Abstract
Background
Wild edible plants (WEPs) are usually considered to constitute all plant resources that are neither cultivated nor domesticated but are used as nutritional supplements by local people. WEPs play a vital role in ensuring food and livelihood security for countless families and communities around the world. The objective of the study was to assess and document wild edible plants used by communities in the Arsi Robe district as food.
Methods
Semi-structured interviews, market surveys, and guided field walks were employed as data collection tools. The data were analyzed using preference ranking, direct matrix ranking, and Jaccard’s similarity index.
Results
The present study revealed various types of wild edible plants consumed by local communities in the Arsi Robe district of the East Arsi Zone. This could be justified by the documentation of 36 different wild edible plants in the study area. These WEPs belong to 31 genera and 25 families. Most of the growth forms of the edible wild plants in the district were shrubs (16, 44.44%) and trees (14, 38.88%). Amaranthus caudatus and Bridelia micrantha are the most preferred WEPs in the study area. The finding of the study also revealed that Lepidotrichilia volkensii and Premna schimperi are among the novel WEPs that had not before been documented as food items in other areas. Olea europaea subsp. cuspidata, Ficus sycomorus, Cordia africana, and Ficus sur are species with multiple uses. Agricultural expansion, charcoal production, the construction of different materials, making agricultural tools, deforestation, and other factors were the top prioritized threats affecting the abundance and diversity of wild edible plants.
Conclusion
Along with the sustainable utilization and conservation of the existing WEPs of the study district, priority should be given to the urgent collection, domestication, and cultivation of multiuse wild edible plant species such as Olea europaea subsp. cuspidata, Ficus sycomorus, Cordia africana, and Ficus sur in the study area.
Similar content being viewed by others
Introduction
Wild edible plants refer to plant species that are harvested from their natural habitats rather than being cultivated by humans on farms. The documentation of ethnobotanical information on WEPs is important from a scientific point of view, especially in the present day and in the age of rapidly changing societies, declining plant diversity, and the loss of traditional knowledge about the uses of these wild plants [1]. Without a question, WEPs have been valuable additions to a diet that is well-balanced, but global consumption has been declining and is currently at an all-time low [2]. Reduced consumption of WEPs leads to an unhealthy and unbalanced diet, which has been connected to a number of diseases and health issues as well as higher death rates, all of which can have an impact on the economy of a country [2].
There is a long tradition of using wild edible plants for nutrition in Africa. Different ethnobotanical studies in Africa have indicated that WEPs are an essential component of many diets. A study conducted in Kenya revealed that 73 WEPs are used by local communities in Turkana County [3]. One hundred WEPs were documented in the Teso-Karamoja region of Uganda [4]. Ethnobotanical studies conducted in the region also indicated that all sampled communities do not practice the consumption of WEPs, although extremely high levels of severe food insecurity are observed [3]. This could be due to the diminishing knowledge of WEPs, seasonal availability, and/or lack of awareness [5].
Throughout human history, communities around the world have used approximately 7000 WEPs as food [6]. Gathering and consuming edible wild plants is an example of a cultural activity that is still practiced worldwide and is crucial to food security [7, 8]. In Ethiopia, 413 WEPs belonging to 224 genera and 77 families were identified and documented in a review by Lulekal et al. [9]. In Ethiopia, these WEPS are used during both abundance and scarcity periods [10]. Approximately 70% of the WEPs observed in the lower river valley of the Omo zone are consumed during periods of severe food deprivation and famine [10]. In addition to being directly used for snacks and/or side dishes [10], the WEPs offer an opportunity for trade, which, with the right support from extension services [11] and an all-around supportive environment, can result in increased income and improved livelihoods [12].
However, for various socioeconomic reasons, indigenous populations underuse and/or disregard many of these plants [13]. Additionally, WEPs are underappreciated by various indigenous groups because in some Ethiopian communities, they are regarded as foods of the poor [14,15,16] and the information associated with them is passed down orally [17]. The diversity of WEPs was reported to gradually decrease due to agricultural expansion, overexploitation of fuel wood collection, and overgrazing [17,18,19]. The loss of these WEPs could risk the food security [20]. Furthermore, ethnobotanical knowledge and related indigenous knowledge are fast vanishing; thus, future WEPs nutritional analyses and use recommendations should document and disseminate this knowledge [12].
Numerous ethnobotanical studies have been carried out in Ethiopia, with a primary focus on evaluating the medical benefits of these plants. As a result, most ethnobotanical studies have not considered how various Ethiopian communities use wild plants for food. Of the few studies on edible wild plants, Ashagrie et al. [21] reported that Burji communities consume 46 wild plants, and 88 wild edible plants were identified in a related study by Dejene et al. [22] that was carried out in several regions of Ethiopia. Recently, 43, 54, 59, and 64 WEPs were recorded in the Ensaro, Dibatie, Raya, and Soro, districts, respectively [18, 19, 23, 24]. More recently, Fassil et al. [12] documented 39 WEPs used by Awi Agaw communities.
Despite the widespread availability and use of WEPs in Ethiopia, nutritional analysis, domestication, and conservation measures are scarce [9, 25]. This is demonstrated by the fact that many species are harvested from wild resources, with minimal attention given to cultivation and conservation [25].
Given their nutritional, medical, economic, and other benefits, as well as the threat they face today, WEPs in Ethiopia still require documentation and further research, even if certain studies have attempted to document these plant species in various areas. For the conservation and use of WEPs in their surroundings, the rural inhabitants of the Arsi Robe district rely in part on their traditional knowledge. The study district is well known for the diversity of WEPs and the cultural diversity of WEP uses. Although WEPs are widely available and used in the district, thus far, there has been no research on the ethnobotany of WEPs or the knowledge associated with them that has been documented. Furthermore, for future conservation and sustainable use of WEPs in the area, the site-specific identification, and preferences of the local WEP communities, the investigation of potential and serious threats of WEPs in the district have not yet been documented. Thus, the objectives of the current study were to (1) identify and document WEPs used in the Arsi Robe district, (2) assess the threats that currently exist to the WEPs of the study area, and (3) assess the most preferred and widely used WEPs in the Arsi Robe district, which may serve as a basis for conservation and nutritional analysis.
Materials and methods
Description of the study area
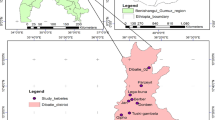
The Arsi Robe district is one of the 180 districts found in the Oromia Regional State in the Arsi Zone. The Arsi Robe district has an area of 1275 km2 (127,500 hectares). The district is bordered to the south by the Wabe Shebelle River, which separates it from the Gasera district of the Bale Zone, to the southwest by the Shirka district, to the west by the Tena district, to the north by the Sude and Dikisis districts, to the northeast by the Amigna district, and to the east by the Bale Gesgar district. It is located approximately 225 km southeast of Addis Ababa and 98.1 km east of Asella, the capital of the eastern Arsi zone (Fig. 1).
Climate
There are two main rainy seasons in the Robe district. These are ganna, the biggest rainy season, and arfaasaa, the little rainy season. The rainy season, during which almost 65% of the total amount of rainfall in the Robe district falls, begins in June and ends in September. The brief rainy season, which constitutes 35% of the total rainfall in the Robe district catchment, begins in March and ends in May.
In the Arsi Robe district, three main agroclimatic zones range from warm to cool thermal zones. Consequently, the district has 85% Baddaa or highland, 10% Badda-daree or midland, and 5% Gammoojjii or lowland climatic zones. The mean annual temperature is 16 °C, while the mean annual rainfall in the area is 1435 mm (Fig. 2).
Topography
The topography of the district is not uniform. The topographic features of the study area are mostly ragged and undulating, representing approximately 40% and 60%, respectively, of the total area of flat plains. The altitude of the study area ranges from 1200 m above sea level around Wabe Gorge Valley to 4000 m above sea level on Shato Mountain.
Vegetation of the district
The Arsi Robe district is rich in natural resources and natural vegetation that are a direct reflection of the climate of the town and the district. The district has different forests and grass types, broad-leaved plant species, and Eucalyptus (Baargamoo) trees. There are various indigenous tree species in the Robe district, such as Waleenaa (Erythrina brucei), Bakkanniisa (Croton macrostachys), Gaattiraa habashaa (Juniperus procera), Birbirsa (Afrocarpus falcatus), and Harbuu (Ficus sur). However, it is essential to note that Eucalyptus tree plantations are predominant in Robe town and its surroundings in very large numbers. Most indigenous tree species have been significantly reduced due to frequent cutting of forests for crop cultivation, wood for the manufacture of charcoal, and for the building of houses and fences. This large amount of clearing and burning of trees has led to severe soil erosion and environmental degradation [26].
