Abstract
Purpose
Exosomal microRNAs have been identified as important mediators of communication between tumor cells and macrophages in the microenvironment. miR-541-5p was reported to be involved in hepatocellular carcinoma progression, but its role in gastric cancer (GC) and in GC cell-macrophage crosstalk is unknown.
Methods
Cell proliferation, migration and invasion were respectively assessed by CCK-8 assay, scratch and Transwell assays. RT-qPCR was used to detect the level of miR-541-5p, macrophage markers and DUSP3. The percentage of CD11b+CD206+ cell population was analyzed by flow cytometry. Western blotting was employed to evaluate DUSP3-JAK2/STAT3 pathway proteins and exosome markers. The interaction between miR-541-5p and DUSP3 was verified by luciferase assay.
Results
The results showed that miR-541-5p was upregulated in GC tissues and cells, and stimulated GC cell growth, migration and invasion in vitro. GC cells induce M2 macrophage polarization by secreting the exosomal miR-541-5p. Exosomal miR-541-5p maintained JAK2/STAT3 pathway activation in macrophages by targeting negative regulation of DUSP3. Inhibiting miR-541-5p significantly limited tumor growth in vivo.
Conclusion
In conclusion, miR-541-5p promotes GC cell progression. GC cells may induce macrophage M2 polarization through the exosomal miR-541-5p-mediated DUSP3/JAK2/STAT3 pathway. miR-541-5p may be a potential therapeutic target for GC.
Similar content being viewed by others
Introduction
Gastric cancer (GC) is a widespread digestive system malignancy, with over 1 million new cases of GC being reported globally annually [1]. Despite partially improved clinical outcomes with surgery, radiotherapy and chemotherapy, GC remains the fifth most prevalent cancer and the third most frequent contributor to cancer-related deaths worldwide [2]. Recently, a remarkable advancement in GC molecular targeted therapies and immunotherapies has been incredible, indicating a promising outlook for therapeutic intervention [3]. Early detection of GC has been shown to improve patient survival and prognosis, yet the biomarkers currently employed for GC diagnosis and prognostic evaluation are not very sensitive or specific [4]. Hence, it is essential to explore new biomarkers and targets for early diagnosis and treatment of GC.
Tumor-associated macrophages are the main immune cells for tumor infiltration, dividing into two subtypes: M1, typically activated, and M2, specifically activated macrophages. M1 macrophages have a pro-immune, pro-inflammatory, and anti-tumor effect, while M2 macrophages have an immunosuppressive and anti-inflammatory effect, which helps to advance tumor progression [5]. In tumor tissues, monocytes were recruited into the tumor microenvironment to differentiate into macrophages, which polarized into M1 or M2 macrophages under multiple stimuli, including cytokines and chemokines. M1 macrophages are characterized by CD80, CD86, TNF-α, IL-8, and iNOS, while M2 macrophages are characterized by CD163, CD206, TGF-β, IL-10, and Arg1 [6].
Exosomes are 30 ~ 150 nm extracellular vesicles that contain a variety of biomolecules from parent cells, such as lipids, proteins, and microRNAs (miRNAs). Exosomes are taken up by target cells through endocytosis, transmitting biological signals between cells [7]. MiRNAs are small, non-coding RNAs that carry out a variety of biological activities, such as breaking down mRNAs or obstructing their translation to control post-transcriptional gene expression [8]. The link between miRNAs and cancer has been extensively studied, with multiple miRNAs playing a role in cancer progression and regulating tumor immunity and microenvironment [9]. There is growing evidence that miRNAs are tissue-specific and can be used as noninvasive, sensitive tumor diagnostic markers [10]. MiRNA therapy by controlling the expression of miRNAs with carcinogenic or tumor-suppressing properties may be an effective cancer treatment strategy [11]. LI et al. found that miR-541-5p could promote the proliferation, migration and invasion of liver cancer cells in a laboratory environment, and knocking down miR-541-5p can obstruct the growth of liver cancer tumors in nude mice [12]. Studies have reported that exosome miRNAs derived from tumor cells can be uptake by tumor-associated macrophages and promote macrophage M2 polarization, accelerating tumor progression [13]. But the role of miR-541-5p in GC cell-macrophage crosstalk is not clear.
This research aims to investigate the precise function of miR-541-5p in GC and to determine if miR-541-5p can be released in exosome form and involved in the interaction between GC cells and macrophages.
Materials and methods
Patient tissue samples
Tissue samples including GC tissues and normal tissues were provided by GC patients. all of which were obtained with informed consent and approved by the ethics committee of Shaanxi Provincial People’s Hospital. The fresh tissues were frozen and stored in liquid nitrogen until they were used.
Database analysis
Stomach adenocarcinoma is the main type of GC. The UALCAN database (https://ualcan.path.uab.edu/) was utilized to analyze the expression of miR-541-5p in normal samples and stomach adenocarcinoma samples. The expression levels of miR-541-5p were also analyzed in samples with different stages and grades as well as lymph node metastases. With the Kaplan-Meier Plotter online tool (https://kmplot.com/analysis/), Kaplan-Meier survival analysis and log-rank test were conducted to ascertain the correlation between miR-541-5p level and the overall survival of stomach adenocarcinoma patients.
