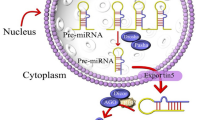
Abstract
MicroRNAs, as non-coding transcripts, modulate gene expression through RNA silencing under normal physiological conditions. Their aberrant expression has strongly associated with tumorigenesis and cancer development. MiR-20b is one of the crucial miRNAs that regulate essential biological processes such as cell proliferation, apoptosis, autophagy, and migration. Deregulated levels of miR-20b contribute to the early- and advanced stages of cancer. On the other hand, investigations emphasize the tumor suppressor ability of miR-20b. High-throughput strategies are developed to identify miR-20b potential targets, providing the proper insight into its molecular mechanism of action. Moreover, accumulated results suggest that miR-20b exerts its effects through diverse signaling pathways, including PI3K/AKT/mTOR and ERK axes. Restoration of the altered expression levels of miR-20b induces cell apoptosis and reduces invasion and migration. Further, miR-20b can be used as a biomarker in cancer. The current comprehensive review could lead to a better understanding of the miR-20b in either tumorigenesis or tumor regression that may open new avenues for cancer treatment.
Video Abstract
Similar content being viewed by others
Introduction
Investigations in the recent two decades changed the researchers' point of view from mRNA to non-coding RNAs as a pivotal regulator of the human genome [1]. The term "non-coding" in biology generally refers to RNA that does not translate to protein. In mammalian genomes, approximately 97% of the genome is transcribed as non-coding RNA, including transfer RNA (tRNA), ribosomal RNA (rRNA), circular RNA, transposons, and microRNAs [2, 3].
MicroRNAs (miRNAs) represent short non-coding RNAs binding to the 3′ untranslated region (UTR) of specific mRNA and suppressing its expression [4]. On the other hand, recent investigations have implied that miRNAs can interact with 5′ UTR of mRNA, promoter of genes, and protein to exert their effects [5]. Indeed, they profoundly impact the transcription and translation of genes through either inhibition or induction. MiRNAs subsequently modulate a wide range of cellular functions [6]. Interaction between miRNAs and other non-coding RNAs like circular RNA to control cellular function make miRNAs essential cell regulators [7]. Furthermore, the comprehensive analysis estimated that one-third of the human genome is modulated through miRNAs [8]. It seems reasonable that impaired miRNA expression may underly human diseases such as cancer.
Cancer is a complex disease comprising uncontrolled growth and division of cells to form tumors in part of the body [9]. Tumor cells can spread to other organs and create a secondary tumor called metastasis [10]. Multiple cell effectors, such as miRNAs, are associated with tumorigenesis [11]. A broad spectrum of reports has evinced that the aberrant expression of miRNAs may cause cancer. Indeed, miRNA levels vary in tumor cells because of diverse mechanisms, including disruption in miRNA biogenesis, deletion and amplification of related genes, dysregulation in transcriptional control mechanisms, and abnormal epigenetic changes [12]. In the next step, miRNA-mediated gene modulation and underlying signaling pathways are disrupted [13]. Then these effects are traceable at the cellular levels, where cell functions may be impaired, and symptoms of the diseases appear. The miRNAs are classified into tumor suppressors and oncogenes according to their regulatory role in target genes [14]. Loss of tumor suppressor miRNAs facilities tumor formation and progression by up-regulating oncogenes expression and activation. In contrast, enhanced levels of oncogenic miRNAs down-regulate tumor suppressor genes and result in malignancy.
IsomiRs refer to non-canonical variants of the reference miRNA that can differ in sequence, length, or both [15]. The miRNAs like miR-20a and miR-20b with one or two nucleotide differences are denoted with a lowercase letter. Besides, based on the origin of sequence from opposite arms of pre-miRNA, mature miRNA is annotated with a -5p or -3p, such as miR-20b-5p [16]. The most important of these findings is that the functions and biological activity of isomiRs are diverse from each other and canonical miRNA [17].
MiR-20a is a member of the miR-17-92 cluster and is located on chromosome 13q31.3, while miR-20b belongs to the miR-106b-25 cluster and is found on chromosome Xq26.2 [18]. Based on different gene regulatory elements, the expression pattern of these two clusters varies in a cell- and tissue-dependent manner. On the other hand, both of these clusters are called the miR-17 family [19]. Members of this family appear to be the result of gene duplication occurrences. The seed sequence (AAAGUG) of members, despite minor differences in length and nucleotide composition, is the same which suggests functional redundancy [20]. As a result, miR-20a and miR-20b have different targets and regulate a variety of downstream biological processes. Plenty of studies have reviewed the miR-20a role in a variety of malignancies, including thyroid cancer, gastric cancer, gliomas, cervical cancer, colon adenocarcinoma, and prostate cancer [21].
The miR-20b is a well-studied regulator in healthy and pathological human conditions (Table 1). Cumulative evidence has illustrated that the miR-20b is involved in normal development, aging, immune diseases, neurodegenerative diseases, and cardiovascular diseases via cell cycle regulation accompanied by moderating cell proliferation and apoptosis [22,23,24]. Besides, animal modeling has been performed to study miR-20b function in healthy human physiology and related disorders (Table 2). Due to the extensive scope of this topic, we focused on miR-20b potential role in tumorigenesis and cancer development.
In contrast to its tumor-supportive role, miR-20b could elicit anti-tumor responses, thus decreasing tumor progression. Based on miR-20b levels and its various targets in a wide range of signaling pathways, major cellular processes such as proliferation, cell cycle, apoptosis, autophagy, and migration are affected. As a result, its role can be different. Aberrant expression of miRNAs has been noted as a molecular tool for the diagnosis and prognosis of cancer. Indeed, surveillance of tumor markers may have advantages for patients before the onset of symptoms, during, and after treatment. Several studies have revealed that miR20b is found in biological fluid and tissue specimens, which have potential applications as biomarkers. These findings are essential to clarifying the therapeutic and diagnostic role of miR-20b in cancer.
Approaches to determine the targets of miR-20b
Identifying potential candidate genes targeted through miRNAs is essential to discovering the function of microRNAs in gene regulatory networks. As a consequence, miRNA-related cellular processes can be determined. It can be considered a pivotal procedure to illuminate the pathogenesis of cancer. In the following, different approaches to predicting targets of miR-20b are introduced.
The miR-20b in normal physiological condition
Growing evidence has revealed the fundamental roles of miRNAs in normal physiological conditions [53]. In this regard, miRNAs act toward maintaining cell function and homeostasis through cooperation with other cellular components and plenty of effectors. It has been authenticated that miR-20b is involved in the regulatory genes network within human cells. In Table 1, we summarized the findings from related investigations around the regulatory role of miR-20b in human biological specimens and cell lines. Evidence has indicated that fundamental cellular processes such as apoptosis, proliferation, differentiation, and T-cell activation are regulated upon miR-20. On the other hand, certain levels of miR-20b in cells are associated with their required function. As a result, changes in miR-20b levels lead to dysregulation of vital biological functions, which triggers human diseases.