Population
Oromos are the predominant ethnic group that lives in the Arsi Robe district. According to the report of the Central Statistical Agency [27], the total population of the Arsi Robe district is 250,495, of which 124,805 are females and 125,690 are males. Afan Oromo is the predominant spoken language in the district.
Study sites and informant selection techniques
Study sites and informants were selected based on information collected from the Arsi Robe district administration office, the health office, the agricultural office, and other people in the study area through a reconnaissance survey before data collection. Of the 34 total Kebeles in the district, six Kebeles—Sebiro Chefe, Jawi Sire, Jena Gedemsa, Sedika Burka, Indato Weltae, and Qilisa Dembel—were sampled using purposive sampling techniques so that all agroecological conditions were met to ensure the availability of wild edible plants in the respective agroecosystems.
A total of 387 informants were selected (80 from Jawi Sire, 104 from Sebito Chefe, 62 from Sedika Burka, 56 from Jena Gedemsa, 43 from Indeto Weltae, and 42 from Qilisa Dembel). Of the total informants, 248 were men and 139 were women (Table 1). Of these, 377 informants were randomly selected, while 10 key informants were selected purposefully based on recommendations from local people.
Ethnobotanical data collection
Ethnobotanical data were collected from February 2023 to May 2023. The data were compiled through semi-structured interviews during guided field walks (Fig. 3) and group discussions. The interviews used a semi-structured checklist of topics, which was prepared in English and translated into the Afaan Oromoo. The interviews covered the following main components: (a) personal data of the informants, e.g., name, address, age, gender, and literacy status; (b) respondent knowledge of local names of the plants and parts used and the conditions of the plant parts used (fresh/dried); and (c) the uses of wild edible plants other than food. Photographic records were also taken in the field to capture field sites, plants, and other useful information, such as preparation methods and storage methods.
All semi-structured interviews were followed by guided walks-in-the-forest, which allowed us to collect and identify traditionally used wild edible plants. During these guided field walks that were guided by informants, all relevant data, including the vernacular names of plants, habits, and parts used, were collected. Additionally, focus group discussions were conducted in each sampled Kebeles with key informants.
Market surveys
Three markets were selected from the district. These markets are found in Robe Didea town, Sedika town, and Endeto town. A market survey was conducted to record the names of wild edible plants within costs and other aspects of the edible parts being sold. Direct observations in the market and interviews on the aspects of wild plant products were conducted to identify wild edibles sold on the market following the methods described by [28] and [29]. The survey was carried out to assess the variety and amount of wild edible plants supplied from the area and to record information that wild edibles are marketable for the use of remedies or for any other use.
Collection and identification of wild edible plants
The reported WEPs were collected from natural vegetation during the field walks. Preliminary identification was done at the field. Moreover, we also recorded the local names of the plant species to aid the identification using Flora of Ethiopia and Eritrea [30, 31] and validated using databases such as IPNI [32] and POWO [33]. Finally, all the identified specimens were collected, processed, and stored in the mini herbarium of Madda Walabu University.
The ethnobotanical data collected through interviews and FGDs were analyzed through descriptive statistics such as frequency and percentages using Excel spread sheets. The results are presented in tables and figures. In addition, preference ranking, direct matrix ranking, priority ranking, and Jaccard similarity coefficient were employed to analyze ethnobotanical data.
Preference ranking
Preference ranking of the WEPs was performed following Martin [28] and Cotton [34]. In the preference ranking process, the values (1–10) were given by ten key informants, each value was summed, and the average was taken to determine the preference of one over the other. For the preference ranking exercise, informants were given the eight most preferred wild edible plants based on their taste and ranked according to their sweetness. The wild edible plant that was believed to have the sweetest fruit was given the highest value of 10, and the plant with the least sweetest fruit was given a value of 1. Preference classification was determined based on the total score of each species. The total rank of preferences was obtained by adding the number given by each informant.
Direct matrix ranking
In addition to food, local people have used edible wild plants for a variety of other purposes, such as fuel wood, fences, house construction, medicine, farm implements, and furniture. The seven use values recorded for six different plant species were totaled and ranked. Direct matrix classification was performed to compare the multipurpose properties of wild edible plants commonly reported by informants following Cotton [34]. Based on the relative benefits obtained from each plant, eight multipurpose (multiuse) WEP species were listed among the total number of wild edible plants and seven use diversities of these plants. The purpose is to assess their relative importance to local people and the extent of existing threats related to their use values. Based on the service categories, 10 KIs were asked to assign use values for each attribute. The list of these attributes included medicinal, fuel wood (firewood and charcoal), construction, farm and household tools, feed, live shade, honeybee forage, and soil and water conservation. The use values (0–5) are given. Ten key informants were chosen to assign use values to each attribute (4 = best, 3 = very good, 2 = good, 1 = less, 0 = none). The average use values given for each multipurpose species in each use category by each KI were recorded, and the values were summed for each species and ranked.
Priority ranking
Ten KIs also ranked as the priority of the main recorded threat factors to these plants based on their degree of destruction. Values of 1–17 were given; all values given by each KI were summed to report the most concerning factor.
Jaccard similarity coefficient
The Jaccard similarity index (JSC) was used to calculate the degree of similarity and difference in composition [35] of the WEPS among the Kebeles in Arsi Robe and with WEPs documented from other different study areas or districts. The Jaccard similarity index was calculated as follows.
where
JSC = Jaccard's similarity coefficient.
c is the number of species shared by or common to any pair compared in the Kebeles and Arsi Robe district and other districts, and b and c are the numbers of WEPS reported solely in one of the compared groups (b for one group and c for the other).
Results
Sociodemographic characteristics of the informants
A total of 387 informants were selected for the study, and all informants willingly participated in the study, resulting in a response rate of 100%. Among the selected informants, 248 (64%) were men, and 139 (36%) were women (Table 2). Most of the informants were aged 31 to 40 years (114, 29.46%), followed by those aged 20 to 30 years (95, 24.55%). In terms of ethnicity, Oromo (344, 89%) was the highest ethnic group, while other ethnic groups (43, 11%) accompanied the other ones. Regarding marital status, the majority (312, 80.50%) of the informants were married, followed by single individuals (37, 9.50%). In terms of religion, Muslims made up the majority (190, 49.00%), followed by Orthodox (143, 37%). In terms of educational status, 64 (16.50%) of the informants had no formal education, while 252 (65%) had a primary level of education (Table 2).
Wild edible plants (WEPs) of the district
Thirty-six wild edible plants used by communities in the Arsi Robe district were documented. The WEPs collected belonged to 31 genera and 24 families. Rosaceae and Anacardiaceae were represented by four and three species, respectively. Potentilla spp. Deolf, Rubus apetalus Poir, Rubus steudneri Schweinf, and Rosa abbyssinica Lindl. are plant species belonging to the Rosaceae family, while Anacardiaceae, represented by Lannea rivae (Chiov.) Sac, Searsia natalensis (Bernh. exKrauss) F.A. Barkley, and Sclerocarya birrea (A.Rich.) Hoechst. The other families were represented by one or two species each.
Habit or growth form of WEPs
Among the collected plant species, most were shrubs (16, 44.44%), followed by trees (14, 38.88%) (Fig. 4).
Edible parts
The local people in the study area consume different parts of wild plants. Most plants consumed are ripe or cooked. The majority of plants (29, 78.4%) consumed uncooked fresh ripe fruits (Fig. 5), followed by eating leaves in cooked or raw form (5, 13.5%), as indicated in Fig. 5. The result also revealed that herbs were mostly consumed for their leaves, whereas trees and shrubs were the main contributors of fruits. For instance, Cordia africana, Ficus sycomorus, Mimusops kummel, and Opuntia ficus-indica fruits were widely consumed in the study area (Fig. 6).
Main collectors and consumers of WEPs in the district
Children or shepherds (178, 45.99%) and women (113, 29.20%) were the main gatherers, followed by other members of the household (57, 14.73%) and men (26, 6.72%). The majority (307, 79.33%) reported that WEPs were consumed by all household members. In terms of consumption mode, 31 species were consumed raw or without further processing, three were consumed cooked and two species were consumed either raw or cooked. Children in the study area mostly consume raw fruits of Embelia schimperi. Similarly, fruits of Opuntia ficus-indica, Syzygium guineense subsp. Guineense, and Mimusops kummel widely consumed by communities of the study area. In the study area, the cooked leaves of Urtica simensis (Doobbii) are widely consumed by communities of the Arsi Robe district. The entire task of gathering, preparing, and serving meals made with U. simensis to the family falls on women and girls. Because the leaves are stinging, hands covered by protector and harvested with a knife. After collection, the leaves crushed using any material to prevent the burning sensation of the leaves. The leaves can then be crushed by hand and boiled for approximately twenty-five minutes. When boiled the leaves have to be grid one more time to become smooth. Then, the smooth sauce will be mixed in cold water and boiled for about 40 min followed by adding barley powder and then the cooled sauce is served with injera. Injera is a traditional dish in some parts of Ethiopia which is made up of Eragrostis tef.