Cell culture and treatment
The human monocyte THP1, human gastric epithelial cell (GES-1), and GC cell lines (SGC-7901, AGS, MKN-45, and HGC-27) were obtained from ATCC (Manassas, VA, USA). GES-1 cells were cultured in DMEM medium (Gibco, USA), and other cells in RPMI-1640 medium (Gibco, USA). The nutrient medium was enriched with 10% fetal bovine serum (FBS, Gibco, USA), 1% antibiotic (HyClone, USA), and 0.1% mycoplasma antibiotic (Invitrogen, USA). All cells were grown and proliferated in a humidified incubator at 37 °C with 5% CO2. To explore macrophage polarization, THP1 cells were cultured with medium containing 100 ng/mL phorbol-12-myristate-13-acetate (PMA) (Selleck, China) for 36 h and then cultured in medium without PMA for 3 days to induce THP1 differentiation into macrophage THP1 (Mφ). To inhibit exosome secretion by SGC-7901 and HGC-27 cells, GW4869 (10 µM, Selleck, China) was added to the culture medium for 24 h.
Cell transfection assay
The cells were introduced into a 6-well, with a density of 5 × 106 per well, and subsequently, delicate cells were transfected after achieving growth stabilization. SGC-7901 cells were transfected with miR-NC (Invitrogen, Catalog # 4,464,059) or miR-541-5p mimic (Invitrogen, Assay ID MC12516, Catalog # 4,464,066) via lipofectamine 2000 (Invitrogen, USA) following the given instructions. HGC-27 cells were transfected with inhibitor-NC (Invitrogen, Catalog # 4,464,078) or miR-541-5p inhibitor (Invitrogen, Assay ID MH12516, Catalog # 4,464,084). The negative control (ov-NC) and DUSP3 overexpression plasmid (ov-DUSP3) were transfected into THP1 (Mφ) cells. The plasmids utilized in this research were meticulously crafted and engineered by Gene Pharma company (Suzhou, China).
CCK-8 assay
By the guidelines provided by the manufacturer, 10 µl of CCK-8 reagents were incorporated into every pore of transfected cells at intervals of 0, 24, 48, and 72 h, followed by a 2 h incubation at 37 °C. Subsequently, a microplate reader was utilized to measure the optical density (OD) at 450 nm.
Scratch assay
SGC-7901 and HGC-27 cells in the logarithmic growth phase were harvested and seeded at a density of 5 × 105 cells/mL in 6-well plates, with each well containing 2 mL of cell suspension. Following incubation for 24 h, a linear scratch was created across the monolayer using a smooth and continuous motion with a 100 µl pipette tip. The cells crossed off were washed 1 ~ 2 times with PBS buffer, and the photos were taken by an inverted microscope for 0 h. After 24 h, the cells were detected under an inverted microscope, taking care to take another 24 h photo in the same place as the 0 h position. Analyze using ImageJ software.
Transwell assay
Using a Transwell invasion chamber (Corning, USA), the upper and lower chambers with an 8 μm pore diameter were coated with Matrigel Matrix gel. The lower chamber contained 600 µl of culture medium supplemented with 10% FBS, and the upper chamber was filled with 100 µl of culture medium without FBS. A cell suspension of 30 µl was added to the upper compartment and incubated in a CO2 incubator for 24 h. Following fixation with methanol and staining using a solution of 0.2% crystal violet, ten non-repeating fields of view were examined under a microscope to quantify the number of cells that successfully penetrated through Matrigel Matrix within each field. This experimental procedure was repeated three times to ensure reliable results for subsequent analysis.
Co-culture assay
In the co-culture system, 6-well plates and 0.4 μm pore size Transwell chambers (Corning, USA) were used. THP1 (Mφ) cells (3 × 105) were inoculated into the lower chambers, and SGC-7901 or HGC-27 cells (3 × 105) were inoculated into the upper chambers. SGC-7901 and HGC-27 cells were either treated or untreated with GW4869. After 48 h of co-culture, M1 and M2 macrophage markers were detected in THP1 (Mφ) cells.
Exosome isolation
The conditioned medium of GES-1 cells, GC cells and THP1 (Mφ) cells was subjected to centrifugation at 100 × g for 10 min, 2000 × g for 30 min, respectively, in order to remove cell and debris. Total exosome isolation kit (Invitrogen; 4,478,359) was used to isolate exosomes in accordance with the manufacturer’s manual. Exosome precipitates were resuspended in PBS solution and stored at -80 °C. The levels of miR-541-5p detected in the exosomes were normalized to total number of exosomes.