Evolutionarily conserved miR-20b gene and comparative biology
Comparative biology is an approach that examines the differences and similarities between organisms, especially in biology, biochemistry, genetics, and physiology [54]. Conserved intra- and inter-species genes and proteins are of interest in comparative biology as a conserved gene in one organism may have a similar function in another [55]. MiRNAs are mentioned as the conserved genes during evolution [56]. Therefore, studies on animal models provide valuable information about the gene-miRNA network, leading to a further understanding of specific miRNA functions in human physiology. According to Table 2, miR-20b can modulate biological function in different animal models of diseases. Considering affected potential targets of miR-20b in the animal models might give a clue to its mechanism of action in humans.
High-throughput analysis to predict miR-20b potential target
Nowadays, novel approaches are established to determine the miRNA-mRNA interaction and underlying mechanisms of action. A computational method is a mathematical approach to merging biology, chemistry, and computer science [57]. This high-throughput analysis can accurately describe the miRNA-related target genes in various diseases. Using computational methods, Kim et al. determined that miR-20b targets transcription factor E2F1 in breast cancer [58]. E2F1 is a transcription factor that facilitates the synthesis of DNA and cell cycle in mammalian cells [59]. Consequently, targeting E2F1 by miR-20b inhibits tumor cell division. Integrative computational algorithms can analyze dysregulated miRNA targets in autoimmune diseases to identify strong candidate genes. Significantly, miR-20b has been determined as a modulator of helper T-cells differentiation with a high score [60]. Negative and positive regulators of T-cells differentiation, such as FOXO1 (Forkhead Box O1) and STAT3 (Signal transducer and activator of transcription 3), have been defined as significant miR-20b downstream target genes.
The miR-20b and cancer
Aberrant expression patterns of miRNAs influence the critical properties of tumors, such as escaping growth inhibitors, uncontrolled growth, promoting migration and invasion, preserving proliferative signaling pathways, suppressing apoptosis, and activating angiogenesis [12]. In contrast to the tumor-promoting role of miRNAs, these non-coding RNAs can act as tumor suppressors and inhibit tumor formation and progression under certain conditions. The miRNAs exert their effects through target genes and related signaling pathways. As mentioned, miR-20b binds to various targets and regulates signaling pathways and biological processes. Over the past years, much more information has become available on the occurrence of cancer upon disruption in miR-20b function and levels (Table 3). We reviewed the role of miR-20b in different solid tumors with emphasis on either its tumor-supportive (Fig. 1) or suppressor (Fig. 2) activities.
Proliferation and cell cycle
Cell division is strongly associated with tumorigenesis and counted as an essential hallmark of malignancy. At the molecular levels, extra- and intracellular signaling pathways, growth factors, and bona fide of hormones keep an equilibrium between the induction of cell division and its inhibition [12]. Abnormal activity of these regulatory pathways leads to cancerous cells with out-of-control proliferation. Various studies have assessed the efficacy of miR-20b in cell division and cell cycle. It significantly integrates into several vital cell proliferation pathways, and the improper modulation of this miRNA is responsible for preserving proliferative signaling and escaping growth inhibitors in tumor cells.
The miR-20b is overexpressed in MCF-7 and MDA-MB-231 cell lines compared to healthy cells [85]. It may refer to the potential role of miR-20b in breast cancer tumorigenesis. Olga et al. have shown that radiation increases the levels of miR-20b, which targets the Phosphatase and tensin homolog (PTEN) and Breast cancer type 1 (BRCA1) genes, and promotes the proliferation and cell cycle of breast cancer cells [86]. BRCA1 is involved in homologous recombination, a kind of DNA repair mechanism. Impairment in BRCA1 normal activity increases genomic instability and facilitates cancer cell proliferation and tumor progression. Besides, incompetent DNA repair in cancer cells yields increased proliferation. Indeed, miR-20b triggers genomic instability and elevated cell proliferation by targeting BRCA1. The growing evidence suggests that miR-20b broadly targets PTEN in multiple cancer cells. Up-regulated miR-20b negatively correlates with PTEN expression in breast cancer specimens [87]. PTEN serves as a tumor suppressor, tightly deterring transformed cell growth and division [88]. Besides, aberrant expression of miR-20b is closely related to prostate cancer [89]. MiR-20b is elevated in prostate cancer cells and triggers their proliferation. Bioinformatics analysis showed that the PTEN gene is a potential target of miR-20b, as verified by preclinical experiments [67]. Furthermore, it has been indicated that miR-20b leads to hepatocellular and colorectal cancer cell proliferation through binding PTEN and down-regulates its expression [74, 79]. Using the antagomir of miR-20b enhances PTEN levels and causes tumor regression. Antagomirs are synthetic antisense oligonucleotides that bind to the desired miRNA and reduce or block its activity [90].
Overexpression of miR-20b positively correlates with poor prognosis in lung cancer [91]. To understand the working mechanism of miR-20b in lung cancer proliferation, Xuan et al. have demonstrated that miR-20b is up-regulated in non-small cell lung cancer cells [70]. Further, molecular experiments revealed that B-cell translocation gene 3 (BTG3) reduced upon miR-20b overexpression and led to tumor cell proliferation [70]. BTG3 is an anti-proliferative protein that loses its normal function during tumorigenesis in human cancers [45]. The level of miR-20b and its function integrate into proliferation-related signaling pathways. Ectopic low-level of miR-20b is associated with tumorigenesis and progression of thyroid carcinoma [77]. It directly binds to extracellular signal-regulated kinase 2 (ERK2) and son of sevenless homolog 1 (SOS1) [77]. These proteins are member of the mitogen-activated protein kinase (MAPK) signaling pathway, which promote cellular proliferation and differentiation [92].
Despite the oncogenesis effect of miR-20b in promoting cell proliferation, miR-20b has an anti-proliferative ability in some cases. The cell cycle-regulated genes such as cyclin-dependent kinase (CDKs) and cyclin have been recognized as miR-20b target genes. In ovarian cancer, overexpression of miR-20b arrests the cell cycle in the G1 phase by reducing cyclin D1 levels [66]. The transfection of EJ cells, an invasive bladder carcinoma, with miR-20b triggers G1 phase arrest by directly targeting CDK6, CDK2, and cyclin D [76]. Furthermore, p21, a well-known inhibitor of CDKs, was indirectly increased in miR-20b-transfected cells and thus reinforced cell cycle arrest.
Metastasis
Cancerous cells acquire several properties and transform into metastatic cells, which can migrate from the original location to distant sites and form secondary tumors [93]. This advanced cancer stage is responsible for patients' illness severity and death. Compelling evidence has indicated that various steps, such as epithelial-mesenchymal transition (EMT), migration, invasion, and angiogenesis, are involved in metastasis.
