B). All WT mice died at 7 days post infection (dpi), whereas only half of Trim32−/− mice perished, indicating that trim32 deletion had a protective impact during Lm infection. To further confirm the involvement of TRIM32 in antimicrobial immunity against Lm infection, we infected WT and Trim32−/− mice via i.p. infection and measured bacterial load at 1 and 3 dpi in the spleen and liver, which were the major organs colonized by Lm [18]. As shown in Fig. 1C, D, WT mice had approximately 100-fold higher Lm load in their livers and spleens than Trim32−/− mice. Also, we examined the spleen white plaques and liver histopathological of Lm-infected mice. At 3 dpi, WT mice displayed severe white plaques in spleen (Fig. 1E) and erosion in liver (Fig. 1F), and Trim32−/− mice showed relatively only minor damage. Together, the Lm infection and the increased expression of TRIM32 promoted bacterial growth and proliferation in mice.
Trim32 deficiency improved resistance against Lm infection. A Immunoblot analysis of inducible TRIM32 expression in THP-1 cells upon Lm infection. THP-1 was differentiated with 100 nM PMA for 2 days and replaced by RPMI media, followed by Lm infection at a MOI of 0.1 for indicated times. B Survival curves for WT and Trim32−/− mice infected with Lm. Mice (n = 10 per group) were injected i.p. with 1.0 × 105 CFU of Lm, and their survival was monitored daily. C, D Bacteria counts in the spleen and liver of mice infected i.p. with 1.0 × 106 CFUs of Lm. Bacterial counts were determined by colony plate count. Showed were the combined results of two independent experiments with a total of 8 to 10 mice per experimental group and time point. Mean value for each experimental group was shown by a bar and each symbol represents one mouse. Macroscopic examination of the spleen E and HE staining of liver F from infected Trim32−/− and WT mice, and the red circles indicated areas of erosion. Mice were injected i.p. with 1.0 × 106 CFUs of Lm, spleens and livers were inspected at 3 dpi. Data were a representative of three independent experiments. Statistical significance was calculated by means of (B) log-rank Mantel-Cox test, (C, D) two-way ANOVA followed by Dunnett’s multiple comparisons test; * P < 0.05, ** P < 0.01
Trim32 deficiency reduced production of pro-inflammatory cytokines while increased chemokines
We evaluated serum cytokine levels in WT and Trim32−/− mice at 0, 1, 2, and 3 dpi, as cytokines have previously shown to be involved in the early pathogenesis of Lm infection and disease progression. Compared to WT mice, the proinflammatory cytokines IL-12, IL-18, IL-6, and TNFα were significantly reduced in Trim32−/− mice at 1 dpi (Fig. 2A–D), whereas the chemokines CXCL1 (neutrophil trafficking, Fig. 2E), CCL2 and CCL7 (monocytes trafficking, Fig. 2F, G), and CCL5 (macrophages trafficking, Fig. 2H) were significantly increased in Trim32−/− mice at 3 dpi. As a result, TRIM32 promoted the secretion of pro-inflammatory cytokines while inhibiting the secretion of chemokines in mice sera during the early stages of Lm infection.
Trim32 deficiency reduced pro-inflammatory cytokine production while increased chemokine production. Trim32−/− and WT mice were i.p. injected with 1.0 × 106 CFUs of Lm. Levels of pro-inflammatory cytokines (A–D) and chemokines (E–H) in sera of mice at indicated time points were measured using Luminex. Shown were mean ± SEM of 3 mice per experimental group. Data were a representative of three independent experiments. Statistical significance was calculated by means of two-way ANOVA followed by Dunnett’s multiple comparisons test; * P < 0.05, ** P < 0.01
Trim32 deficiency reduced production of IFN-β and IFN-γ
We measured IFN-β and IFN-γ in serum, spleens and livers of WT and Trim32−/− mice at 1 and 3 dpi, respectively. IFN-β was significantly lower in Trim32−/− mice serum at 1 and 3 dpi (Fig. 3A), spleens at 1 dpi (Fig. 3B), and livers at 3 dpi (Fig. 3C) compared to WT mice. Furthermore, IFN-γ was also significantly reduced in Trim32−/− mice serum at 1 dpi (Fig. 3D), spleens at 1 and 3 dpi (Fig. 3E), and livers (Fig. 3F). Overall, our findings showed that TRIM32 increased the production of IFN-β and IFN-γ in mice following Lm infection.