Seasons for harvesting WEPs
The key informants explained that the time or season and the frequency of harvesting vary from plant to plant depending on the availability of WEPs. It varies from place to place due to ecological and seasonal conditions. They were more abundant during the main rainy season (Ganna) from June to August, during the spring season from September to November, and during autumn from March to April, while they were less abundant during the dry season (winter) from December to February (Table 3).
Consumer perceptions about WEPs
The perceptions and/or attitudes of the communities and informants toward WEPs were not the same, as there were different local taboos associated with the consumption and utilization of WEPs in the Arsi Robe district. Table 4 presents herein details some of the connotations of the surveyed informants related to the consumption and utilization of wild edible plant species. Most (275, 71.06%) of the indigenous peoples of the district occasionally consider WEPs to be famine foods or foods for conditions of starvation, as the general public consumes most of the WEPs as snacks, supplements, or refreshment foods. Approximately 284 (73.39%) of the informants reported using WEPs as supplementary food, while the remaining 103 (26.61%) reported using WEPs as regular food or as famine meals. According to the responses of 87 (22.48%) of the informants surveyed, the consumption of wild edible plants carries the connotation of belonging to a lower stratum of society and is considered an insult due to a lack of knowledge (Table 4).
Preference ranking
In the preference ranking process, the values (1–10) were given by ten KIs, each value was summarized, and the average was taken to determine the preference of one over the other. The wild edible plant that was believed to have the most multiple uses was given the highest value of 10, and the one with the lowest preference ranking was determined based on the total score of each species. The total rank of preferences was obtained by adding the number given by each informant. Consequently, Amaranthus caudatus, Bridelia micrantha, Capparis tomentosa, and Carisa spinarum were the five WEP species preferred by the key informants (Table 5).
Direct matrix ranking
Cordia africana, Embelia schimperi, Ficus sur, Ficus sycomorus, Myrsine africana, Olea europaea subsp. cuspidata, and Searsia natalensis were the eight most multi-use wild edible plant species in the Arsi Robe district. They are used for various purposes, such as forage/fodder, medicine, fuel wood (charcoal and firewood), material culture, and miscellaneous uses. Based on the relative benefits obtained from each plant, seven multipurpose WEP species out of the total wild edible plants and nine use diversities of these plants were listed. The purpose is to assess their relative importance to the local population. On the basis of the data collected from the informants, ten multiuse species were chosen in the Arsi Robe district. These attributes included medicinal use (MU), fuel wood (FW), construction (C), charcoal production (CP), agricultural tools (AT), cattle feed (CF), bee forage (BF), shade (S), and furniture making (FMF). The average use values given for each multipurpose species in each use category by each KI were recorded, and the values were summed for each species and ranked (Table 6).
Marketability of WEPs
The marketability and price of wild edible plants in Arsi Robe were identified. People collect wild fruits from the wild and sell them directly to retailers in formal markets, and people profit by selling them to consumers in towns at a better price. Mimusops kummel fruits were found to be sold in the local market of the study area to support household income. A market survey and an interaction with business residents in the study area in local markets showed that the average price of WEP fruits ranges from 10 birrs to 15 Ethiopian birrs per cup (Fig. 7).
Priority ranking of threats to WEPs in the district
A priority ranking of the main recorded threats to wild edible plants was also performed with 10 KIs based on their degree of destruction. Values ranging from 0 to 5 were given. Consequently, agricultural expansion, charcoal production, the construction of different materials, the production of agricultural tools, deforestation, human settlement, selective harvesting, the removal of woody plants for different purposes, and overexploitation were the top nine threats affecting the abundance and biodiversity of wild edible plants in the district that need priority consideration (Table 7).
Jaccard's similarity coefficient
The Jaccard similarity coefficient (JSC) was used to calculate the degree of similarity and the difference in composition of the WEPS among the six selected Kebeles in the Arsi Robe district. The Jaccard similarity index for Jawi Sire and Sebro Chefe was 0.80, and that for Jawi Sire and Sedika Burka Kebeles was 0.77. When collected and documented WEPs from the Arsi Robe district were compared with similar plants documented by other researchers from other study areas, the Arsi Robe district had a higher Jaccard similarity index with the Midakegn district (18.1%), followed by the Ensaro district (16.8), and the least similarity was observed with the study conducted in the Quara district (6.5%) of northwest Ethiopia (Table 9).
Data novelty
The relevant ethnobotanical studies carried out in Ethiopia were compared with the data in order to assess any potential novelty in the use of WEPs. According to our comparative analysis with the previous ethnobotanical studies conducted in Ethiopia, 5 WEPs, including Lepidotrichilia volkensii, Nicandra physalodes, Potentilla spp., Premna schimperi, and Salvia nilotica, had not before been documented as food items in other areas (Table 8). It was previously known that the remaining 31 species were consumed in different parts of Ethiopia.
Discussion
The results of the current study demonstrate that there is wide use of edible wild plants in the Arsi Robe District of the Oromia Regional State. Thirty-six WEPs were documented in the study area (Table 8). This number is comparable to those reported from other districts in Ethiopia. Tahir et al. [17] documented 41 species in the Mieso district, [11] collected 34 species in the Bereket forest, [36] documented 36 species in the Quara district, and [37] documented 36 species in the Tach Gaynt district. However, studies by [16, 18, 19, 23, 24] in Ethiopia and another study from Turkana County of Kenya [3] and Morocco [20] reported higher numbers of WEPs than did the current study. The variation could be attributed to variations in agroclimate, study area size, cultural context, and research effort, as described by [16] and [24]. Rosaceae and Anacardiaceae were the dominant families that contributed the highest number of WEPs in this study. Rosaceae contributed to four wild edible plants, as reported by [16] and [38]. The finding that Anacardiaceae was the highest contributor of nutritionally useful species is in agreement with a previous study by [21]. Unlike in the present study, the Moraceae family was reported to contribute the highest number of WEPs in the studies by [18] and [39]. Moraceae also reported as the highest contributor of WEPs with six species in the study conducted by [12]. Rubiaceae [19] and Malvaceae [17] were also reported to be the highest contributors to WEPs.
The number of WEPs reported in the study area could provide some essential nutrients, and they can contribute to the food security and promote a balanced diet as stated by [18]. The use of WEPs can be a benefit in enhancing food security [40]. It provides a wide range of nutrients, for example, C. spinarum in the present study contains high minerals such as potassium and calcium [41]; likewise, C. africana is also rich in vitamins A, and C [42], potassium, calcium, and iron [43]. Their inclusion in the diets can improve overall health and well-being [14]. The food security can be in jeopardy and poverty may rise as the plants which are mainly collected from the wild are being diminished [44]. A possible reason for threats on WEPs could be because of a lack of knowledge about the importance of wild plants for daily uses and food security [45].
According to Ngome et al. [44], WEPs have four different roles in local diets, food security, and nutrition. The first is the daily net function, which summarizes how WEPs are used and contribute to family food demands on a daily basis. WEPs serve as safety nets during famines and are eaten as famine foods [10], which is their second contribution. The third dimension is the utilization of WEPs to generate cash income [16, 19, 46, 47]. This is an indirect contribution to food security and nutrition because the money made enables households to buy other essential foods such as proteins or staples [44]. The fourth role is their significance in the local culture. Food security incorporates the crucial component of food preferences, even if the nutritional results of food security are essential for both physical and cognitive function [48].
The documented WEPs in the study area also play a vital role in health-related issues. There is a strong association between the health and nutrition of WEPs [40]. In the study area, Embelia schimperi, Myrsine africana, and Opuntia ficus-indica are among species reported to have medicinal properties. Several studies in Ethiopia have reported that WEPs are used for medicinal purposes [21, 49]. The fruits of M. africana were used as food and medicine to expel ascaris in children [21]; likewise, consuming fruits of Grewia ferruginea and C. africana were reported to prevent constipation in children [39].
The study area shared 19 wild edible plant species with the Midakegn district [16], 16 with the Ensaro [18] and Soro [24] districts, and 13 with the Dibatie [19] and Raya Azebo districts. The higher similarity of wild edible plants in the study area to those in the Midakegn district [16] might be due to vegetation cover, indigenous knowledge practices among communities, and language and cultural similarities. The least similarity was observed between Chilga district [58] and Quara district [36], as they shared 9 and 5 overlapping wild edible plant species, respectively (Table 9).