EMT
Reversible and rapid modulation of phenotype transition from epithelium to mesenchyme is termed EMT. During this peculiar process, primary tumor cells lose their surface adhesion molecules and obtain migration capabilities [94]. Many experiments have recognized the different signaling pathways, including transforming growth factor beta (TGF-β) participating in EMT [95]. In prostate cancer, the TGF-β signaling pathway significantly promotes EMT, while miR-20b mimic represses EMT by targeting the receptor of TGF- β [68]. The microRNA mimic approach has focused on using synthetic miRNA-like fragments for gene silencing [96]. Furthermore, incorporating long-noncoding RNA with miRNAs to regulate biological processes has become a field of interest for investigators. LncRNA operates as a miRNA sponge that binds to the desired miRNA with high affinity and inhibits their downstream functions [97]. H19, a long non-coding RNA, interacts with miR-20b and suppresses its biological function [84]. Mechanistically, miR-20b downregulates HIF-1 alpha, while downregulation of miR-20b activates hypoxia signaling pathways and stimulates EMT in endometrial cancer [84].
Migration and invasion
Cell migration typically occurs in embryonic development, nervous system formation, wound healing, vascular sprouting, and immune-cell trafficking [61]. The migration enables cells to change their location in either tissue or among various organs. Uncontrolled cell movement is related to pathological circumstances, including the invasion behavior of tumor cells. In invasion, malignant cells can penetrate tissue and vascular barriers into the bloodstream. Characterizing cancer cells' migration and invasive potential and interaction with various underlying regulation mechanisms, such as miRNA, is relevant for developing therapeutic strategies against cancer.
The results of the microarray assay emphasize miR-20b key role in the miRNA-mRNA network in cervical cancer migration [69]. Cervical cancer is a prevalent malignancy in women after breast and intestine cancer [98]. Papillomavirus (HPV) infection may trigger tumorigenesis and cervical cancer progression. This virus increases miR-20b levels and leads to migration and invasion via targeting tissue inhibitor of metalloproteinases 2 (TIMP2, an inhibitor of MMP2 [69]. Metalloproteases (MMPs) such as MMP2 are complicated in the degradation of the extracellular matrix, thus enabling cell migration [99]. There is another relevance between changed miR-20b levels and MMPs in bladder cancer occurrence and progression. It has been indicated that miR-20b expression is reduced in bladder tumor cells, and elevated miRNA levels can act as a tumor suppressor [76]. MMP2 is described as the potential target of miR-20b [76]. Downregulation of miR-20b predominantly yields sustained migration and invasion of bladder tumor cells.
The miR-20b also participates in tumorigenesis and esophageal cancer progression. Indeed, miR-20b activity brings about esophageal cancer migration and invasion through the downregulation of PTEN levels [71]. The regular role of PTEN is suppressing migration through its phosphatase activity [100]. PTEN inactivates the phosphoinositide 3-kinase (PI3K) signaling pathway, which is vital to provide a front-rear gradient of molecular and chemotaxis levels for cell migration. Consequently, targeting PTEN augments PI3K signaling pathway and cancer cell migration.
Angiogenesis
Angiogenesis is the formation of blood vessels to supply cancer cells within the tumor. As a result of its unique capacity to inspire new blood vessel formation, angiogenesis plays an essential role in tumor formation and metastasis [101]. The miRNAs regulate several angiogenesis-related signaling pathways and master genes [102]. Vascular endothelial growth factor (VEGF) is secreted by various cells, such as cancer cells, to stimulate angiogenesis. Targeting VEGF either directly or indirectly through miR-20b leads to down-regulation of angiogenesis. However, the interaction between long non-coding RNAs and miR-20b leads to the induction of VEGF expression in some cases. Enhanced levels of lncCAMTA1, a long non-coding RNA, are found mainly in the MDA-MB-231 breast cancer cell line. Further analysis indicated that lncCAMTA1 binds to miR-20b, thus up-regulating VEGF level as the miR-20b target gene [62]. In addition, VEGF downstream signaling pathways such as MAPK, ERK, Janus kinase (JAK), and Signal transducer and activator of transcription (STAT) are up-regulated in MDA-MB-231 cells, and angiogenesis occurs. Interestingly, miR-20b can target the upstream regulator of the VEGF gene and trigger angiogenesis in hepatocellular carcinoma [81]. STAT3, as the miR-20b target, negatively regulates VEGF in mRNA and protein levels. Overexpression of miR-20b in hepatocellular carcinoma cells inhibits STAT3 function, leading to increased VEGF levels [81].
Cancer stem cells
Recent developments in miRNA-mRNA interaction networks have led to an interest in miR-20b in cancer stem cells (CSCs), believed to play a critical role in tumor progression and drug resistance [103]. The miR-20b may be a double-edged sword in cancer stem cell regulation.
The stemness of cancer stem cells that causes the malignancy of colorectal tumor have been explored [73]. The expression of Oct4, a stem cell marker, and MALAT1, a long non-coding RNA, are negatively related by miR-20b levels. Overexpression of miR-20b attenuates the proportion of cancer stem cells via the direct targeting of Oct4 and MALAT1, critical positive regulators of cancer stem cell stemness [73, 81, 102, 103], and finally eases tumor regression [104]. However, miR-20b could promote breast cancer stem cell proliferation and cell cycle [64]. It indirectly elevates Cyclin D1 and E2F1 mRNA levels and promotes cell proliferation in breast cancer stem cells [64]. Cyclin D1 is a crucial regulator of cell cycle progression from G1 to the S phase [104]. Overexpression of cyclin D1 and E2F1 mediates the high rate of cell cycle and division [105]. Further, an examination has revealed that miR-20b may inhibit cyclin D1 and E2F1 inhibitors, ultimately increasing cyclin D1 and E2F1 expression.
Autophagy
Autophagy is a conserved self-degradative cellular procedure to remove damaged organelles, intracellular pathogens, and misfolded proteins [106]. This fundamental process balances cell energy and response to nutrient stress [106]. The relevance of autophagy in early- and advanced cancer remains disputable. While investigations have illustrated that autophagy inhibits tumorigenesis by suppressing tumor cell growth and inducing apoptosis, it may also cause tumor cell migration and invasion [107, 108]. Depending on a wide range of regulators, including miRNAs, the role of autophagy can be different in cancer. It has been shown that miR-20b can downregulate autophagy via inhibiting RB1CC1/FIP200, autophagosome formation-related proteins [63]. Then breast cancer cells' survival decreased. Further research should be carried out to establish closer links between miR-20b and autophagy in cancer.