Trim32 deficiency reduced IFN-β and IFN-γ production. Trim32−/− and WT mice were i.p. injected with 1 × 106 CFUs of Lm. Levels of IFN-β A-C and IFN-γ D-F in serum and in supernatants of homogenized spleens and livers at indicated time points were determined using ELISA. A, D Showed were the combined results of two independent experiments with a total of 10 mice per experimental group and time point B, C, E, F. Data were a representative of three independent experiments. Each symbol represented one mouse. Statistical significance was calculated by means of two-way ANOVA followed by Dunnett’s multiple comparisons test; * P < 0.05, ** P < 0.01
Trim32 deficiency enhanced recruitment of macrophages and neutrophils but decreased that of DCs
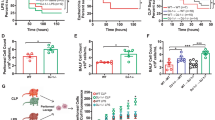
Given that bacterial elimination at early stages of Lm infection mainly relied on innate immune cells such as macrophages, neutrophils and DCs [19], we measured the abundance of these three cells in the spleen and liver of Trim32−/− and WT mice at 0, 1, 2, and 3 dpi. The gating strategy analyzed by flow cytometry was shown in Fig. 4A. Uninfected Trim32−/− and WT mice had similar populations of innate immune cells in the spleen and liver (Additional file 1: Fig S2). Trim32−/− mice had higher proportions of Gr1+ CD11b+ neutrophils in the spleen than WT mice at 2 and 3 dpi (Fig. 4B), and there was an increased trend in the liver at 3 dpi, but no statistical difference (Fig. 4B). Trim32−/− mice had more F4/80+ CD11b+ macrophages in their spleens than WT mice at 1 dpi (Fig. 4C). Similarly, at 1 dpi, there was an increased trend in liver but no statistical difference (Fig. 4C). Trim32−/− mice had a lower proportion of CD11b+ CD11c+ DCs in their livers at 1 dpi than WT mice (Fig. 4D). Overall, TRIM32 inhibited the recruitment of macrophages and neutrophils but promoted that of DCs at early stages of Lm infection.
Trim32 deficiency enhanced recruitment of macrophages and neutrophils but decreased that of DCs. A Schematic illustration of the gating strategy. (B-D) Relative proportions of neutrophils, macrophages, and DCs in spleen and liver. Trim32−/− and WT mice were i.p. injected with 1 × 106 CFUs of Lm. Mice were sacrificed at 0, 1, 2, and 3 dpi to analyze composition of innate immune cells. Shown were mean ± SEM of 3 mice per experimental group. Data were a representative of three independent experiments. Statistical significance was calculated by means of B–D two-way ANOVA followed by Dunnett’s multiple comparisons test; * P < 0.05, ** P < 0.01
Trim32 deficiency increased macrophage-associated iNOS production
To begin, we infected two primary mouse macrophages, BMDMs and PMs, with Lm at a MOI of 0.25 and then measured the intracellular bacterial growth at 0.5, 2 and 6 hpi (Fig. 5A, B). Trim32−/− macrophages had a lower bacterial load at 6 hpi than WT. Furthermore, we examined cell viability and the iNOS production in BMDMs following Lm infection (Fig. 5C, D). FVD-eF600 (a marker of cell viability) expression was decreased in Trim32−/− BMDMs at 6 and 24 hpi, while iNOS was increased at 6 and 24 hpi. In all, TRIM32 inhibited bactericidal activity by decreasing iNOS production in macrophages.
Trim32 deficiency increased macrophage bactericidal activity. A, B Growth of Lm in macrophages. Macrophages were seeded in 12 mm coverslips in 24-well culturing plates and infected with Lm at a MOI of 0.25 for 0.5 h. Lm media was then discarded and replaced with DMEM containing 50 μg/mL gentamicin, then the mixture was incubated for 1 h. Bacterial counts were determined for infected BMDMs (A) and PMs (B) at 0.5, 2, and 6 hpi. Shown were mean ± SEM of 3 mice per experimental group. Data were a representative of three independent experiments. C, D Representative flow cytometry plots. BMDMs were infected by Lm at a MOI of 0.1 for indicated times, and then viability (C) and iNOS expression (D) in BMDMs were analyzed by flow cytometry. E–F Statistical analysis of flow cytometry data from multiple replicates. BMDMs were infected by Lm at a MOI of 0.1 for indicated times, and then viability of BMDMs (E), iNOS expression in CD11b+ BMDMs (F). Data were a representative of three independent experiments. Statistical significance was calculated by means of A, B unpaired two-tailed Student’s t-test and E–F two-way ANOVA followed by Dunnett’s multiple comparisons test. * P < 0.05, *** P < 0.001
Discussion
The role of TRIM32 in the pathogenesis of Lm-induced sepsis in mice was investigated in this study. Our findings showed that TRIM32 was induced to be expressed in mice after Lm infection, whereas deletion of the Trim32 gene in mice resulted in increased animal survival and decreased bacterial load in the major infected organs, which was later linked to the down-regulation of serum proinflammatory cytokines (TNF-α, IL-6, IL-18, and IL-12p70) and interferons (IFN-β and IFN-γ). Trim32 deficiency also increased neutrophil and macrophage recruitment to the spleen following Lm infection. Also, we discovered that Trim32 deficiency increased BMDMs bactericidal activity and macrophage iNOS production. Collectively, TRIM32 aggravated Lm-induced sepsis in mice at early stages of infection, which was associated with increased cytokine production, decreased leucocyte recruitment and iNOS production.