In the present study, the majority of wild edible plants identified were shrubs and trees, indicating that most of the WEPs in the study area are woody species. This finding aligns with previous research findings by [18, 21, 57]. Similarly, Ojelel et al. [4] reported dominant shrub growth habits in Uganda. In contrast, trees were reported as the main sources of wild food in different parts of Ethiopia [17, 24, 39, 66] and other localities, such as Nepal [69, 70] and Uganda [71]. Herbs were rarely reported as most of the growth forms for WEPs. However, only a few studies, mostly from Asia, have reported herbs as the most prevalent growth form of WEPs throughout various study areas [72,73,74,75]. The dominance of growth form for the highest contributor of WEPs could be attributed to the use categories of plant parts. For example, the dominance of herbs is due to the high number of edible succulent stems and leaves that are used as food [75]. Furthermore, agroecology and local culture in a given area contribute to this variation [76].
The majority of the plants in the district consumed for their fruits which is raw. For instance, raw fruits of C. africana [62, 65], Dovyalis abyssinica [53, 58], Ficus sur [11, 37], and Syzygium guineense subsp. guineense [22, 24] widely consumed in the study area and other parts of Ethiopia. The preference of raw fruits could be due to the sweetness and high nutritional content, good taste, and high sugar concentration of the fruits [23, 77]. Moreover, the ease of consuming fruits raw without processing could be a reason why fruits are consumed widely [19]. Many other studies conducted in Ethiopia [16, 18, 19, 23, 24, 39] and elsewhere in the world [3, 4, 69, 78] also revealed the dominance of wild edible fruits. Children were the main consumers of fruits, particularly when herding animals in distant areas of homesteads. In contrast, women were often responsible for gathering and preparing green vegetables for domestic use at home. This result is consistent with recent studies [16, 18, 23].
According to the direct matrix ranking analysis, F. sur, O. europaea subsp. cuspidata, and C. africana were identified as the most significant in terms of their multiple usage values. The multi-used plant species in Ethiopian communities according to studies performed in different regions of the country and similar to the current findings are O. europaea subsp. cuspidata and C. africana [11, 16, 18, 19, 24, 37, 65]. As a result, O. europaea subsp. cuspidata and C. africana have strongly declined in natural forests, as evidenced by the results of the current study and other studies conducted in Ethiopia. This indicates the urgency of conserving O. europaea subsp. cuspidata and C. africana due to their multiple values within the country.
In addition to being used for household consumption, M. kummel fruits were sold in the local market of the study area to support household income. This result is in agreement with the reports of previously conducted studies in different parts of Ethiopia [16, 19, 46, 47]. In a similar study conducted in Ethiopia, Arisaema schimperianum [21], Ximenia americana [19, 36, 47], Balanites aegyptiaca [23, 36, 39, 49], and Syzygium guineense subsp. guineense [24] are plant species sold in open markets. Temarindus indica, Balanites aegyptiaca, and Adansonia digitata have been reported as plant species exported to the Republic of Sudan [36, 39]. The authors also reported that Balanites aegyptiaca was the most expensive species exported to Sudan.
Literature review on the phytochemical, nutritional, and antioxidant properties of frequently cited WEPs
Most plant species identified in the study area for use as food have been reported elsewhere in Ethiopia. Here, we discuss some of these plants that are most commonly reported by local communities in the study area during ethnobotanical surveys and other similar studies conducted in Ethiopia. Carissa spinarum emerged as the most frequently mentioned plant among wild edible plants in Ethiopia, with 31 authors citing this species, followed by Cordia africana, with 30 authors citing the plant.
The most effective method used to determine whether people's nutritional needs are being successfully met is through nutritional assessment [79]. To eradicate hunger, poverty, and the burden of malnutrition in developing nations, nutritional analysis of native wild edible fruits provides quality, quantity, and evidence-based information for future research, planning, commercialization, and utilization [80]. Different studies have demonstrated that wild fruits have great potential as sources of vital nutrients, especially for growing children who are prone to malnutrition [5]. Children's participation in the collection and consumption of WEPs may be a sign that these resources are still valued for food security and are known to younger generations. To spread awareness among the next generation, schools and other institutions should take into account the significance of WEPs for food security.
Carissa spinarum
Carissa spinarum is a tropical plant that produces delicious crimson berry fruit called by different names in Ethiopia. The Oromo communities in Ethiopia are called Agamsa [11, 17], the plant is known by Agama in the Burji language [21], and the Amhara and Tigray communities are called Agam [18, 23, 37]. The native range of this species is Africa to Indo-China and Australia to New Caledonia [33]. It is a scrambling shrub or tree that belongs to the Apocynaceae family. Different parts of C. spinarum are traditionally used in various areas of Ethiopia to cure diseases. The root is used to treat malaria, gonorrhea, and diarrhea [81,82,83]; the leaf is used to treat headache, stomachache, and skin diseases [81, 84]; and fresh bark and seeds are used to treat fibril illness [71].
Phytochemical analysis by many research groups has shown that fruit extracts are rich in polyphenols, flavonoids, and flavanones [85]. A recent review by Dhatwalia et al. [86] reported that 93 compounds from C. spinarum were extracted, including 27 polyphenols, 27 lignans, 23 terpenoids, 8 steroids, 2 coumarins, and 6 cardiac glycosides. Nazareth et al. [87] reported that the phenolic and flavonoid contents were 273.20 mg GAE/100 g and 453.78 mg QE/100 g, respectively, in ripe fruits. Additionally, fruit extracts exhibit high contents of syringic acid, resveratrol, and quercetin in ripe fruits [87]. The antioxidant activity of the ripe fruit extract exhibited a higher potential to scavenge DPPH radicals, with an IC50 of 4.69 mg/ml [87]. In a similar study, Mundaragi and Thangadurai [88] reported that the radical scavenging activity of C. spinarum juice was 51.3 ± 3.1%. This may be attributed to the content of polyphenols and ascorbic acid present in fruits [88]. The fruits of C. spinarum possess a potential antidiabetic effect, corroborating the ethnomedical uses of the fruits [89].
The moisture content of the fruit ranges between 58.62 and 91.15% [41, 87, 89]. A study by Siyum and Meresa [41] revealed that 1.42% protein, 5.33% fat, and 4.92% fiber were present in the fruits of C. spinarum. The fruits of C. spinarum have also been reported to have a high mineral content. For example, [41] reported that the fruit has a high content of Fe (1.49), Ca (83.54), K (816.69), P (28.02), Mn (0.93), and Zn (0.798) mg/100 g. The authors also reported that the fruit extracts have a high percentage of constituents compared to the cultivated fruits such as banana and apple. The consumption of these fruits can also alleviate vitamin A deficiencies in areas affected by malnutrition [85]. In addition to its nutritional value, the consumption of fruits of C. spinarum promotes good health and well-being and reduces the risk of chronic diseases [86]. In a recent study, Mamoona et al. [90] formulated a beverage drink from C. spinarum fruit. The formulated beverage is a rich source of anthocyanins, phenolics, and antioxidants, which are essential for human health.
Cordia africana
Cordia africana is a tree of the family Boraginaceae that produces edible fruits. Its natural distribution ranges from South Africa to the Arabian Peninsula, Comoros, and Central Madagascar. It is a shrub or tree and grows primarily in the seasonally dry tropical biome [33]. The plant has multiple uses in Ethiopia, in addition to its food value. Local communities in Ethiopia use C. africana for construction, timber production, charcoal, and traditional medicine [81, 91]. Various ethnobotanical studies have reported that the plant is also one of the most threatened species in Ethiopia [92].
The nutritional analysis by [42] indicated that the fresh and dried fruits of C. africana were rich in vitamin C and vitamin A. Furthermore, the authors reported that the vitamin A and vitamin C levels were still good after drying and storage. The moisture content of fresh fruits of this plant species is reported to be 11.15–56.89% [43, 93], which has an impact on the concentration of total phenol [93]. The fruits of C. africana were reported to contain 8.1% crude protein, 0.69% crude fat, 3.86% crude fiber, 60.26% carbohydrate, and 277.97 kcal/100 g of energy [43]. Mineral composition analysis of C. africana fruits indicated that the fruit contained a higher concentration of potassium (2102.96 mg/100 g), followed by calcium (96.55 mg/100 g) and iron (28.51 mg/100 g). Furthermore, copper and zinc were also found at good concentrations [43]. The study also indicated that mineral concentrations were strongly influenced by geographic region.
The antioxidant power decreased by ferric acid, and the total phenols in the fruit extracts were 30.8 ± 1.45 mg Trolox equivalent 100 g−1 of fruit and 2317.0 ± 104.0 mg of gallic acid equivalent 100 g−1 of fruit [93]. Agroecology has been reported to have an impact on the concentration of antioxidants and the total phenol content [93]. Although the fruit of C. africana is widely eaten in different parts of Ethiopia, its nutritional, phytochemical, and antioxidant properties have not been widely evaluated.