RT-qPCR
TRIZOL reagents (Invitrogen, USA) were employed to obtain total RNA from THP1 (Mφ) cells, GC cells, and tissues. The New Poly (A) tail kit (ThermoFisher, USA) and PrimeScript RT Master Mix Kit (TaKaRa, Japan) were utilized to transcribe miRNA and mRNA into cDNA, adhering to the instructions provided by the manufacturer. The FastStart® General SYBR Green Master Kit (Roche, Mannheim, Germany) was utilized to amplify these cDNAs, which were subsequently examined using an ABI PRISM 790HT Sequencing System (Applied Biosystems, Waltham, MA, USA). The primer sequences were as follows: miR-541-5p (F: 5’-CGAAAGGATTCTGCTGTCGGT-3’; R: 5’-AGTGCAGGGTCCGAGGTATT-3’), U6 (F: 5’-CTCGCTTCGGCAGCACATATACTA-3’; R: 5’-ACGAATTTGCGTGTCATCCTTGCG-3’), DUSP3 (F: 5’-GGGTGATGCCCAGTTTCT-3’; R: 5’-GATCTCAACGACCTGCTCTC-3’), GAPDH (F: 5’-GCACCGTCAAGGCTGAGAAC-3’; R: 5’-GCCTTCTCCATGGTGGTGAA-3’), CD163 (F: 5’-CGAGTTAACGCCAGTAAGG-3’; R: 5’-GAACATGTCACGCCAGC-3’), TGF-β (F: 5’-CGTGGAGCTGTACCAGAAATAC-3’; R: 5’-CACAACTCCGGTGACATCAA-3’), IL-10 (F: 5’-GAGAACCAAGACCCAGACATCA-3’; R: 5’-AAGGCATTCTTCACCTGCTCCAC-3’), Arg1 (F: 5’-TGCCCTTTGCTGACATCCCTAAT-3’; R: 5’-CTTCTTGACTTCTGCCACCTT-3’), TNF-α (F: 5’-TGACAAGCCTGTAGCCC-3’; R: 5’-CCCTTGAAGAGGACCTGG-3’), iNOS (F: 5’-GAGGCAAACAGCACATTCAG-3’; R: 5’-CAGGAGAGTTCCACCAGGAT-3’).
Western blot
The RIPA lysis buffer (Beyotime, Shanghai, China) supplemented with protease and phosphatase inhibitors (NCM Biotech, Suzhou, China) was employed to acquire cells and tissue proteins. Subsequently, the lysate was augmented with SDS-PAGE protein buffer (Beyotime, Shanghai, China) and subjected to denaturation in a 100 °C water bath for 5 min. The protein samples were isolated using SDS-polyacrylamide gel electrophoresis and transferred onto a polyvinylidene difluoride membrane (Thermo Fisher, MA, USA). A 5% rapid blocking buffer (Beyotime, Shanghai, China) was used to obstruct the membrane for 45 min. The membranes were cut and then incubated overnight with the primary antibody at 4 °C. Subsequently, the membrane blots underwent immunoblotting with the secondary antibody for a duration of 2 h, and subsequently captured utilizing a Tanon4600 imaging system (Biotanon, China) with chemiluminescent reagents (Advansta, CA, USA). The antibodies used in this study were as follows: anti-DUSP3 (ab125077, 1: 1,0000); anti-p-JAK2 (ab32101, 1:1,000); anti-JAK2 (ab108596, 1:1,000); anti-p-STAT3 (ab76315, 1:1,000); anti-STAT3 (ab68153, 1:1,000); anti-GAPDH (ab9485, 1:1,0000); anti-CD63 (ab68418, 1:1,000); anti-TSG101 (ab125011, 1:1,000); anti-Alix (ab275377, 1:1,000); anti-CD163 (ab182422, 1:1,000) (Abcam, Cambridge, MA, USA), and horseradish peroxidase-conjugated goat anti-rabbit secondary antibody (ZB-5301, ZSGB-BIO, Beijing, China).
Flow cytometry
To identify CD11b+CD206+ macrophages, THP-1(Mφ) cells were first collected and fixed with 1% paraformaldehyde at 4 °C overnight. The cells were then immersed in flow cytometry buffer (1×PBS buffer containing 1% FSA) and stained with anti-CD11b-FITC and anti-CD206-APC (Invitrogen, USA) for 30 min at ambient temperature. Finally, the labeled cells were analyzed on a flow cytometer (BD Biosciences, USA).
Dual luciferase reporter assay
The TargetScan database (http://www.targetscan.org/vert_71) was used to anticipate the binding site of miR-541-5p and DUSP3 (DUSP3-WT) and carry out site mutation (DUSP3-MUT). WT/MUT plasmids cloned into pmirGLO. mimic-NC, miR-541-5p mimic, DUSP3-WT or DUSP3-MUT were transfected into THP1 (Mφ) cells. Two days later, the Dual-Luciferase Reporter Kit (Promega, San Luis Obispo, WI, USA) was employed to assay luciferase activity utilizing the SpectraMax i3X Enzyme Labeler (Molecular Devices, San Jose, CA, USA).
Animal models
The Laboratory Animal Centre is where mice were acquired and kept in a sterile environment. Six-week-old female nude mice were randomly allocated to either the inhibitor-NC group or the miR-541-5p inhibitor group to generate a model of subcutaneous tumorigenesis. Each mouse’s forelimb axils were injected with 1 × 107 HGC-27 cell suspension subcutaneously. Following a period of 4 weeks, the mice were sacrificed and their subcutaneous tumors were extracted for weight evaluation. Regarding euthanasia, we employed cervical dislocation for the mice. The mice were not anesthetized before the procedure. The cervical vertebrae were dislocated by grasping the back of the head and neck firmly and applying a rapid, forceful upward and backward movement, resulting in immediate death. The Animal Ethics Committee of Shaanxi Provincial People’s Hospital reviewed and approved the animal experimental protocol of this study.
ELISA assay
According to the manufacturer’s manual, anti-inflammatory factors TGF-β and IL-10 levels in nude mouse tumor tissues were measured by using the mouse TGF-β ELISA kit and the mouse IL-10 ELISA kit (Abcam, USA). Within 20 min at the end of the reaction, the absorbance was quantified at 450 nm using a spectrophotometer (Thermo Fisher Scientific, USA), and then the TGF-β and IL-10 content was calculated from the normalized curve.