Excessive serum inflammatory cytokines such as IL-6, TNF-α, and IL-18 damage host homeostasis during the early stages of innate immune defense, eventually leading to multi-organ failure and death [20, 21]. Enhanced IFN-β production not only contributes to the above-mentioned excessive cytokine production, but also facilitates Lm proliferation in mice [7], and mice lacking IFN-β receptor or with IFN-β production deficiency are more resistant to Lm-induced infection in mice [7, 22]. Similar to IFN-β, excessive IFN-γ production has a negative impact on mouse survival after Lm infection: Lm-induced infection causes excessive IFN-γ production, which inhibits granulocyte recruitment to infected sites in mice, but when mice are rejected with an IFN-γ-neutralizing monoclonal antibody, mouse survival rates improve [23]. The findings support the link discovered in this study between TRIM32 and Lm-induced sepsis in mice: inducible expression of TRIM32 in response to Lm infection results in excessive production of inflammatory cytokines and interferons, which may be the primary cause of acceleration of multi-organ lesion and increased bacterial loads. Furthermore, the increased chemokines expression in Trim32−/− mice on day 3 may facilitate recruitment of immune cells, such as neutrophils and macrophages, thus contribute to the increased Lm bactericidal ability and survival rate. Moreover, increased iNOS expression which required to clear intracellular Lm infection [24,25,26] in macrophages maybe another major reason to increase Trim32−/− bactericidal ability. In brief, low cytokines and high chemokines and iNOS expression may help Trim32−/− mice resist the attack of Lm.
Inflammasomes activation is a well-known function of the innate immune response to Lm [27], leading to activates both the NLRP3 and the AIM2 inflammasome, thus leading to the secretion of IL-1β and IL-18 [28]. The roles of TRIM family members in regulating inflammasome activation are crucial in manipulating this process. Several TRIM proteins have been found to regulate NLRP3 inflammasome activation. TRIM33 positive regulates NLRP3 inflammasome activation upon stimulation with cytosolic dsRNA and bacterial RNA through the ubiquitylates dsRNA sensor DHX33 via K63 specific linkage, inducing the cleavage of caspase-1 and maturation of IL-1β and IL-18 [29, 30]. TRIM21 triggers the activation of NLRP3 by targeting STING for K63-linked ubiquitination at K20/150/224/236 through its E3 ubiquitin ligase activity, which promoted the interaction of STING with TBK1 upon RNA and DNA virus infection [31]. Similarly, TRIM32 is also an E3 ubiquitin ligase [32], but the role of TRIM32 on Lm-triggered inflammasome activation remains elucidated. In this study, we found decreased IL-18 level in Trim32−/− mice serum upon Lm infection, suggesting a potential correlation between TRIM32 and inflammasomes. The further study will unpack the function of TRIM32.In a mouse model of Salmonella typhimurium (St) infection [33], deletion of the trim32 gene reduced animal survival and decreased bacteria burden in the spleen and liver; these findings are contradictory and consistent with those for Lm-induced infection, respectively. The above contradiction may be due to the fact that St, as a Gram-negative bacterium, and Lm, as a Gram-positive bacterium, respectively, stimulate host immune response via toll-like receptor 4 (TLR4) [34, 35] and TLR2 [36, 37]. TRIM32 has been shown to ubiquitinate STING, thereby activating downstream signaling pathways such as NF-B and IRF3 [32, 38]. TRIM32 inhibits inflammatory gene transcription after St infection via TLR4/Myd88/TRIF-activated downstream LC3-associated selective autophagy [33]. Lm infection activates the TLR2-NF-κB signaling pathway to promote the production of inflammatory cytokines [39]. It is thus hypothesized that TRIM32 contributes to increased production of inflammatory cytokines in response to Lm infection, most likely via the TLR2-NF-κB pathway.