Apoptosis
Apoptosis is called programmed cell death (PCD), mainly containing intrinsic and extrinsic pathways. Each pathway requires a specific initiator caspase protein which activates the downstream molecular cascade and induces apoptosis [109]. Improper apoptosis, either activation or suppression, is a key factor in human diseases like cancer [110]. A strong relationship between miR-20b and apoptosis has been reported in ovarian cancer. Increased miR-20b levels remarkably down-regulate B-cell lymphoma 2 (Bcl2) as an anti-apoptotic protein [65]. As a result, Bax protein is oligomerized on the mitochondria surface and causes induces apoptosis by releasing cytochrome c [65]. Furthermore, Increased miR-20b levels induce apoptosis in renal cell carcinoma. MAPK signaling pathway components are considered miR-20 putative targets based on the bioinformatic assay [82]. Depending on cell type, the MAPK cascade has a dual role in inducing or inhibiting apoptosis [111]. Although the MAPK signaling pathway is up-regulated in renal cancer cells and inhibits apoptosis [112], over-expression of miR-20b could inhibit this axis and induce apoptosis.
The miR-20b can inhibit apoptosis. The expression of miR-20b and its underlying mechanism of action has been investigated in cancerous thyroid cells. LncRNA- miRNA crosstalk can regulate apoptosis by affecting associated signaling pathways. double homeobox A pseudogene 8 (DUXAP8), a lncRNA, suppresses apoptosis in papillary thyroid tumor cells. Indeed, DUXAP8 binds to miR-20b and inhibits its function [78]. SOS-1 of the ERK signaling pathway, which causes cell survival, is known as the miR-20b target. Mechanistically, miR-20b inhibition by DUXAP8 improves cell proliferation while impeding apoptosis [78].
Drug resistance
Drug resistance is the main challenge in cancer treatment, primarily preventing chemotherapy's efficacy in cancer cells and promoting cancer cell survival. MiR-20b has an essential role in chemotherapy resistance. It has been demonstrated that the activation of Syndecan-2, a transmembrane receptor, is positively correlated with chemotherapy resistance [75]. Mechanistically, miR-20b diminishes the resistance of cancer cells to chemotherapy by targeting Syndecan-2 [75]. Its capability to regulate the sensitivity of cancer cells to chemotherapy drugs through hypoxia signaling pathways has profoundly been displayed.
Interestingly, the downregulation of miR-20b in gastric cancer induces drug resistance [83]. Hypoxia-inducible factor 1 subunit alpha (HIF1A) negatively correlates with miR-20b levels in gastric cancer. As a result, increased levels of HIF1A activate downstream genes like multidrug resistance (MDR) and facilitate drug resistance [83].
An investigation of hepatocellular carcinoma cells has indicated that drug resistance negatively associates with reduced levels of miR-20b [80]. Overexpression of miR-20b leads to the sensitivity of hepatocellular cancer cells to chemotherapy drugs. From the molecular aspect, the cell division cycle 37-like 1 (CDC37L1) gene enhances drug resistance of hepatocellular cancer cells [80]. Targeting CDC37L1 by miR-20b reverses its effects. CDC37L1 is a cochaperone protein that promotes cell survival [113]. Besides, miR-20b can induce apoptosis of drug-resistance colon cancer cells by suppressing ADAM9 (A disintegrin and a metalloprotease 9), an activator of MMPs [72]. Silencing of ADAM9 expression encourages apoptosis in various tumor cells, such as ovarian and prostate cancer [114, 115].
The miR-20b as a biomarker
Biomarkers are an increasingly important area in biomedical science [116]. These measurable indicators are biological molecules that can be found in body fluids, including blood, plasma, urea, and semen, as well as related tissue biopsy. Biomarkers provide essential information on either normal or abnormal cellular function [116]. Due to identifying miRNAs in biological fluids, miRNAs could be applied as potential biomarkers for a wide range of abnormalities such as cancer [117]. Using miRNAs as a biomarker can evolve the prognosis of malignant cancers and make it possible to evaluate tumors and the treatment protocols' efficacy.
A study on renal cell carcinoma determined that miR-20b was downregulated during tumorigenesis. Consequently, tumor cell proliferation and cell cycle were increased [82]. Therefore, miR-20b may be a biomarker for prognosis prediction and early detection of renal cell carcinoma. Besides, an elevated level of miR-20b found in breast cancer patients' serum and plasma has a positive correlation with tumor malignancy [118]. Consequently, it has the potential to be considered a biomarker with significant sensitivity and specificity. Evaluation of miR-20b level in plasma samples of endometrial cancer patients showed that this miRNA has prognostic value in advanced tumors compared to the early stage [119]. Up-regulated miR-20b in HPV- associated oropharyngeal carcinoma specimens is negatively correlated with patient survival [120], and elevated miR-20b level is associated with the shorter survival rate of breast cancer patients [121].
A recent study in breast cancer patients exhibited down-regulated levels of miR-20b in procured exosomes from tumor biopsy samples with recurrence [122]. Interestingly, exosome-packaged miRNAs are secreted into the cell microenvironment to regulate target cells and underlying signaling pathways [123].
Conclusion
The abbrent expression of miR-20b affects tumor-related signaling pathways, underlying genes, and biological functions. Despite acting as an oncogene and tumor progression factor, miR-20b has tumor suppressor function in cancers. This dual role of miR-20 may depend on target genes in specific cellular conditions. Furthermore, integrating miR-20b function with long non-coding RNAs such as DUXAP8 in regulating fundamental cellular processes leads to inconsistent effects. Different vital tumor-related genes, such as PTEN, VEGF, MMP2, Oct4, and ADAM9, were defined as miR-20b targets. Many signaling pathways, including ERK, STAT, and TGF-beta, were regulated through miR-20b in tumor cells. Multiple comprehensive analyses revealed that the miR-20b could consider as a biomarker to evaluate tumor progression and the effectiveness of therapeutic approaches. This review provides a better understanding of miR-20b function during tumorigenesis and cancer progression. More information on miR-20b would help to establish a greater degree of accuracy on this matter. Therefore, further investigations are required to investigate the interaction between miR-20b and cancer.
Availability of data and materials
Not applicable.
References
Hombach S, Kretz M. Non-coding RNAs: classification, biology and functioning. Adv Exp Med Biol. 2016;937:3–17.
Sevignani C, Calin GA, Siracusa LD, Croce CM. Mammalian microRNAs: a small world for fine-tuning gene expression. Mamm Genome Off J Int Mamm Genome Soc. 2006;17(3):189–202.
Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, et al. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature. 2005;433(7027):769–73.
Saliminejad K, Khorram Khorshid HR, Soleymani Fard S, Ghaffari SH. An overview of microRNAs: biology, functions, therapeutics, and analysis methods. J Cell Physiol. 2019;234(5):5451–65.
Ørom UA, Nielsen FC, Lund AH. MicroRNA-10a binds the 5′UTR of ribosomal protein mRNAs and enhances their translation. Mol Cell. 2008;30(4):460–71.