Ficus sur
Ficus sur, commonly known as mulberry, is a widely distributed tropical African tree that belongs to Moraceae. F. sur is a productive multipurpose tree that occurs in cooler climates up to more than 2800 m above sea level. Adapted to a wide range of climatic conditions and soil types, the tree can grow to a height of at least 35 m, attains a breast height diameter of over 1 m, and is considered a keystone tree species [94]. F. sur is also a widely used plant in traditional medicine and wild edible food in Ethiopia. Approximately 26 authors mentioned that the plant is widely consumed in different parts of Ethiopia [24, 37, 47]. F. sur produces fruit throughout the year [95].
Phytochemical analysis indicated that the ripe fruits of F. sur possess alkaloids, flavonoids, saponins, glycosides, tannins, terpenoids, and anthocyanins [96,97,98]. F. sur fruits also contain metabolites with potential antioxidant activity [96]. An antioxidant analysis by [96] indicated that the fruit extract had a DPPH radical scavenging activity close to 7.3 ± 0.30 μg QE/mg of dry extract. Similarly, the FRAP value was 19.61 ± 0.22 μmol FeSO4 Eq/mg dry extract. In a similar study, Ogunlaja et al. [97] reported that the methanolic extract of the fruits showed (IC50 9.06 μg/mL) antioxidant activity. This could be attributed to the higher phenolic content and presence of epicatechin [97].
The results of the proximate analysis showed that F. sur fruits contain 88.8% moisture, 65.6% carbohydrate, 2% protein, and 4.7% fat [99]. The percentage protein and percentage lipid content of fruit pulp was 8.37% and 5.88% respectively [100]. The high level of carbohydrates in fruits indicates that they can serve as an immediate and alternative source of energy [97]. Nutritional analysis also indicated that the fruits were rich in Ca, Mg, Fe, Zn, Cu, Na, and Mn [99]. The presence of essential minerals in the fruits of F. sur indicated that it could overcome the lack of minerals and serve as a possible dietary supplement, especially in rural areas [96].
Syzygium guineense subsp. guineense
Syzygium guineense is a medium-sized or tall evergreen tree 15–30 m high. It is widely distributed in the tropical regions of Australia, Asia, and Africa [101]. The bark varies in subspecies and is grayish-white or silver mottled and smooth in young trees, turning rough, flaky, creamy, light gray, dark brown, or black in older trees [102]. Edible fruit has a very short shelf life and should therefore be consumed soon after harvesting; it is used to make beverages or vinegars [80].
A proximate analysis conducted by Sibiya et al. [80] revealed that the protein, fiber, carbohydrate, and fat contents of the fruit are 1.66, 3.84, 1.01, and 7.74 g/100 g, respectively. The results of the amino acid analysis indicated that the fruits have a lower amount of indispensable amino acids [80]. In a similar study, Tesfay et al. [47] reported that the proximate analysis of fresh fruit revealed 2.21 mg/100 g of fiber, 0.28 mg/100 g of fat, 1.48 mg/100 g of protein, and 26.47 mg/100 g of carbohydrates. Iron and zinc were also reported as constituents of the fruit, where the concentrations were 1.56 mg/100 g and 0.15 mg/100 g, respectively [47]. Mineral content analysis also revealed that the fruit is rich in Al (12 mg/kg), Ca (1725 mg/kg), Mg (1549 mg/kg), and 1243 mg/kg P [103]. The fruit also contained a significant amount of vitamin A (1.7 mg/100 g). In a similar study, Maregesi et al. [104] reported that 20,477 mg/100 g of calcium, 443 mg/g of potassium, 8392 mg/g of phosphorous, and 268.3 mg/g of iron and zinc were not detected. Possible reasons for the compositional differences could be differences in geography, soils, and climates, as well as differences in the analytical methods, maturity stages, and storage conditions of the fruit samples and intraspecies differences due to genetics [105].
A recent study by Asfaw et al. [106] indicated that drying and extraction methods have a profound impact on the biological constituents and antioxidant potential of fruits of S. guineense. For instance, compared with sun drying, vacuum freeze drying, and drying at room temperature, room drying methods preserve the total phenolic content, total flavonoid content, antioxidant potential, and ascorbic acid content [106]. The authors indicated that the total phenolic content of the dried fruit at room temperature was 1130.11 ± 32.33 mg/100 g and 1828.54 ± 6.74 mg/100 × g for Soxhlet and ultra-assisted extraction, respectively, while the total flavonoid content was 498.143 ± 4.93 and 1324.20 ± 13.31, respectively. In a similar study, Mapunda and Mligo [107] reported that the total phenolic content and total flavonoid content of fresh fruit samples were 137.37 ± 2.3 and 45.24 ± 0.4 mg/100 g, respectively.
Mimusops kummel
Mimusops kummel is an evergreen tree that can reach 30 m and has a diameter of more than 1 m, with a leafy and oval crown. The fruit is a hard 2-cm-long drupe that is pointed and orange yellow and contains one red‒brown seed [108]. The native range of M. kummel is Tropical Africa [33]. The phytochemical, antioxidant, and nutritional properties of M. kummel fruits are not well documented in the literature. However, there are few available papers on this topic. A preliminary phytochemical screening of 80% ethanol from M. kummel fruits revealed the presence of alkaloids, saponins, tannins, phenols, terpenoids, and flavonoids. Steroids, anthraquinones, and glycosides are absent in fruit extracts [109]. The proximate analysis of the M. kummel fruits revealed that the fruits consisted of 1.62 mg/100 g of fat, 2.19 mg/100 g of protein, 20.46 mg/100 g of fiber, and 80.49 mg/100 g of carbohydrate [5]. The authors also indicated that phosphorus, iron, and vitamin C were 31.28 mg/100 g, 2.95 mg/100 g, and 148.6 mg/100 g, respectively.
Dovyalis abyssinica
Dovyalis abyssinica is a spiny shrub or tree found mainly in Ethiopia and Malawi [108]. This plant is often found in upland rainforests, dry evergreen forests, riverbanks, and sometimes more open forests [15]. Most people who gather and consume raw D. abyssinica fruits are children who like the pleasant, sweet, sour flavor of the fruits [15].
Proximate analysis of dried fruit pulps demonstrated that D. abyssinica has 3.11 g/100 g of protein, 2.55 g/100 g of fat, 16.08 g/100 g of fiber, and 54.77 g/100 g of carbohydrate [110]. A similar study by Jiru et al. [111] indicated that the protein, fat, and fiber contents were approximately 3.01%, 1.46%, and 2.11%, respectively, on the basis of dry weight. The fruit also contained Ca (120.18 mg/100 g), Na (10.86 mg/100 g), Fe (2.09 mg/100 g), K (183.39 mg/100 g), and Mg (5.63 mg/100 g). On a fresh basis, the Ca and Fe contents of the fruit were 15.19 and 1.88 mg/100 g, respectively.
The fruits of D. abyssinica have been reported to be excellent sources of ascorbic acid, from which 109.07 mg/100 g was obtained [110]. In addition, the authors reported that the total phenolic content was 1128 mg GAE/100 g. In a similar study by Jiru et al. [111], the total phenolic content in the fruits of D. abyssinica was 196.36 mg GAE/100 g. The extract of the fruit of the plant species exhibited moderate antioxidant potential compared to that of Pyrus communis fruits, which can be used in food products to prevent lipid oxidation [110].
Conclusions
The present study indicated that local communities in the Arsi Robe district used wild edible plants for consumption. Thirty-six wild edible plants belonging to 31 genera and 25 families were documented in the study area. In addition to their use as food, wild edible plants serve local communities for medicinal purposes, fuel wood, construction, and charcoal production. This multipurpose use places these plants under serious threat. Ficus sur, Olea europaea subsp. cuspidata, and Cordia africana are among the threatened species that need proper conservation to save them from further loss. Domestication of these WEPs with the integration of local communities is a wise approach to conserving plant species. Some of the WEPs in the present study have been reported elsewhere in Ethiopia, which could be due to their availability and nutritional value. For example, Carisa spinarum, Cordia africana, Ficus sur, Syzygium guineense, Mimusops kummel, Dovyalis abyssinica, Opuntia ficus-indica, Rosa abbyssinica, Grewia villosa, and Grewia bicolor are frequently reported WEPs. Therefore, a detailed nutritional, antinutritional, and antioxidant assessment of these plant species is vital to aid one of the SDGs, which calls for the end of hunger.
Availability of data and materials
All the data used to support this study are presented in the tables and figures in this manuscript.