Statistical analysis
The experiments were conducted over three repetitions. The quantitative data was expressed using the Mean ± standard deviation method. The t-test was used to compare the statistical differences between the two groups, and the ANOVA was used to assess the differences between the groups. The Kaplan-Meier method and log-rank were utilized to evaluate the overall survival. A p-value of less than 0.05 was deemed to be statistically significant.
Results
MiR-541-5p expression was upregulated in GC tissues and cells
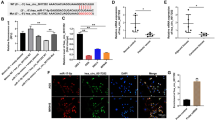
Bioinformatics analysis revealed that miR-541-5p was increased in individuals with GC (Fig. 1A). As tumor stage, grade, and lymph node metastasis progressed, miR-541-5p expression increased (Fig. 1B-1D), suggesting a potential link between miR-541-5p and the advancement of GC. The poorer prognosis of GC patients may be related to miR-541-5p levels, as evidenced by the significantly lower overall survival of GC patients with higher miR-541-5p expression (Fig. 1E). Furthermore, the up-regulation of miR-541-5p expression in GC tissues was confirmed by RT-qPCR (Fig. 1F). GC cell lines also exhibited a higher miR-541-5p expression than normal cell lines (Fig. 1G). These results suggest that miR-541-5p may be associated with the development of GC.
The expression of miR-541-5p was increased in GC tissues and cells. (A) The expression of miR-541-5p in normal and GC samples was analyzed using the UALCAN database. (B-D) The expression of miR-541-5p was characterized in normal samples and GC samples with different stages, grades as well as lymph node metastases using the UALCAN database. (E) Kaplan-Meier survival analysis and log-rank test were used to determine the association of miR-541-5p with overall survival. (F) The expression of miR-541-5p in collected GC tissues and adjacent normal tissues. (G) The expression of miR-541-5p in the human gastric epithelial cell line GES-1 and GC cell lines SGC-7901, AGS, MKN-45 and HGC-27 (*P < 0.05 vs. normal or GES-1 group)
MiR-541-5p stimulated the proliferation, migration, and invasion of GC cells
Figure 1G illustrated that miR-541-5p exhibited the lowest expression in SGC-7901 cells and the highest expression in HGC-27 cells. Therefore, miR-541-5p mimic was transfected into SGC-7901 cells to promote miR-541-5p expression (Fig. 2A), and miR-541-5p inhibitor was transfected into HGC-27 cells to inhibit miR-541-5p expression (Fig. 2B). Subsequent cell function experiments showed that overexpression of miR-541-5p enhanced the capacity of SGC-7901 cells to proliferate, migrate and invade (Fig. 2C, E and G). In contrast, inhibiting miR-541-5p weakened the ability of HGC-27 cells to proliferate, migrate and invade (Fig. 2D, F and H). The results indicated that miR-541-5p stimulated malignant activity in GC cells.
miR-541-5p promoted proliferation, migration, and invasion of GC cells. (A) miR-541-5p overexpression efficiency in SGC-7901 cells. (B) miR-541-5p interference efficiency in HGC-27 cells. (C) The effect of overexpression of miR-541-5p on the proliferation of SGC-7901 cells. (D) Inhibition of miR-541-5p on the proliferation of HGC-27 cells. (E) The effect of overexpression of miR-541-5p on cell migration in SGC-7901 cells. (F) The effect of inhibition of miR-541-5p on HGC-27 cell migration. (G) The effect of overexpression of miR-541-5p on cell invasion in SGC-7901 cells. (H) The effect of inhibition of miR-541-5p on HGC-27 cell invasion. *P < 0.05 vs. control group
GC cells induce M2 macrophage polarization through exosome secretion
To investigate the influence of GC cells on macrophage polarization, PMA-induced differentiated THP1 (Mφ) macrophages were cultured alone or co-cultured with SGC-7901 cells and HGC-27 cells. SGC-7901 cells and HGC-27 cells were treated with or without the exosome inhibitor GW4869. The M1 (TNF-α and iNOS) and M2 markers (CD163, CD206, TGF-β, IL-10 and Arg1) of THP1 (Mφ) cells were detected. Co-culture with SGC-7901 and HGC-72 cells resulted in increased levels of CD163, TGF-β, IL-10 and Arg1 (Fig. 3A and B) and decreased levels of TNF-α and iNOS (Fig. 3C and D), as well as an increased proportion of CD11b+CD206+ cells in THP1 (Mφ) cells (Fig. 3E). The above results suggest that co-culture with GC cells promotes M2 polarization of THP1 (Mφ) cells. As shown in Fig. 3A-E, the addition of GW4869 significantly attenuated the effect of co-culture of SGC-7901 and HGC-27 cells on the expression of M1 and M2 markers in THP1 (Mφ) cells. This implies that SGC-7901 and HGC-27 cells may regulate THP1 (Mφ) cell polarization through exosomes. To verify this, exosomes from SGC-7901 and HGC-27 cells were isolated. The exosome markers CD63, TSG101 and Alix were all detected and both groups with GW4869 added had very little marker proteins (Fig. 3F). The exosomes isolated from SGC-7901 and HGC-27 cells significantly increased the percentage of CD11b+CD206+ cells in THP1 (Mφ) cells (Fig. 3G). This suggests that GC cells are able to induce M2 macrophage polarization by secreting exosomes.