Liu B, Li J, Cairns MJ. Identifying miRNAs, targets and functions. Brief Bioinform. 2014;15(1):1–19.
Ratti M, Lampis A, Ghidini M, Salati M, Mirchev MB, Valeri N, et al. MicroRNAs (miRNAs) and long non-coding RNAs (lncRNAs) as new tools for cancer therapy: first steps from bench to bedside. Target Oncol. 2020;15(3):261–78.
Urbich C, Kuehbacher A, Dimmeler S. Role of microRNAs in vascular diseases, inflammation, and angiogenesis. Cardiovasc Res. 2008;79(4):581–8.
Roy PS, Saikia BJ. Cancer and cure: a critical analysis. Indian J Cancer. 2016;53(3):441–2.
Suhail Y, Cain MP, Vanaja K, Kurywchak PA, Levchenko A, Kalluri R, et al. Systems biology of cancer metastasis. Cell Syst. 2019;9(2):109–27.
Hill M, Tran N. miRNA interplay: mechanisms and consequences in cancer. Dis Models Mech. 2021;14(4):dmm047662.
Peng Y, Croce CM. The role of MicroRNAs in human cancer. Signal Transduct Target Ther. 2016;1(1):15004.
Acunzo M, Romano G, Wernicke D, Croce CM. MicroRNA and cancer–a brief overview. Adv Biol Regul. 2015;57:1–9.
Otmani K, Lewalle P. Tumor suppressor miRNA in cancer cells and the tumor microenvironment: mechanism of deregulation and clinical implications. Front Oncol. 2021;11:708765.
Magee RG, Telonis AG, Loher P, Londin E, Rigoutsos I. Profiles of miRNA isoforms and tRNA fragments in prostate cancer. Sci Rep. 2018;8(1):5314.
Tomasello L, Distefano R, Nigita G, Croce CM. The MicroRNA family gets wider: the IsomiRs classification and role. Front Cell Dev Biol. 2021;9: 668648.
Zelli V, Compagnoni C, Capelli R, Corrente A, Cornice J, Vecchiotti D, et al. Emerging role of isomiRs in cancer: state of the art and recent advances. Genes. 2021;12(9):1447.
Nunes DN, Dias-Neto E, Cardó-Vila M, Edwards JK, Dobroff AS, Giordano RJ, et al. Synchronous down-modulation of miR-17 family members is an early causative event in the retinal angiogenic switch. Proc Natl Acad Sci USA. 2015;112(12):3770–5.
Tanzer A, Stadler PF. Molecular evolution of a microRNA cluster. J Mol Biol. 2004;339(2):327–35.
Ventura A, Young AG, Winslow MM, Lintault L, Meissner A, Erkeland SJ, et al. Targeted deletion reveals essential and overlapping functions of the miR-17 through 92 family of miRNA clusters. Cell. 2008;132(5):875–86.
Huang W, Wu X, Xiang S, Qiao M, Cen X, Pan X, et al. Regulatory mechanism of miR-20a-5p expression in Cancer. Cell Death Discovery. 2022;8(1):262.
Basak J, Majsterek I. miRNA-Dependent CD4(+) T cell differentiation in the pathogenesis of multiple sclerosis. Multiple Sclerosis International. 2021;2021.
Zhou ZQ, Chen SW, Tian ZM, Deng SB, Yi XY, Yang SN, et al. miR-20b-5p attenuates hypoxia-induced apoptosis in cardiomyocytes via the HIF-1 alpha/NF-kappa B pathway. Acta Biochim Biophys Sin. 2020;52(9):927–34.
Wang RZ, Chopra N, Nho K, Maloney B, Obukhov AG, Nelson PT, et al. Human microRNA (miR-20b-5p) modulates Alzheimer’s disease pathways and neuronal function, and a specific polymorphism close to the MIR20B gene influences Alzheimer’s biomarkers. Mol Psychiatry. 2022;27(2):1256–73.
Xu MZ, Yu T. MiR-20b-5p contributes to the dysfunction of vascular smooth muscle cells by targeting MAGI3 in hypertension. J Mol Histol. 2022;53(2):187–97.
Lu Y, Wang SJ, Cai SY, Gu XX, Wang JJ, Yang Y, et al. Propofol-induced MiR-20b expression initiates endogenous cellular signal changes mitigating hypoxia/re-oxygenation-induced endothelial autophagy in vitro. Cell Death Dis. 2020;11(8):1–4.
Zhang SS, Kan XQ, Liu P, Yin LZ, Li QY, Xu HY. MiR-20b is implicated in preeclampsia progression via the regulation of myeloid cell leukemin-1. J Biol Regul Homeost Agents. 2020;34(5):1709–17.
Tang J, Luo LY. MicroRNA-20b-5p inhibits platelet-derived growth factor-induced proliferation of human fetal airway smooth muscle cells by targeting signal transducer and activator of transcription 3. Biomed Pharmacother. 2018;102:34–40.
Jin MY, Li HY, Xu HF, Huo GX, Yao YY. MicroRNA-20b inhibits trophoblast cell migration and invasion by targeting MMP-2. Int J Clin Exp Pathol. 2017;10(11):10901–9.
Xin YZ, Cai HF, Lu TY, Zhang Y, Yang Y, Cui YB. miR-20b Inhibits T Cell proliferation and activation via NFAT signaling pathway in thymoma-associated myasthenia gravis. Biomed Res Int. 2016;2016.
Wang HY, Ban WR, Wang T, Li Z, Dang XQ. miR-20b/106a modulate Ngn2 gene expression during neural differentiation of human umbilical cord mesenchymal stem cells. NeuroReport. 2017;28(18):1225–31.
Mu GH, Deng YJ, Lu ZQ, Li X, Chen YB. miR-20b suppresses mitochondrial dysfunction-mediated apoptosis to alleviate hyperoxia-induced acute lung injury by directly targeting MFN1 and MFN2. Acta Biochim Biophys Sin. 2021;53(2):220–8.
Zhu SH, Hu XS, Yu ZB, Peng YZ, Zhu JG, Liu XH, et al. Effect of miR-20b on apoptosis, differentiation, the bmp signaling pathway and mitochondrial function in the P19 cell model of cardiac differentiation in vitro. PLoS ONE. 2015;10(4):e0123519.
He WD, Cheng Y. Inhibition of miR-20 promotes proliferation and autophagy in articular chondrocytes by PI3K/AKT/mTOR signaling pathway. Biomed Pharmacother. 2018;97:607–15.
Cui Y, Han J, Xiao ZF, Chen T, Wang B, Chen B, et al. The miR-20-Rest-Wnt signaling axis regulates neural progenitor cell differentiation. Sci Rep. 2016;6:1–3.