Abbreviations
- CSA:
-
Central Statistical Authority
- IPNI:
-
International Plant Names Index
- JSC:
-
Jaccard similarity coefficient
- KI:
-
Key informants
- POWO:
-
Plants of the World Online
- WEPs:
-
Wild edible plants
References
Łuczaj Ł. Descriptive ethnobotanical studies are needed for the rescue operation of documenting traditional knowledge. J Ethnobiol Ethnomed. 2023;19:37.
Hussain ST, Muhammad S, Khan S, Hussain W, Pieroni A. Ethnobotany for food security and ecological transition: wild food plant gathering and consumption among four cultural groups in Kurram District, NW Pakistan. J Ethnobiol Ethnomed. 2023;19:1–35.
Oduor FO, Kaindi DWM, Abong GO, Thuita F, Termote C. Diversity and utilization of indigenous wild edible plants and their contribution to food security in Turkana County, Kenya. Front Sustain Food Syst. 2023;7:14.
Ojelel S, Kakudidi EK. Wild edible plant species utilized by a subsistence farming community in Obalanga sub-county, Amuria district. Uganda J Ethnobiol Ethnomed. 2015;11:1–8.
Fentahun MT, Hager H. Exploiting locally available resources for food and nutritional security enhancement: wild fruits diversity, potential and state of exploitation in the Amhara region of Ethiopia. Food Secur. 2009;1:207–19.
Carvalho AM, Barata AM. The consumption of wild edible plants. In: Wild plants, mushrooms and nuts. Wiley; 2016. p. 159–98.
Pieroni A, Soukand R. Ethnic and religious affiliations affect traditional wild plant foraging in Central Azerbaijan. Genet Resour Crop Evol. 2019;66:1495–513.
Leon-Lobos P, Diaz-Forestier J, Díaz R, Celis-Diez JL, Diazgranados M. Ulian Patterns of traditional and modern uses of wild edible native plants of Chile: challenges and future perspectives. Plants. 2022;11:744.
Lulekal E, Asfaw Z, Kelbessa E, Van Damme P. Wild edible plants in Ethiopia: a review on their potential to combat food insecurity. Afr Focus Brill. 2011;24:71–122.
Teklehaymanot T, Giday M. Ethnobotanical study of wild edible plants of Kara and Kwego semi-pastoralist people in Lower Omo River Valley, Debub Omo Zone, SNNPR. Ethiopia J Ethnobiol Ethnomed. 2010;6:1–8.
Kidane L, Kejela A. Food security and environment conservation through sustainable use of wild and semi-wild edible plants: a case study in Berek Natural Forest, Oromia special zone, Ethiopia. Agric Food Secur. 2021;10:29.
Fassil A, Mazengia E, Gebreamanuel B, Dessie Y, Kumera B, Atnkut B, Mullualem D, Tsega A, Van Damme P. Harvesting nature’s bounty: exploring the ethnobotanical landscape of wild edible plants in the Awi Agäw community, Northwestern Ethiopia. J Ethnobiol Ethnomed. 2024;20:59.
Ajayi OC, Mafongoya PL. Indigenous knowledge systems and climate change management in Africa. CTA; 2017.
Duguma HT. Wild edible plant nutritional contribution and consumer perception in Ethiopia. Int J Food Sci. 2020;1–16.
Gebru M, Oduor F, Lochetti G, Kennedy G, Baye K. Ethiopia’s food treasures: revitalizing Ethiopia’s underutilized fruits and vegetables for inclusion in the food-based dietary guidelines for improved diet diversity, nutrition and health of the population. Rome: Bioversity International; 2019. p. 36.
Guzo S, Lulekal E, Nemomissa S. Ethnobotanical study of underutilized wild edible plants and threats to their long-term existence in Midakegn District, West Shewa Zone, Central Ethiopia. J Ethnobiol Ethnomed. 2023;19:30.
Tahir M, Abrahim A, Beyene T, Dinsa G, Guluma T, Alemneh Y, et al. The traditional use of wild edible plants in pastoral and agro-pastoral communities of Mieso District, eastern Ethiopia. Trop Med Health. 2023;51:10.
Asfaw A, Lulekal E, Bekele T, Debella A, Tessema S, Meresa A, et al. Ethnobotanical study of wild edible plants and implications for food security. Trees for People. 2023;14:100453.
Anbessa B, Lulekal E, Getachew P, Hymete A. Ethnobotanical study of wild edible plants in Dibatie district, Metekel zone, Benishangul Gumuz Regional State, western Ethiopia. J Ethnobiol Ethnomed. 2024;20:1.
Ghanimi R, Ouhammou A, Ahouach A, Cherkaoui M. Ethnobotanical study on wild edible plants traditionally used by Messiwa people, Morocco. J Ethnobiol Ethnomed. 2022;18:16.
Ashagre M, Asfaw Z, Kelbessa E. Ethnobotanical study of wild edible plants in Burji District, Segan Area Zone of Southern Nations, Nationalities and Peoples Region (SNNPR), Ethiopia. J Ethnobiol Ethnomed. 2016;12:32.
Dejene T, Agamy MS, Agúndez D, Martín-Pinto P. Ethnobotanical survey of wild edible fruit tree species in lowland areas of Ethiopia. Forests. 2020;11:177.
Giday M, Teklehaymanot T. Use of wild edible and nutraceutical plants in Raya-Azebo District of Tigray Region, northern Ethiopia. Trop Med Health. 2023;51:58.
Hankiso M, Warkineh B, Asfaw Z, Debella A. Ethnobotany of wild edible plants in Soro District of Hadiya Zone, southern Ethiopia. J Ethnobiol Ethnomed. 2023;19:21.
Melaku A, Ebrahim MA. Critical review on wild-edible fruit species in Ethiopia. Int J For Res. 2021;2021:1–2.
ARDNRCO (Arsi Robe District Natural Resources Conservation Office). Unpublished report of Arsi Robe District Natural Resources Conservation Office for the fiscal year 2021/2022. Robe, Ethiopia; 2022.
Central Statistical Authority (Csa). The 2023 Population Projection of Ethiopia, Central Statistical Authority (CSA), Addis Ababa, Ethiopia. http://www.statsethiopia.gov.et/population-projection/; 2023.
Martin GJ. Ethnobotany: a methods manual. 1st ed. Falmouth, Cornwall: Chapman & Hall; 1995.
Alexiades MN. Collecting ethnobotanical data: an introduction to basic concepts and techniques. Adv Econ Bot. 1996;10:53–94.
Hedberg I, Edwards S. Flora of Ethiopia and Eritrea. Volume 3: Pittosporaceae to Araliaceae. Addis Ababa: The National Herbarium; Department of Systematic Botany; 1989.
Hedberg I, Edwards S, Nemomissa S. Flora of Ethiopia and Eritrea. Volume 4: Apiaceae to Dipsacaceae. Addis Ababa: The National Herbarium, Department of Systematic Botany; 2003.
IPNI. The Royal Botanic Gardens, Kew, Harvard University Herbaria & Libraries and Australian National Herbarium; 2023. http://www.ipni.org. Accessed Aug 2023.
POWO. Facilitated by the Royal Botanic Gardens, Kew; 2023. http://www.plantsoftheworldonline.org/. Accessed Oct 2023.
Cotton CM. Ethnobotany: principles and applications. New York: Wiley; 1996.
Kent M, Coker P. Vegetation description and analysis: a practical approach. London: Belhaven Press; 1992.
Tebkew M, Gebremariam Y, Mucheye T, Alemu A, Abich A, Fikir D. Uses of wild edible plants in Quara district, northwest Ethiopia: implication for forest management. Agric Food Secur. 2018;7:1–14.
Yiblet Y, Adamu E. An ethnobotanical study of wild edible plants in Tach Gayint District, South Gondar Zone, Amhara Region, Northwestern Ethiopia. Evid Based Complement Altern Med. 2023;2023:1–11.
Aragaw HS, Nohr D, Callo-Concha D. Nutritional potential of underutilized edible plant species in coffee agroforestry systems of Yayu, southwestern Ethiopia. Agrofor Syst. 2021;95:1047–59.
Masresha G, Melkamu Y, Walle GC. Ethnobotanical study on wild edible plants in Metema District, Amhara Regional State, Ethiopia. Int J For Res. 2023;2023:1–10.
Lalmuanpuii R, Zodinpuii B, Bohia B, Zothanpuia, Lalbiaknunga J, Singh PK. Wild edible vegetables of ethnic communities of Mizoram (Northeast India): an ethnobotanical study in thrust of marketing potential. J Ethnobiol Ethnomed. 2024;20:58.