GC cells induced M2 macrophage polarization through exosome secretion. (A, B) The expression of M2 markers CD163, TGF-β, IL-10, and Arg1 in THP1 (Mφ) cells. (C, D) The expression of M1 markers TNF-α and iNOS in THP1 (Mφ) cells. (E) Percentage of CD11b+CD206+ cells in THP1 (Mφ) cells (*P < 0.05 vs. NC group, #P < 0.05 vs. SGC-7901 group, &P < 0.05 vs. HGC-27 group). (F) The expression of exosome markers CD63, TSG101, and Alix (*P < 0.05 vs. SGC-7901-Exo group, #P < 0.05 vs. HGC-27-Exo group). (G) Percentage of CD11b+CD206+ cells in THP1 (Mφ) cells (*P < 0.05 vs. PBS group, #P < 0.05 vs. SGC-7901-Exo group, &P < 0.05 vs. HGC-27-Exo group)
GC cells induced polarization of M2 macrophages through exosomal miR-541-5p
The exosomes secreted by GC cells usually contain biological molecules such as miRNA. Consequently, miR-541-5p was identified in the exosomes isolated from GES-1 and GC cells (SGC-7901, AGS, MKN-45, and HGC-27). The findings indicated that miR-541-5p expression in the exosomes of GC cells was markedly higher than that of GES-1 (Fig. 4A). THP1 cells themselves also release exosomes containing miR-541-5p, but the miR-541-5p level is low (Fig. 4A). To investigate whether GC cells regulate M2 macrophage polarization through exosomal miR-541-5p, we up-regulated miR-541-5p in SGC-7901 cells and down-regulated it in HGC-27 cells, and then treated THP1 (Mφ) cells with cell exosomes. The findings showed that miR-541-5p was significantly increased in SGC-7901 (miR-541-5p mimic)-Exo group (Fig. 4B), and the expression of M2 markers CD163, TGF-β, IL-10 and Arg1 was up-regulated considerably (Fig. 4D). The percentage of CD11b+CD206+ cells was also obviously increased (Fig. 4F). When THP1 (Mφ) cells were exposed to exosomes from HGC-27 (miR-541-5p inhibitor) cells, the expression of miR-541-5p was decreased (Fig. 4C), the M2 markers was reduced (Fig. 4E), and the amount of CD11b+CD206+ cells was decreased (Fig. 4G). This suggests that GC cells may promote M2 macrophage polarization by secreting the exosomal miR-541-5p. To further clarify the transfer of miR-541-5p from GC cells to THP1 (Mφ) cells, THP1 (Mφ) cells were cultured alone (NC group) or co-cultured with un-transfected/transfected SGC-7901 cells and HGC-27 cells. The results showed that both co-cultures significantly promoted miR-541-5p expression in THP1 (Mφ) cells (Fig. S1A, S1B). SGC-7901 cells transfected with miR-541-5p mimic further increased miR-541-5p levels, while HGC-27 cells transfected with miR-541-5p inhibitor decreased miR-541-5p levels (Fig. S1A, B). In addition, it’s observed that the use of miR-541-5p mimic or inhibitor similarly changed the level of miR-541-5p in exosomes in the co-culture medium (Fig. S1C, S1D).
GC cells induced polarization of M2 macrophages through exosome miR-541-5p. (A) The expression of miR-541-5p in exosomes from human gastric epithelial cell line GES-1, GC cell lines SGC-7901, AGS, MKN-45 and HGC-27, as well as human monocyte cell line THP1 (*P < 0.05 vs. GES-1-Exo group). (B, C) The expression of miR-541-5p in THP1 (Mφ) cells treated with exosomes. (D, E) The expression of M2 markers CD163, TGF-β, IL-10 and Arg1 in THP1 (Mφ) cells treated with exosomes. (F, G) The percentage of CD11b+CD206+ cells in THP1 (Mφ) cells (*P < 0.05 vs. SGC-7901-Exo or HGC-27-Exo group)
MiR-541-5p promoted polarization of M2 macrophages through the DUSP3-JAK2/STAT3 pathway
To gain a deeper understanding of how miR-541-5p affects M2 macrophage polarization, the TargetScan website was used to identify the target gene of miR-541-5p, and it was discovered that miR-541-5p had binding sites in the 3’-UTR region of DUSP3 mRNA (Fig. 5A). The binding of miR-541-5p to DUSP3 was confirmed through a dual luciferase assay. The transfection of miR-541-5p mimic resulted in a significant decrease in luciferase activity for the DUSP3- WT group, while the DUSP3-MUT group showed no substantial alteration (Fig. 5B). The miR-541-5p mimic caused a reduction in DUSP3 mRNA, whereas miR-541-5p inhibitor caused an increase in DUSP3 mRNA (Fig. 5C). Additionally, overexpression of DUSP3 attenuated SGC-7901 (miR-541-5p mimic)-Exo inhibition of DUSP3 expression, suggesting that the GC cell-derived exosomal miR-541-5p can target DUSP3 in macrophages (Fig. 5D and E). The JAK2/STAT3 signaling pathway regulates cell proliferation differentiation and is associated with the polarization of M2 macrophages [14, 15]. DUSP3 is a phosphatase that dephosphorylates substrate proteins [16]. Therefore, the effect of DUSP3 on the JAK2/STAT3 pathway was further examined. The results showed that overexpression of DUSP3 decreased the phosphorylation of JAK2 and STAT3 (Fig. 5F). Furthermore, overexpression of DUSP3 attenuated the induction of M2 macrophage polarization by exosome miR-541-5p (Fig. 5G and H). Thus, miR-541-5p may promote polarization of M2 macrophages by inhibiting DUSP3 to maintain JAK2/STAT3 signaling activation.