Liang B, Wang X, Song XS, Bai R, Yang HY, Yang ZM, et al. MicroRNA-20a/b regulates cholesterol efflux through post-transcriptional repression of ATP-binding cassette transporter A1. BBA-Mol Cell Biol L. 2017;1862(9):929–38.
Pernaute B, Spruce T, Smith KM, Sanchez-Nieto JM, Manzanares M, Cobb B, et al. MicroRNAs control the apoptotic threshold in primed Pluripotent stem cells through regulation of BIM. Genes Dev. 2014;28(17):1873–8.
Ghafouri-Fard S, Niazi V, Taheri M. Role of miRNAs and lncRNAs in hematopoietic stem cell differentiation. Non-Coding Rna Res. 2021;6(1):8–14.
Zaiou M, Rihn BH, Bakillah A. Epigenetic regulation of genes involved in the reverse cholesterol transport through interaction with miRNAs. Front Biosci-Landmark. 2018;23:2090–105.
Tai LH, Huang CJ, Choo KB, Cheong SK, Kamarul T. Oxidative stress down-regulates MiR-20b-5p, MiR-106a-5p and E2F1 expression to suppress the G1/S transition of the cell cycle in multipotent stromal cells. Int J Med Sci. 2020;17(4):457–70.
Dong FF, Dong SH, Liang Y, Wang K, Qin YW, Zhao XX. miR-20b inhibits the senescence of human umbilical vein endothelial cells through regulating the Wnt/beta-catenin pathway via the TXNIP/NLRP3 axis. Int J Mol Med. 2020;45(3):847–57.
Robichaud K, Craig PM. Nuclear microRNAs may regulate mitochondrial gene expression following effluent exposure in darter (Etheostoma) species. Comp Biochem Physiol B-Biochem Mol Biol. 2022;262:110754.
Cui JX, Gu LP, Zhong LC, Liu XZ, Sun YN, Xu TJ. microRNA-20–1 and microRNA-101a suppress the NF-kappa B-mediated inflammation production by targeting TRAF6 in Miiuy croaker. Infect Immun. 2022;90(1):e00585.
Zhang MJ, Yin JW, Wu JH, Gu J, Yuan CY, Miao HJ, et al. Circular RNAs are abundant and dynamically expressed during the embryonic lung development of C57BL/6 mice. Heliyon. 2020;6(3):e03437.
Cheng Y-C, Chiang H-Y, Cheng S-J, Chang H-W, Li Y-J, Shieh S-Y. Loss of the tumor suppressor BTG3 drives a pro-angiogenic tumor microenvironment through HIF-1 activation. Cell Death Dis. 2020;11(12):1046.
Lou J, Wang YL, Zhang ZM, Qiu WQ. MiR-20b inhibits mycobacterium tuberculosis induced inflammation in the lung of mice through targeting NLRP3. Exp Cell Res. 2017;358(2):120–8.
Ma H, Wang HL, Luo YL, Guo SJ, Song CW. Mir-20b-induced increase in myeloid-derived suppressor cells in the lungs of mice with chronic asthma. Ann Clin Lab Sci. 2017;47(1):76–82.
Shi J, Duan JY, Gong HJ, Pang YW, Wang L, Yan YJ. Exosomes from miR-20b-3p-overexpressing stromal cells ameliorate calcium oxalate deposition in rat kidney. J Cell Mol Med. 2019;23(11):7268–78.
You HP, Zhang LH, Chen ZY, Liu WF, Wang HG, He HF. MiR-20b-5p relieves neuropathic pain by targeting Akt3 in a chronic constriction injury rat model. Synapse. 2019;73(12):e22125.
Tang GX, Yang MS, Xiang KM, Yang BC, Liu ZL, Zhao SP. MiR-20b-5p modulates inflammation, apoptosis and angiogenesis in severe acute pancreatitis through autophagy by targeting AKT3. Autoimmunity. 2021;54(7):460–70.
Liang ZG, Yao H, Xie RS, Gong CL, Tian Y. MicroRNA-20b-5p promotes ventricular remodeling by targeting the TGF beta-/Smad signaling pathway in a rat model of ischemia-reperfusion injury. Int J Mol Med. 2018;42(2):975–87.
Liu J, Liu YA, Zhang LD, Chen YZ, Du HX, Wen ZL, et al. Down-regulation of circDMNT3B is conducive to intestinal mucosal permeability dysfunction of rats with sepsis via sponging miR-20b-5p. J Cell Mol Med. 2020;24(12):6731–40.
O’Brien J, Hayder H, Zayed Y, Peng C. Overview of MicroRNA biogenesis, mechanisms of actions, and circulation. Front Genet. 2018;9:402.
Tirosh I, Bilu Y, Barkai N. Comparative biology: beyond sequence analysis. Curr Opin Biotechnol. 2007;18(4):371–7.
Morandin C, Tin MMY, Abril S, Gómez C, Pontieri L, Schiøtt M, et al. Comparative transcriptomics reveals the conserved building blocks involved in parallel evolution of diverse phenotypic traits in ants. Genome Biol. 2016;17(1):43.
Warnefors M, Liechti A, Halbert J, Valloton D, Kaessmann H. Conserved microRNA editing in mammalian evolution, development and disease. Genome Biol. 2014;15(6):R83.
Lin H. Computational method in protein structure and function data. Protein Pept Lett. 2020;27(4):257–8.
Kim S. Computational model for predicting the relationship between Micro-RNAs and their target messenger RNAs in breast and colon cancers. Cancer Inform. 2018;17:1176935118785145.
Denechaud PD, Fajas L, Giralt A. E2F1, a novel regulator of metabolism. Front Endocrinol. 2017;8:311.
Honardoost MA, Naghavian R, Ahmadinejad F, Hosseini A, Ghaedi K. Integrative computational mRNA-miRNA interaction analyses of the autoimmune-deregulated miRNAs and well-known Th17 differentiation regulators: An attempt to discover new potential miRNAs involved in Th17 differentiation. Gene. 2015;572(2):153–62.
Kramer N, Walzl A, Unger C, Rosner M, Krupitza G, Hengstschläger M, et al. In vitro cell migration and invasion assays. Mutat Res/Rev Mutat Res. 2013;752(1):10–24.
Lu PW, Gu YT, Li L, Wang F, Yang X, Yang YQ. Long noncoding RNA CAMTA1 promotes proliferation and mobility of the human breast cancer cell line MDA-MB-231 via targeting miR-20b. Oncol Res. 2018;26(4):625–35.
Li SF, Qiang Q, Shan HT, Shi MK, Gan GM, Ma F, et al. MiR-20a and miR-20b negatively regulate autophagy by targeting RB1CC1/FIP200 in breast cancer cells. Life Sci. 2016;147:143–52.
Xia LQ, Li F, Qiu J, Feng ZM, Xu ZH, Chen ZT, et al. Oncogenic miR-20b-5p contributes to malignant behaviors of breast cancer stem cells by bidirectionally regulating CCND1 and E2F1. BMC Cancer. 2020;20(1):1–3.