Overall, our findings show that TRIM32 modulates innate immune response and iNOS production in macrophages, assisting the host in fighting Lm-induced sepsis at the early stages of infection. TRIM32 is an intriguing host therapeutic target for the treatment of Lm-induced sepsis.
Availability of data and materials
The original data for this study are available from the corresponding author.
References
Schlech WF. Epidemiology and clinical manifestations of listeria monocytogenes infection. Microbiol Spectr. 2019. https://doi.org/10.1128/microbiolspec.GPP3-0014-2018.
Lecuit M. Listeria monocytogenes, a model in infection biology. Cell Microbiol. 2020;22(4):e13186.
Radoshevich L, Cossart P. Listeria monocytogenes: towards a complete picture of its physiology and pathogenesis. Nat Rev Microbiol. 2018;16(1):32–46.
Regan T, MacSharry J, Brint E. Tracing innate immune defences along the path of listeria monocytogenes infection. Immunol Cell Biol. 2014;92(7):563–9.
Huggins MA, et al. Microbial exposure enhances immunity to pathogens recognized by TLR2 but increases susceptibility to cytokine storm through TLR4 Sensitization. Cell Rep. 2019;28(7):1729–43.
Dussurget O, Bierne H, Cossart P. The bacterial pathogen listeria monocytogenes and the interferon family: type I, type II and type III interferons. Front Cell Infect Microbiol. 2014;4:50.
Auerbuch V, et al. Mice lacking the type I interferon receptor are resistant to listeria monocytogenes. J Exp Med. 2004;200(4):527–33.
Carrero JA, Calderon B, Unanue ER. Type I interferon sensitizes lymphocytes to apoptosis and reduces resistance to listeria infection. J Exp Med. 2004;200(4):535–40.
Liu M, et al. Formylpeptide receptors are critical for rapid neutrophil mobilization in host defense against listeria monocytogenes. Sci Rep. 2012;2:786.
Aoshi T, et al. The cellular niche of listeria monocytogenes infection changes rapidly in the spleen. Eur J Immunol. 2009;39(2):417–25.
Cinelli MA, et al. Inducible nitric oxide synthase: regulation, structure, and inhibition. Med Res Rev. 2020;40(1):158–89.
Miller BH, et al. Mycobacteria inhibit nitric oxide synthase recruitment to phagosomes during macrophage infection. Infect Immun. 2004;72(5):2872–8.
Chaudhuri S, et al. The Listeria monocytogenes ChiA chitinase enhances virulence through suppression of host innate immunity. MBio. 2013;4(2):e00617.
Lazzari E, Meroni G. TRIM32 ubiquitin E3 ligase, one enzyme for several pathologies: from muscular dystrophy to tumours. Int J Biochem Cell Biol. 2016;79:469–77.
Cui H, Liu Y, Huang Y. Roles of TRIM32 in corneal epithelial cells after infection with herpes simplex virus. Cell Physiol Biochem. 2017;43(2):801–11.
OuYang X, et al. TRIM32 drives pathogenesis in streptococcal toxic shock-like syndrome and streptococcus suis meningitis by regulating innate immune responses. Infect Immun. 2020. https://doi.org/10.1128/IAI.00957-19.
O’Riordan M, et al. Innate recognition of bacteria by a macrophage cytosolic surveillance pathway. PNAS. 2002;99:13861–6.
Vazquez-Boland JA, et al. Listeria pathogenesis and molecular virulence determinants. Clin Microbiol Rev. 2001;14(3):584–640.
Edelson BT, Unanue ER. Immunity to listeria infection. Curr Opin Immunol. 2000;12:425–31.
Jennifer R, et al. Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76(1):16–32.
Ragab D, et al. The COVID-19 cytokine storm; what we know so far. Front Immunol. 2020;11:1446.
Archer KA, Durack J, Portnoy DA. STING-dependent type I IFN production inhibits cell-mediated immunity to listeria monocytogenes. PLoS Pathog. 2014;10(1):e1003861.
Viegas N, et al. IFN-gamma production by CD27(+) NK cells exacerbates listeria monocytogenes infection in mice by inhibiting granulocyte mobilization. Eur J Immunol. 2013;43(10):2626–37.
MacMicking JD. Interferon-inducible effector mechanisms in cell-autonomous immunity. Nat Rev Immunol. 2012;12(5):367–82.
Myers JT, Tsang AW, Swanson JA. Localized reactive oxygen and nitrogen intermediates inhibit escape of listeria monocytogenes from vacuoles in activated macrophages. J Immunol. 2003;171(10):5447–53.