miR-541-5p promoted polarization of M2 macrophages through the DUSP3-JAK2/STAT3 pathway. (A) The binding site of miR-541-5p to DUSP3 was predicted by the TargetScan website. (B) Relative luciferase activity in THP1 (Mφ) cells after transfected with miR-541-5p mimic (*P < 0.05 vs. mimic-NC group). (C) The mRNA level of DUSP3 in THP1 (Mφ) cells (*P < 0.05 vs. mimic-NC group, #P < 0.05 vs. inhibitor-NC group). (D-F) The protein expression of DUSP3, p-JAK2, JAK2, p-STAT3 and STAT3 in THP1 (Mφ) cells. (G) The expression of M2 markers CD163 and Arg1 in THP1 (Mφ) cells. (H) The percentage of CD11b+CD206+ cells in THP1 (Mφ) cells (*P < 0.05 vs. SGC-7901 (mimic-NC)-Exo group, #P < 0.05 vs. ov-NC group, &P < 0.05 vs. SGC-7901 (miR-541-5p mimic)-Exo group)
The suppression of miR-541-5p caused a decrease in tumor growth
Tumorigenesis assays were performed in nude mice to investigate the influence of miR-541-5p on GC development. The results showed that tumor weight was significantly reduced in miR-541-5p inhibitor group (Fig. 6A), and miR-541-5p expression was decreased (Fig. 6B). The inhibition of miR-541-5p resulted in increased DUSP3 expression and decreased phosphorylation levels of JAK2 and STAT3 in miR-541-5p inhibitor group (Fig. 6C and D). What’s more, the suppression of miR-541-5p decreased the levels of the M2 macrophage marker CD163 protein, as well as the levels of the anti-inflammatory factors TGF-β and IL-10 in tumor tissues (Fig. 6E and F). The findings imply that suppressing miR-541-5p may impede GC progression in vivo.
Inhibition of miR-541-5p slowed tumor growth in vivo. (A) Tumor weight. (B) The expression of miR-541-5p in tumor tissues. (C, D) The protein expression of DUSP3, p-JAK2, JAK2, p-STAT3 and STAT3 in tumor tissues. (E) The protein level of CD163 in tumor tissues. (F) The TGF-β and IL-10 levels in tumor tissues were measured by ELISA kit (*P < 0.05 vs. inhibitor-NC group)
Discussion
The transfer of exosomes-mediated miRNAs to specific recipient cells via vectors contributes to tumor metastasis and drug resistance [17, 18]. The expression of abnormal miRNA can activate or deactivate tumor-related genes, thus aiding in the beginning, development, infiltration, and spread of tumors by controlling cell differentiation, apoptosis, and proliferation [19, 20]. In vitro and in vivo, miR-541 hinders the growth and turnover of HCC cells. miR-541p plays a role in cell growth, differentiation, cell death, and the cell cycle by controlling the transcription of specific genes. A limited number of investigations have indicated the involvement of miR-541-5p in cancer. Xu [21] et al. demonstrated a decrease in miR-541 expression in human HCC tissues with a shift in clinicopathological phenotype and autophagy in HCC. Tang [22] et al. showed that the uptake of miR-541-5p by circRTN4IP1 caused HIF1A to form and hindered the growth of intrahepatic cholangiocarcinoma cell malignancies. This research demonstrated that miR-541-5p expression was increased in gastric cancer tissues and cells, and we further corroborated that miR-541-5p stimulated the growth, movement, and infiltration of gastric cancer cells. The evidence implies that miR-541-5p has beneficial and detrimental effects on tumors and cancers.
DUSP3, also known as V vaccinia H1-R elated (VHR), is a crucial member of bispecific protein phosphatase, a small bispecific phosphatase. Earlier researches have indicated that DUSP3 plays a role in the cell cycle [23], differentiation [24], adhesion, and migration [25, 26] and possesses intricate and contradictory functions in tumorigenesis, potentially associated with cell type. Subsequent studies [27] revealed the regulatory function of DUSP3 interaction with target protein networks in various cancers. DUSP3 deletion reduces inflammatory response to septic shock by increasing the number and activity of M2 macrophages [28]. In this study, miR-541-5p binds to the 3 ‘-UTR regions of DUSP3 mRNA by predicting miR-541-5p’s target genes, and we validated miR-541-5p binding to DUSP3 by double-luciferase assays. However, overexpression of DUSP3 attenuated the inhibition of DUSP3 by SGC-7901-Exo, suggesting that miR-541-5p derived from gastric cancer cell exosomes can target and regulate DUSP3 expression in macrophages.