Zhan ZJ, Xu XY, Li ZN, Chen XJ, Li Q, Wang XL. Effect of miR-20 on apoptosis of ovarian cancer cells by regulating TIMP-2 pathway. J Biomater Tissue Eng. 2020;10(6):858–61.
Zhong QA, Xiong Y, Ling C, Qian YP, Zhao X, Yang HS. Enhancing the sensitivity of ovarian cancer cells to olaparib via microRNA-20b-mediated cyclin D1 targeting. Exp Biol Med. 2021;246(11):1297–306.
Guo J, Xiao ZW, Yu XW, Cao RF. miR-20b promotes cellular proliferation and migration by directly regulating phosphatase and tensin homolog in prostate cancer. Oncol Lett. 2017;14(6):6895–900.
Qi JC, Yang Z, Zhang YP, Lu BS, Yin YW, Liu KL, et al. miR-20b-5p, TGFBR2, and E2F1 form a regulatory loop to participate in epithelial to mesenchymal transition in prostate cancer. Front Oncol. 2020;9:1535.
Cheng Y, Geng L, Zhao LJ, Zuo P, Wang JL. Human papillomavirus E6-regulated microRNA-20b promotes invasion in cervical cancer by targeting tissue inhibitor of metalloproteinase 2. Mol Med Rep. 2017;16(4):5464–70.
Peng LJ, Li SB, Li YC, Wan MH, Fang XS, Zhao YX, et al. Regulation of BTG3 by microRNA-20b-5p in non-small cell lung cancer. Oncol Lett. 2019;18(1):137–44.
Wang B, Yang J, Xiao B. MicroRNA-20b (miR-20b) promotes the proliferation, migration, invasion, and tumorigenicity in esophageal cancer cells via the regulation of phosphatase and tensin homologue expression. PLoS ONE. 2016;11(10):e0164105.
Fu Q, Cheng J, Zhang JD, Zhang YL, Chen XB, Luo SX, et al. miR-20b reduces 5-FU resistance by suppressing the ADAM9/EGFR signaling pathway in colon cancer. Oncol Rep. 2017;37(1):123–30.
Tang DX, Yang Z, Long FX, Luo L, Yang B, Zhu RY, et al. Long noncoding RNA MALAT1 mediates stem cell-like properties in human colorectal cancer cells by regulating miR-20b-5p/Oct4 axis. J Cell Physiol. 2019;234(11):20816–28.
Zhu JJ, Chen LX, Zou LT, Yang PP, Wu RR, Mao Y, et al. MiR-20b,-21, and-130b inhibit PTEN expression resulting in B7–H1 over-expression in advanced colorectal cancer. Hum Immunol. 2014;75(4):348–53.
Hua RH, Zhang Y, Yan XY, Tang D, Li XL, Ni QF, et al. Syndecan-2, negatively regulated by miR-20b-5p, contributes to 5-fluorouracil resistance of colorectal cancer cells via the JNK/ERK signaling pathway. Acta Biochim Biophys Sin. 2021;53(11):1547–57.
Park SL, Cho TM, Won SY, Song JH, Noh DH, Kim WJ, et al. MicroRNA-20b inhibits the proliferation, migration and invasion of bladder cancer EJ cells via the targeting of cell cycle regulation and Sp-1-mediated MMP-2 expression. Oncol Rep. 2015;34(3):1605–12.
Hong SB, Yu S, Li J, Yin YL, Liu YJ, Zhang Q, et al. MiR-20b displays tumor-suppressor functions in papillary thyroid carcinoma by regulating the MAPK/ERK signaling pathway. Thyroid. 2016;26(12):1733–43.
Pang RZ, Yang S. lncRNA DUXAP8 inhibits papillary thyroid carcinoma cell apoptosis via sponging the miR-20b-5p/SOS1 axis. Oncol Rep. 2021;45(5):1.
He J, Mu MM, Luo YL, Wang HL, Ma H, Guo SJ, et al. MicroRNA-20b promotes proliferation of H22 hepatocellular carcinoma cells by targeting PTEN. Oncol Lett. 2019;17(3):2931–6.
Li L, Yu SJ, Chen JD, Quan M, Gao Y, Li YD. miR-15a and miR-20b sensitize hepatocellular carcinoma cells to sorafenib through repressing CDC37L1 and consequent PPIA downregulation. Cell Death Discov. 2022;8(1):1.
Luo YL, He J, Tao XN, Wang HL, Fang Q, Guo SJ, et al. miR-20b negatively regulates VEGF expression by targeting STAT3 in H22 hepatocellular carcinoma cells. Oncol Rep. 2018;40(5):2806–13.
Li YF, Chen DQ, Jin L, Liu JJ, Su ZM, Li YC, et al. MicroRNA-20b-5p functions as a tumor suppressor in renal cell carcinoma by regulating cellular proliferation, migration and apoptosis. Mol Med Rep. 2016;13(2):1895–901.
Danza K, Silvestris N, Simone G, Signorile M, Saragoni L, Brunetti O, et al. Role of miR-27a, miR-181a and miR-20b in gastric cancer hypoxia-induced chemoresistance. Cancer Biol Ther. 2016;17(4):400–6.
Zhu H, Jin YM, Lyu XM, Fan LM, Wu F. Long noncoding RNA H19 regulates HIF-1 alpha/AXL signaling through inhibiting miR-20b-5p in endometrial cancer. Cell Cycle. 2019;18(19):2454–64.
Abasi M, Kohram F, Fallah P, Arashkia A, Soleimani M, Zarghami N, et al. Differential maturation of miR-17 similar to 92 cluster members in human cancer cell lines. Appl Biochem Biotechnol. 2017;182(4):1540–7.
Li DP, Ilnytskyy Y, Kovalchuk A, Khachigian LM, Bronson RT, Wang B, et al. Crucial role for early growth response-1 in the transcriptional regulation of miR-20b in breast cancer. Oncotarget. 2013;4(9):1373–87.
Zhou WD, Shi GX, Zhang QY, Wu QW, Li BA, Zhang ZM. MicroRNA-20b promotes cell growth of breast cancer cells partly via targeting phosphatase and tensin homologue (PTEN). Cell Biosci. 2014;4:1176935118785145.
Worby CA, Dixon JE. PTEN. Annu Rev Biochem. 2014;83:641–69.
Zhang RK, Li FW, Wang YC, Yao M, Chi CL. Prognostic value of microRNA-20b expression level in patients with prostate cancer. Histol Histopathol. 2020;35(8):827–31.
Atri C, Guerfali FZ, Laouini D. Chapter 6: MicroRNAs in diagnosis and therapeutics. In: Mallick B, editor. AGO-Driven Non-Coding RNAs. Academic Press; 2019. p. 137–77.