Lipinski S, et al. DUOX2-derived reactive oxygen species are effectors of NOD2-mediated antibacterial responses. J Cell Sci. 2009;122(Pt 19):3522–30.
Eitel J, Suttorp N, Opitz B. Innate immune recognition and inflammasome activation in listeria monocytogenes infection. Front Microbiol. 2010;1:149.
Yang L, Xia H. TRIM proteins in inflammation: from expression to emerging regulatory mechanisms. Inflammation. 2021;44(3):811–20.
Mitoma H, et al. The DHX33 RNA helicase senses cytosolic RNA and activates the NLRP3 inflammasome. Immunity. 2013;39(1):123–35.
Weng L, et al. The E3 ubiquitin ligase tripartite motif 33 is essential for cytosolic RNA-induced NLRP3 inflammasome activation. J Immunol. 2014;193(7):3676–82.
Labzin LI, et al. Antibody and DNA sensing pathways converge to activate the inflammasome during primary human macrophage infection. Embo J. 2019;38(21):e101365.
Zhang J, et al. TRIM32 protein modulates type I interferon induction and cellular antiviral response by targeting MITA/STING protein for K63-linked ubiquitination. J Biol Chem. 2012;287(34):28646–55.
Yang Q, et al. TRIM32-TAX1BP1-dependent selective autophagic degradation of TRIF negatively regulates TLR3/4-mediated innate immune responses. PLoS Pathog. 2017;13(9):e1006600.
O’Brien AD, et al. Genetic control of susceptibility to salmonella typhimurium in mice: role of the LPS gene. J Immunol. 1980;124(1):20–4.
Tötemeyer S, et al. Sublethal infection of C57BL/6 mice with salmonella enterica serovar typhimurium leads to an increase in levels of toll-like receptor 1 (TLR1), TLR2, and TLR9 mRNA as well as a decrease in levels of TLR6 mRNA in infected organs. Infect Immun. 2005;73(3):1873–8.
Wang G, et al. TLR2 Promotes Monocyte/Macrophage Recruitment Into the Liver and Microabscess Formation to Limit the Spread of Listeria Monocytogenes. Front Immunol; 2019. 10.3389/fimmu.2019.01388https://doi.org/10.3389/fimmu.2019.01388.
Nguyen BN, et al. TLR2 and endosomal TLR-mediated secretion of IL-10 and immune suppression in response to phagosome-confined listeria monocytogenes. PLoS Pathog. 2020;16(7):e1008622.
Gogoi H, Mansouri S, Jin L. The age of cyclic dinucleotide vaccine adjuvants. Vaccines. 2020;8(3):453.
Machata S, et al. Lipoproteins of listeria monocytogenes are critical for virulence and TLR2-mediated immune activation. J Immunol. 2008;181(3):2028–35.
Funding
This work was supported by Beijing Natural Science Foundation (5222034) and National Natural Science Foundation of China (82002115, 82002116).
Author information
Authors and Affiliations
Contributions
XOY, PL, have designed and conceived the experiments equally in this work. YJ, DK, YP and XOY, conducted the data analysis, wrote and revised the manuscript. XOY and PL performed the experiments mainly. YZ and HJ contributed and shared some experimental materials, regents and instruments. QL and WH and conducted the formal analysis. YP and HH revised and edited the manuscript.
Corresponding authors
Ethics declarations
Ethical approval and consent to participate
All animals used in this study were housed at the Academy of Military Medical Sciences (AMMS) animal center in Beijing, China. Animals were cared for in accordance with the approved principles of laboratory animal care in China. All experimental procedures were approved by the Institutional Animal Care and Use Committee of the AMMS (IACUC-DWZX-2021-023).
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
Validation of Trim32 knockout. (A) Diagram of trim32 knockout. (B) Gene sequencing validation of trim32 knockout. (C) Western blot validation of trim32 knockout. Data were a representative of three independent experiments. Figure S2. Trim32 deficiency did not affect the components of innate immune cells. (A) Schematic illustration of the gating strategy. (B) Relative proportions of innate immune cells in the spleen and liver. Shown were mean ± SD of 3 mice per experimental group. Data were a representative of three independent experiments
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
OuYang, X., Liu, P., Zheng, Y. et al. TRIM32 reduced the recruitment of innate immune cells and the killing capacity of Listeria monocytogenes by inhibiting secretion of chemokines. Gut Pathog 15, 32 (2023). https://doi.org/10.1186/s13099-023-00558-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13099-023-00558-9