The JAK2/STAT3 signaling pathway is critical for regulating cell growth and differentiation and significantly contributes to the formation of tumors, drug resistance, and the progression of malignancies. Research has demonstrated that decreasing the phosphorylation of JAK2 and STAT can obstruct the growth of diffuse large B-cell lymphoma in mice, amplifying the antitumor effects of vincristine and prednisone [29] and induce apoptosis in breast cancer cells by stimulating the production of reactive oxygen species [30], as well as preventing the emergence of chemotherapy resistance in breast cancer during paclitaxel treatment by controlling the expression of genes related to epithelial-mesenchymal transition [31]. The JAK2/STAT3 signaling pathway is responsible for regulating M2 macrophage polarization. Research has suggested that the JAK2/STAT3 signaling pathway alters the polarization of microglia/macrophages (particularly M2 polarization) during CIRI, which assists in lessening brain harm and contributes to the progression of Cucurbitacin B-induced M2-like macrophage inhibition [14]. DUSP3 has been shown to dephosphorylate MAPKs, ERKs, and JNKs, and other substrates have also been shown to be targets of DUSP3 in recent years. In non-small cell lung cancer, DUSP3 can dephosphorylate tyrosine Tyr, explicitly targeting the tyrosine kinase receptor (ErBb) and epidermal growth factor receptor (EGFR) [32]. (2) In immune cells stimulated by cytokines and growth factors, DUSP3 can dephosphorylate signal transducers and activators of transcription (STAT) [33]. This research revealed that an increase in DUSP3 expression caused a decrease in the phosphorylation of JAK2 and STAT3. Furthermore, the overexpression of DUSP3 reduced the stimulation of M2 macrophage polarization by exosomal miR-541-5p, implying that miR-541-5p may sustain JAK2/STAT3 signaling activation by restraining DUSP3, thus encouraging M2 macrophage polarization, which was further corroborated by in vivo experiments.
In summary, miR-541-5p was upregulated in gastric cancer tissues. Macrophages took up the exosomal miR-541-5p secreted by gastric cancer cells, and miR-541-5p promoted JAK2/STAT3 signaling activation by targeting DUSP3, thereby promoting macrophage M2 polarization. Our research indicates that miR-541-5p could be a viable target for treating gastric cancer.
Despite this, there are certain restrictions to this research. At the outset, there is a need for more evidence to back up the role of miR-541-5p in controlling tumor growth and M2 macrophage polarization in living organisms. Furthermore, there needs to be more pertinent clinical trials to corroborate the study’s novel discoveries. Consequently, in subsequent investigations, we will concentrate on these two queries to enhance the intrinsic process of miR-541-5p in controlling gastric cancer tumors and M2 macrophages.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Thrift AP, El-Serag HB. Burden of gastric Cancer. Clin Gastroenterol Hepatol. 2020;18:534–42.
Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396:635–48.
Patel TH, Cecchini M. Targeted therapies in Advanced Gastric Cancer. Curr Treat Options Oncol. 2020;21:70.
Necula L, Matei L, Dragu D, Neagu AI, Mambet C, Nedeianu S, et al. Recent advances in gastric cancer early diagnosis. World J Gastroenterol. 2019;25:2029–44.
Pan Y, Yu Y, Wang X, Zhang T. Tumor-Associated macrophages in Tumor Immunity. Front Immunol. 2020;11:583084.
Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Invest. 2012;122:787–95.
He C, Zheng S, Luo Y, Wang B. Exosome Theranostics: Biology and Translational Medicine. Theranostics. 2018;8:237–55.
Lu TX, Rothenberg ME. MicroRNA. J Allergy Clin Immunol. 2018;141:1202–07.
He B, Zhao Z, Cai Q, Zhang Y, Zhang P, Shi S, et al. miRNA-based biomarkers, therapies, and resistance in Cancer. Int J Biol Sci. 2020;16:2628–47.
Lee YS, Dutta A. MicroRNAs in cancer. Annu Rev Pathol. 2009;4:199–227.
Inoue J, Inazawa J. Cancer-associated miRNAs and their therapeutic potential. J Hum Genet. 2021;66:937–45.
Li D, Zhang J, Yang J, Wang J, Zhang R, Li J, et al. CircMTO1 suppresses hepatocellular carcinoma progression via the miR-541-5p/ZIC1 axis by regulating Wnt/β-catenin signaling pathway and epithelial-to-mesenchymal transition. Cell Death Dis. 2021;13:12.
Li W, Wang X, Li C, Chen T, Yang Q. Exosomal non-coding RNAs: emerging roles in bilateral communication between cancer cells and macrophages. Mol Ther. 2022;30:1036–53.
Zhang H, Zhao B, Wei H, Zeng H, Sheng D, Zhang Y. Cucurbitacin B controls M2 macrophage polarization to suppresses metastasis via targeting JAK-2/STAT3 signalling pathway in colorectal cancer. J Ethnopharmacol. 2022;287:114915.
Zhong Y, Gu L, Ye Y, Zhu H, Pu B, Wang J, et al. JAK2/STAT3 Axis intermediates Microglia/Macrophage polarization during cerebral Ischemia/Reperfusion Injury. Neuroscience. 2022;496:119–28.
Monteiro LF, Ferruzo PYM, Russo LC, Farias JO, Forti FL. DUSP3/VHR: a Druggable Dual Phosphatase for Human diseases. Rev Physiol Biochem Pharmacol. 2019;176:1–35.
Azmi AS, Bao B, Sarkar FH. Exosomes in cancer development, metastasis, and drug resistance: a comprehensive review. Cancer Metastasis Rev. 2013;32:623–42.
Sun Z, Shi K, Yang S, Liu J, Zhou Q, Wang G, et al. Effect of exosomal miRNA on cancer biology and clinical applications. Mol Cancer. 2018;17:147.