Zhou C, Chen Z, Zhao L, Zhao W, Zhu Y, Liu J, et al. A novel circulating miRNA-based signature for the early diagnosis and prognosis prediction of non-small-cell lung cancer. J Clin Lab Anal. 2020;34(11): e23505.
Zhang W, Liu HT. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002;12(1):9–18.
Geiger TR, Peeper DS. Metastasis mechanisms. Biochimica et Biophysica Acta (BBa) Rev Cancer. 2009;1796(2):293–308.
Diepenbruck M, Christofori G. Epithelial–mesenchymal transition (EMT) and metastasis: yes, no, maybe? Curr Opin Cell Biol. 2016;43:7–13.
Savagner P. The epithelial–mesenchymal transition (EMT) phenomenon. Ann Oncol. 2010;21:vii89–92.
Wang Z. The guideline of the design and validation of MiRNA mimics. Methods Mol Biol (Clifton, NJ). 2011;676:211–23.
Thomson DW, Dinger ME. Endogenous microRNA sponges: evidence and controversy. Nat Rev Genet. 2016;17(5):272–83.
Olusola P, Banerjee HN, Philley JV, Dasgupta S. Human papilloma virus-associated cervical cancer and health disparities. Cells. 2019;8(6):622.
Mahalanobish S, Saha S, Dutta S, Sil PC. Matrix metalloproteinase: an upcoming therapeutic approach for idiopathic pulmonary fibrosis. Pharmacol Res. 2020;152: 104591.
Peglion F, Capuana L, Perfettini I, Boucontet L, Braithwaite B, Colucci-Guyon E, et al. PTEN inhibits AMPK to control collective migration. Nat Commun. 2022;13(1):4528.
Griffioen AW, Dudley AC. Angiogenesis: a year in review. Angiogenesis. 2021;24(2):195–6.
Tang Y, Zong S, Zeng H, Ruan X, Yao L, Han S, et al. MicroRNAs and angiogenesis: a new era for the management of colorectal cancer. Cancer Cell Int. 2021;21(1):221.
Yu Z, Pestell TG, Lisanti MP, Pestell RG. Cancer stem cells. Int J Biochem Cell Biol. 2012;44(12):2144–51.
Tashiro E, Tsuchiya A, Imoto M. Functions of cyclin D1 as an oncogene and regulation of cyclin D1 expression. Cancer Sci. 2007;98(5):629–35.
Ullah Shah A, Mahjabeen I, Kayani MA. Genetic polymorphisms in cell cycle regulatory genes CCND1 and CDK4 are associated with susceptibility to breast cancer. J BUON Off J Balkan Union Oncol. 2015;20(4):985–93.
Glick D, Barth S, Macleod KF. Autophagy: cellular and molecular mechanisms. J Pathol. 2010;221(1):3–12.
Yun CW, Lee SH. The roles of autophagy in cancer. Int J Mol Sci. 2018;19(11):3466.
Bhutia SK, Mukhopadhyay S, Sinha N, Das DN, Panda PK, Patra SK, et al. Autophagy: Cancer’s friend or foe? Adv Cancer Res. 2013;118:61–95.
Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35(4):495–516.
Shirjang S, Mansoori B, Asghari S, Duijf PHG, Mohammadi A, Gjerstorff M, et al. MicroRNAs in cancer cell death pathways: apoptosis and necroptosis. Free Radical Biol Med. 2019;139:1–15.
Yue J, López JM. Understanding MAPK signaling pathways in apoptosis. Int J Mol Sci. 2020;21(7):2346.
Huang D, Ding Y, Luo WM, Bender S, Qian CN, Kort E, et al. Inhibition of MAPK kinase signaling pathways suppressed renal cell carcinoma growth and angiogenesis in vivo. Can Res. 2008;68(1):81–8.
Li L, Tao X, Li Y, Gao Y, Li Q. CDC37L1 acts as a suppressor of migration and proliferation in gastric cancer by down-regulating CDK6. J Cancer. 2021;12(11):3145–53.
Haoyuan MA, Yanshu LI. Structure, regulatory factors and cancer-related physiological effects of ADAM9. Cell Adh Migr. 2020;14(1):165–81.
Chou CW, Huang YK, Kuo TT, Liu JP, Sher YP. An overview of ADAM9: structure, activation, and regulation in human diseases. Int J Mol Sci. 2020;21(20):7790.
Strimbu K, Tavel JA. What are biomarkers? Curr Opin HIV AIDS. 2010;5(6):463–6.
Condrat CE, Thompson DC, Barbu MG, Bugnar OL, Boboc A, Cretoiu D, et al. miRNAs as biomarkers in disease: latest findings regarding their role in diagnosis and prognosis. Cells. 2020;9(2):276.
Li MH, Zhou Y, Xia TS, Zhou X, Huang ZB, Zhang H, et al. Circulating microRNAs from the miR-106a-363 cluster on chromosome X as novel diagnostic biomarkers for breast cancer. Breast Cancer Res Treat. 2018;170(2):257–70.
Fan XC, Zou X, Liu C, Cheng WF, Zhang SY, Geng XN, et al. MicroRNA expression profile in serum reveals novel diagnostic biomarkers for endometrial cancer. Biosci Rep. 2021;41(4).
Hui ABY, Lin A, Xu W, Waldron L, Perez-Ordonez B, Weinreb I, et al. Potentially prognostic miRNAs in HPV-associated oropharyngeal carcinoma. Clin Cancer Res. 2013;19(8):2154–62.
Mohsenikia M, Khalighfard S, Alizadeh AM, Khori V, Zanjan MG, Zare M, et al. An innovative systematic approach introduced the involved lncRNA-miR-mRNA network in cell cycle and proliferation after conventional treatments in breast cancer patients. Cell Cycle. 2022;21(16):1753–74.
Sueta A, Yamamoto Y, Tomiguchi M, Takeshita T, Yamamoto-Ibusuki M, Iwase H. Differential expression of exosomal miRNAs between breast cancer patients with and without recurrence. Oncotarget. 2017;8(41):69934–44.
Hu G, Drescher K, Chen X. Exosomal miRNAs: biological properties and therapeutic potential. Front Genet. 2012;3:56.
Acknowledgements
Not applicable.
Funding
No funders.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and the main idea of the work. AI, FA, SG, BZTA, SGS, DD, MMN, and AFY drafted the main text, figures, and tables. FK and AFY supervised the work and provided the comments and additional scientific information. F.K also reviewed and revised the text. All authors read and approved the final version of the work to be published.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
İlhan, A., Golestani, S., Shafagh, S.G. et al. The dual role of microRNA (miR)-20b in cancers: Friend or foe?. Cell Commun Signal 21, 26 (2023). https://doi.org/10.1186/s12964-022-01019-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12964-022-01019-7