Siyum ZH, Meresa TA. Physicochemical properties and nutritional values of Carissa spinarum L./ “Agam” fruit. Int J Fruit Sci. 2021;21(1):826–34.
Tewolde-Berhan S, Remberg SF, Abegaz K, Narvhus J, Abay F, Wicklund T. Impact of drying methods on the nutrient profile of fruits of Cordia africana Lam. in Tigray, northern Ethiopia. Fruits. 2015;70(2):77–90.
Mokria M, Gebretsadik Y, Birhane E, McMullin S, Ngethe E, Hadgu KM, Hagazi N, Tewolde-Berhan S. Nutritional and ecoclimatic importance of indigenous and naturalized wild edible plant species in Ethiopia. Food Chem Mol Sci. 2022;4:100084.
Ngome PIT, Shackleton C, Degrande A, Tieguhong JC. Addressing constraints in promoting wild edible plants’ utilization in household nutrition: case of the Congo Basin forest area. Agric Food Secur. 2017;6:20.
Tieguhong JC, Ndoye O, Tchatat M, Chikamai B. Processing and marketing of non-wood forest products: potential impacts and challenges in Africa. Discov Innov. 2009;21(1):60–5.
Wondimu T, Asfaw Z, Kelbessa E. Ethnobotanical study of food plants around’Dheeraa’town, Arsi, Ethiopia. SINET Ethiop J Sci. 2006;29:71–80.
Tesfay A, Tewolde-Berhan S, Birhane E, Rannestad MM, Gebretsadik A, Hailemichael G, Haile M, Gebrekirstos A. Edible indigenous fruit trees and shrubs in Tigray, Ethiopia. Trees For People. 2024;16:100525.
FAO, Wfp, IFAD. The state of food insecurity in the world. Economic growth is necessary but not sufficient to accelerate reduction of hunger and malnutrition. Rome: FAO; 2012.
Teklehaymanot T. An ethnobotanical survey of medicinal and edible plants of Yalo Woreda in Afar regional state, Ethiopia. J Ethnobiol Ethnomed. 2017;13:40.
Regassa T, Kelbessa E, Asfaw Z. Ethnobotany of wild and semi-wild edible plants of Chelia District, West-Central Ethiopia. Sci Technol Arts Res J. 2015;3:122–34.
Addis G, Asfaw Z, Woldu Z. Ethnobotany of wild and semi-wild edible plants of Konso ethnic community, south Ethiopia. Ethnobot Res Appl. 2013;11:121–41.
Emire A, Demise S, Giri T, Tadele W. Ethnobotanical study of wild edible plants in Liben and Wadera Districts of Guji Zone, Southern Ethiopia. Glob J Agric Res. 2022;10:47–65.
Demise S, Asfaw Z. Ethno botanical study of wild edible plants in Adola District, Southern Ethiopia. Int J Res Anal Rev. 2020;7:212–28.
Balemie K, Kebebew F. Ethnobotanical study of wild edible plants in Derashe and Kucha Districts, South Ethiopia. J Ethnobiol Ethnomed. 2006;2:1–9.
Bahru T, Asfaw Z, Demissew S. Wild edible plants: sustainable use and management by indigenous communities in and the buffer area of Awash National Park, Ethiopia. SINET Ethiop J Sci. 2013;36:93–108.
Girmay G, Lulekal E, Belay B, Gebrehiwot K. Wild edible plants study in a dryland ecosystem of Ethiopia. Daagu Int J Basic Appl Res. 2022;4:105–19.
Beleta KG, Jiru DB, Tolera KD. Assessment of edible woody plants’ diversity, their threats, and local people’s perception in Borecha Woreda of Buno Bedele Zone, Southwestern Ethiopia. Int J For Res. 2024;1–7.
Tebkew M, Asfaw Z, Zewudie S. Underutilized wild edible plants in the Chilga District, northwestern Ethiopia: focus on wild woody plants. Agric Food Secur. 2014;3:1–16.
Feyssa DH, Njoka JT, Asfaw Z, Nyangito MM. Comparative analysis of indigenous knowledge on use and management of wild edible plants: the case of central East Shewa of Ethiopia. Ethnobot Res Appl. 2012;10:287–304.
Addis G, Asfaw Z, Woldu Z. The role of wild and semi-wild edible plants in household food sovereignty in Hamer and Konso Communities, south Ethiopia. Ethnobot Res Appl. 2013;11:251–71.
Ocho DL, Struik PC, Price LL, Kelbessa E, Kolo K. Assessing the levels of food shortage using the traffic light metaphor by analyzing the gathering and consumption of wild food plants, crop parts and crop residues in Konso, Ethiopia. J Ethnobiol Ethnomed. 2012;8:30.
Teklu Y. Ethnobotanical study on the diversity and utilization of wild edible plants in Majang Zone of Gambella Region, Southwest Ethiopia. Int J Innov Sci Res Technol. 2019;4:880–6.
Yimer A, Forsido SF, Addis G, Ayelign A. Nutritional composition of some wild edible plants consumed in Southwest Ethiopia. Heliyon. 2023;9:e16541.
Yiblet Y. Nutritional composition and antinutritional factors of five wild edible fruits grown in the Mekdela District, South of Wollo, Ethiopia. Sci World J. 2024;9.
Morka AB. Study on wild edible plant used by Shinasha ethnic community at Metekel Zone, Northwest Ethiopia. Adv Nutr Food Sci. 2024;9(1):01–11.
Hassen A. Diversity and potential contribution of wild edible plants to sustainable food security in North Wollo, Ethiopia. Biodiversitas J Biol Divers. 2021;22:2501–10.
Addis G, Urga K, Dikasso D. Ethnobotanical study of edible wild plants in some selected districts of Ethiopia. Hum Ecol. 2005;33:83–118.
Kassa Z, Asfaw Z, Demissew S. Ethno-ecological study of medicinal and wild edible plants in Sheka Zone, Southern Nations, Nationalities and Peoples Regional State, Ethiopia. Trop Plant Res. 2020;7(1):65–75.
Aryal KP, Poudel S, Chaudhary RP, Chettri N, Chaudhary P, Ning W, et al. Diversity and use of wild and non-cultivated edible plants in the Western Himalaya. J Ethnobiol Ethnomed. 2018;14(1):1–18.
Uprety Y, Poudel RC, Shrestha KK, Rajbhandary S, Tiwari NN, Shrestha UB, Asselin H. Diversity of use and local knowledge of wild edible plant resources in Nepal. J Ethnobiol Ethnomed. 2012;8(1):16.
Tugume P, Nyakoojo C. Traditional use of wild edible plants in the communities adjacent Mabira Central Forest Reserve, Uganda. Ethnobot Res Appl. 2020;20:1–14.
Cao Y, Li R, Zhou S, Song L, Quan R, Hu H. Ethnobotanical study on wild edible plants used by three trans-boundary ethnic groups in Jiangcheng County, Pu’er, Southwest China. J Ethnobiol Ethnomed. 2020;16:1–23.
Wang J, Seyler BC, Ticktin T, Zeng YG, Ayu K. An ethnobotanical survey of wild edible plants used by the Yi people of Liangshan Prefecture, Sichuan Province, China. J Ethnobiol Ethnomed. 2020;16(1):10.
Al-Fatimi M. Wild edible plants traditionally collected and used in southern Yemen. J Ethnobiol Ethnomed. 2021;17:1–21.
Cheng Z, Lu X, Lin F, Naeem A, Long C. Ethnobotanical study on wild edible plants used by Dulong people in northwestern Yunnan, China. J Ethnobiol Ethnomed. 2022;18:1.
Ghorbani A, Langenberger G, Sauerborn J. A comparison of the wild food plant use knowledge of ethnic minorities in Naban River Watershed National Nature Reserve, Yunnan SW China. J Ethnobiol Ethnomed. 2012;8(17):1–9.
Bhatia H, Sharma YP, Manhas RK, Kumar K. Traditionally used wild edible plants of district Udhampur, J&K, India. J Ethnobiol Ethnomed. 2018;14:1.
Mishra A, Swamy SL, Thakur TK, Bhat R, Bijalwan A, Kumar A. Use of wild edible plants: Can they meet the dietary and nutritional needs of indigenous communities in Central India. Foods. 2021;10(7):1453.
Nieman DC, Lee R. Nutritional assessment. 3rd ed. Boston: McGraw-Hill; 2003.
Sibiya NP, Kayitesi E, Moteetee AN. Proximate analyses and amino acid composition of selected wild indigenous fruits of Southern Africa. Plants. 2021;10(4):721.
Megersa M, Nedi T, Belachew S. Ethnobotanical study of medicinal plants used against human diseases in Zuway Dugda District, Ethiopia. Evid Based Complement Altern Med. 2023;2023:1–22.