Jiang C, Chen X, Alattar M, Wei J, Liu H. MicroRNAs in tumorigenesis, metastasis, diagnosis and prognosis of gastric cancer. Cancer Gene Ther. 2015;22:291–301.
Li C, Zhang K, Chen J, Chen L, Wang R, Chu X. MicroRNAs as regulators and mediators of forkhead box transcription factors function in human cancers. Oncotarget. 2017;8:12433–50.
Xu WP, Liu JP, Feng JF, Zhu CP, Yang Y, Zhou WP, et al. miR-541 potentiates the response of human hepatocellular carcinoma to sorafenib treatment by inhibiting autophagy. Gut. 2020;69:1309–21.
Tang J, Wang R, Tang R, Gu P, Han J, Huang W. CircRTN4IP1 regulates the malignant progression of intrahepatic cholangiocarcinoma by sponging mir-541-5p to induce HIF1A production. Pathol Res Pract. 2022;230:153732.
Russo LC, Farias JO, Forti FL. DUSP3 maintains genomic stability and cell proliferation by modulating NER pathway and cell cycle regulatory proteins. Cell Cycle. 2020;19:1545–61.
Qiu W, Guo X, Li B, Wang J, Qi Y, Chen Z, et al. Exosomal miR-1246 from glioma patient body fluids drives the differentiation and activation of myeloid-derived suppressor cells. Mol Ther. 2021;29:3449–64.
Tanaka M, Abe S. Different activities of antitumor immunomodulators to induce neutrophil adherence response. Drug Discov Ther. 2019;13:299–305.
Chen YR, Chou HC, Yang CH, Chen HY, Liu YW, Lin TY, et al. Deficiency in VHR/DUSP3, a suppressor of focal adhesion kinase, reveals its role in regulating cell adhesion and migration. Oncogene. 2017;36:6509–17.
Patterson KI, Brummer T, O’Brien PM, Daly RJ. Dual-specificity phosphatases: critical regulators with diverse cellular targets. Biochem J. 2009;418:475–89.
Singh P, Dejager L, Amand M, Theatre E, Vandereyken M, Zurashvili T, et al. DUSP3 genetic deletion confers M2-like macrophage-dependent tolerance to septic shock. J Immunol. 2015;194:4951–62.
Dong L, Huang J, Gao X, Du J, Wang Y, Zhao L. CircPCBP2 promotes the stemness and chemoresistance of DLBCL via targeting miR-33a/b to disinhibit PD-L1. Cancer Sci. 2022;113:2888–903.
Shakya R, Park GH, Joo SH, Shim JH, Choi JS. Hydroxyzine induces cell death in Triple-negative breast Cancer cells via mitochondrial superoxide and modulation of Jak2/STAT3 signaling. Biomol Ther (Seoul). 2022;30:585–92.
Stevens LE, Peluffo G, Qiu X, Temko D, Fassl A, Li Z, et al. JAK-STAT signaling in inflammatory breast Cancer enables chemotherapy-resistant Cell States. Cancer Res. 2023;83:264–84.
Wang JY, Yeh CL, Chou HC, Yang CH, Fu YN, Chen YT, et al. Vaccinia H1-related phosphatase is a phosphatase of ErbB receptors and is down-regulated in non-small cell lung cancer. J Biol Chem. 2011;286:10177–84.
Kim BR, Ha J, Kang E, Cho S. Regulation of signal transducer and activator of transcription 3 activation by dual-specificity phosphatase 3. BMB Rep. 2020;53:335–40.
Acknowledgements
Not applicable.
Funding
This work was supported by Shaanxi Province Technology Innovation Guidance Special Project-Regional Innovation Capability Guidance Program [grant numbers: 2022QFY01-08].
Author information
Authors and Affiliations
Contributions
Jinyan Yuan contributed to the study conception and design. Jia Fu and Ruiting Liu contributed to the Material preparation and Data collection. Likun Yan and Zheqi Zhou contributed to Data analysis and Software. The first draft of the manuscript was written by Haimin Xiao. All authors commented on previous versions of the manuscript, read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was performed in line with the principles of the Declaration of Helsinki and approval was granted by the Ethics Committee of Shaanxi Provincial People’s Hospital. Written informed consent was obtained from individual or guardian participants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12885_2024_12672_MOESM2_ESM.tif
Supplementary Material 2: Fig. S1: GC cells facilitated miR-541-5p expression in THP1 (Mφ) cells by secreting the exosomal miR-541-5p. SGC-7901 cells were transfected with mimic-NC or miR-541-5p mimic, and HGC-27 cells were transfected with inhibitor-NC or miR-541-5p inhibitor; THP1 (Mφ) cells were cultured alone or co-cultured with un-transfected/transfected SGC-7901 and HGC-27 cells. The level of miR-541-5p was detected by RT-qPCR (A, B) in THP1 (Mφ) cells and in exosomes in the co-culture medium (C, D). *P < 0.05 vs NC group, #P < 0.05 vs SGC-7901 (mimic-NC) or HGC-27 (inhibitor-NC) group
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it.The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xiao, H., Fu, J., Liu, R. et al. Gastric cancer cell-derived exosomal miR-541-5p induces M2 macrophage polarization through DUSP3/JAK2/STAT3 pathway. BMC Cancer 24, 957 (2024). https://doi.org/10.1186/s12885-024-12672-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12885-024-12672-1