Tamene S, Negash M, Makonda FB, Chiwona-Karltun L, Kibret KS. Ethnobotanical study on medicinal plant knowledge among three ethnic groups in peri-urban areas of south-central Ethiopia. J Ethnobiol Ethnomed. 2023;19(1):55.
Alemu M, Asfaw Z, Lulekal E, Warkineh B, Debella A, Sisay B, Debebe E. Ethnobotanical study of traditional medicinal plants used by the local people in Habru District, North Wollo Zone, Ethiopia. J Ethnobiol Ethnomed. 2024;20(1):4.
Zemede J, Mekuria T, Ochieng CO, Onjalalaina GE, Hu GW. Ethnobotanical study of traditional medicinal plants used by the local Gamo people in Boreda Abaya District, Gamo Zone, southern Ethiopia. J Ethnobiol Ethnomed. 2024;20(1):1–28.
Patel S. Food, pharmaceutical and industrial potential of Carissa genus: an overview. Rev Environ Sci Biotechnol. 2013;12:201–8.
Dhatwalia J, Kumari A, Verma R, Upadhyay N, Guleria I, Lal S, Thakur S, Gudeta K, Kumar V, Chao JCJ, Sharma S. Phytochemistry, pharmacology, and nutraceutical profile of Carissa species: an updated review. Molecules. 2021;26(22):7010.
Nazareth MS, Shreelakshmi SV, Shetty NP. Identification and characterization of polyphenols from Carissa spinarum fruit and evaluation of their antioxidant and anti-quorum sensing activity. Curr Microbiol. 2021;78(4):1277–85.
Mundaragi A, Thangadurai D. Process optimization, physicochemical characterization and antioxidant potential of novel wine from an underutilized fruit Carissa spinarum L. (Apocynaceae). Food Sci Technol. 2017;38:428–33.
Ojerinde OS, Gwatau DD, Falang KD, Odumosu PO, Kolawole JA. Nutritional composition, antioxidant assay and α-glucosidase inhibitory flavonoids from the fruits of Carissa edulis Vahl (Apocynaceae). J Pharma Bioresour. 2021;18(2):122–32.
Mamoona T, Rafique N, Zubair KM, Shafique AK, Bashir S, Ali ST, Salamatullah AM, Mekonnen AB, Bourhia M. Phytonutritional and sensorial assessment of a novel functional beverage formulated from an underutilized fruit of Carissa spinarum L. ACS Omega. 2023;8(36):32643–55.
Nuro GB, Tolossa K, Giday M. Medicinal plants used by Oromo Community in Kofale District, West-Arsi Zone, Oromia Regional State, Ethiopia. J Exp Pharmacol. 2024;16:81–109.
Alemayehu G, Asfaw Z, Kelbessa E. Cordia africana (Boraginaceae) in Ethiopia: a review of its taxonomy, distribution, ethnobotany and conservation status. Int J Bot Stud. 2016;1:38–46.
Tewolde-Berhan S, Remberg SF, Abegaz K, Narvhus JA, Abay F, Wicklund T. Ferric reducing antioxidant power and total phenols in Cordia africana. Afr J Biochem Res. 2013;7:215–24.
Negash L. A selection of African native trees: biology, uses, propagation and restoration techniques, Addis Ababa, Ethiopia; 2021. . ISBN 978-99944-3-086-4.
Obi KN, Onyeike EN, Anacletus FC. Studies on in vitro radical scavenging potentials of methanol leaf extract of Ficus sur and its fractions. Free Radic Antioxid. 2022;12(1):15–21.
Saloufou KI, Boyode P, Simalou O, Eloh K, Idoh K, Melila M, Toundou O, Kpegba K, Agbonon A. Chemical composition and antioxidant activities of different parts of Ficus sur. J Herbmed Pharmacol. 2018;7(3):185–92.
Ogunlaja OO, Moodley R, Baijnath H, Jonnalagadda SB. Antioxidant activity of the bioactive compounds from the edible fruits and leaves of Ficus sur Forssk. (Moraceae). S Afr J Sci. 2022;118(3–4):1–5.
Dongmo FL, Kamsu GT, Nanfack AR, Ndontsa BL, Farooq R, Bitchagno GT, Waha A, Tene. Chemical constituents and antibacterial activity from the fruits of Ficus sur Forssk. Investig Med Chem Pharmacol. 2022;5(1):64.
Ogunlaja OO, Moodley R, Baijnath H, Jonnalagadda SB. Nutritional evaluation, bioaccumulation and toxicological assessment of heavy metals in edible fruits of Ficus sur Forssk (Moraceae). J Environ Sci Health B. 2017;52(2):84–91.
Wilson AL, Downs CT. Fruit nutritional composition and nonnutritive traits of indigenous South African tree species. S Afr J Bot. 2012;78:30–6.
Badou RB, Yedomonhan H, Ewedje EEBK, Dassou GH, Adomou A, Tossou M, Akoegninou A. Floral morphology and pollination system of Syzygium guineense (Willd.) DC. subsp. macrocarpum (Engl.) F. White (Myrtaceae), a subspecies with high nectar production. S Afr J Bot. 2020;131:462–7.
Orwa C, Mutua A, Kindt R, Jamnadass R, Simons A. Agroforestree database: a tree reference and selection guide version 4.0. World Agroforestry Centre, Kenya; 2009. https://www.worldagroforestry.org/output/agroforestree-database. Accessed 25 Feb 2024.
Sibiya NP, Kayitesi E, Moteetee A. Mineral composition of selected indigenous wild southern African fruits. S Afr J Bot. 2020;132:87–94.
Maregesi S, Kagashe G, Messo CW, Mugaya L. Determination of mineral content, cytotoxicity and anthelmintic activity of Syzygium guineense fruits. Saudi J Med Pharm Sci. 2016;2(5):95–9.
Stadlmayr B, Charrondiere UR, Eisenwagen S, Jamnadass R, Kehlenbeck K. Nutrient composition of selected indigenous fruits from sub-Saharan Africa. J Sci Food Agric. 2013;93(11):2627–36.
Asfaw TB, Woldemariam HW, Tadesse MG, Tessema FB, Admassie ZG, Esho TB. Method optimization for the determinations of selected phytochemicals and antioxidant activities of wild Ethiopian Syzygium guineense fruit and seed under different drying conditions. Heliyon. 2023;9(6):e16227.
Mapunda EP, Mligo C. Nutritional content and antioxidant properties of edible indigenous wild fruits from miombo woodlands in Tanzania. Int J Biol Chem Sci. 2019;13(2):849–60.
Bekele T, Birnie AA, Tengnas B. Useful trees and shrubs for Ethiopia, vol. 2. Nairobi: Regional Soil Conservation Unit (RSCU), Swedish International Development Authority (SIDA); 1993. p. 123–7.
Molla M, Gemeda N, Abay SM. Investigating potential modes of actions of Mimusops kummel fruit extract and solvent fractions for their antidiarrheal activities in mice. Evid Based Complement Altern Med. 2017;2017:1–11.
Waweru DM, Arimi JM, Marete E, Jacquier JC, Harbourne N. Chemical and antioxidant characterization of Dovyalis caffra and Dovyalis abyssinica fruits in Kenya. Heliyon. 2022;8(10):e11064.
Jiru NA, Gemede HF, Keyata EO. Nutritional composition and antioxidant properties of selected underutilized wild edible fruits in East Wollega zone, Western Ethiopia. Int J Fruit Sci. 2023;23(1):34–45.
Acknowledgements
Our special thanks go to informants from the Arsi Robe district for their participation in providing relevant responses to the questions. Without their participation, the study would not have been carried out. We thank Madda Walabu University for funding the study. We also thank the leaders of the Arsi Robe district for their cooperation in providing us with a support letter to visit the study sites.
Funding
Madda Walabu University supported the study.
Author information
Authors and Affiliations
Contributions
SB designed the study, collected the data, and analyzed the results. BA analyzed the results and critically reviewed the manuscript. GD, AS, FG, BW, and GA analyzed and interpreted the data. ZK prepared the climadiagram, checked the appropriateness of the semi-structured interviews, and critically reviewed the manuscript. MM conceived the research idea, supervised the work, and wrote the manuscript. All the authors have read and approved the final manuscript and agreed on its submission for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Oral and written permission was obtained from the study district administration and the administrative offices of Kebele. A supporting letter was obtained from the Department of Biology of Madda Walabu University to collect data from the study district.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Biri, S., Ayenew, B., Dida, G. et al. Ethnobotanical study of wild edible plants in Arsi Robe district of East Arsi Zone, Ethiopia. J Ethnobiology Ethnomedicine 20, 70 (2024). https://doi.org/10.1186/s13002-024-00703-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13002-024-00703-x