Abstract
Objective
The objective of this systematic review and meta-analysis was to assess the value of uric acid in predicting acute kidney injury caused by traumatic rhabdomyolysis.
Methods
The search was conducted in MEDLINE, Scopus, Embase and Web of Science until November 1, 2023. Based on the inclusion and exclusion criteria, the articles were included by two independent researchers. Data regarding study design, patient characteristics, number of patients with and without AKI, mean and SD of uric acid and prognostic characteristics of uric acid were extracted from relevant studies. STATA version 17.0 was used to compute pooled measures of standardized mean differences, odds ratios, and diagnostic accuracy. I2 and chi-square tests were used to assess heterogeneity between studies.
Results
We found 689 non-redundant studies, 44 of them were potentially relevant. Six articles met the inclusion criteria and were included in the review. The results of the meta-analysis confirmed that there was a significant correlation between serum uric acid levels and the occurrence of AKI (SMD = 1.61, 95% CI = 0.69 to 2.54, I2 = 96.94%; p value = 0.001). There were no significant publication biases.
Conclusion
According to this meta-analysis, uric acid levels could be considered as a predictor of acute kidney injury following traumatic rhabdomyolysis.
Similar content being viewed by others
Introduction
Rhabdomyolysis (RM) is a clinical syndrome distinguished by the disruption of structural integrity in skeletal muscle cells, leading to the release of intracellular constituents into the extracellular space. Trauma, immobilization, sepsis, and surgery are the most frequently reported causes of RM [1,2,3]. In the case of rhabdomyolysis, acute kidney injury (AKI) is a tragic yet preventable complication. Patients with RM are reported to have a 13–50% chance of developing AKI, which is associated with approximately 30% mortality increase [4]. Patient outcomes are worse in RM patients with AKI, which increases medical burden. Therefore, prevention and early diagnosis of AKI improve prognosis for these patients [5,6,7,8].
A severe physical injury or trauma can cause traumatic rhabdomyolysis, which is characterized by rapid breakdown of skeletal muscle tissue [9]. When there is significant muscle compression or crush injuries, such as those sustained in car accidents, crush syndrome, sever burns, compartment syndrome, or natural disasters, this condition may occur [10]. The symptoms of traumatic rhabdomyolysis include muscle pain, weakness, dark urine (due to myoglobinuria), and, in severe cases, renal dysfunction [11]. By hydration and other appropriate measures, and prompt medical intervention, the underlying trauma can be managed, complications prevented, and renal function supported [12]. A mass disaster such as an earthquake will result in a large number of traumatic rhabdomyolysis patients. Management of these patients will be challenging given the special circumstances following the disaster and the shortage of equipment and resources [13].
Furthermore, rhabdomyolysis can present with a variety of symptoms, from asymptomatic to multiple organ failure [14]. In both the treatment and logistic aspects of triaging at-risk patients, it is of utmost importance to identify potential effective factors of rhabdomyolysis-induced complications such as AKI. Several studies have examined clinical and laboratory factors and developed a limited number of clinical prediction rules [13,14,15].
Uric acid(UA) is produced by purine metabolism in the body. During high cell turnover states like hemolysis, rhabdomyolysis, and tumor lysis, uric acid levels can increase [16]. Increased serum uric acid levels have been shown to be associated with kidney disease. Despite uric acid’s intrinsic antioxidant properties, studies have found that increased levels of uric acid promote oxidative stress, inflammation, fibrosis pathways, and endothelial dysfunction [17,18,19,20]. Excessive uric acid increases the risk of acute kidney injury [21], impairs the contractile function of intraglomerular mesangial cells [22], and causes damage to mesangial and proximal tubule epithelial cells through Toll like receptor 4-dependent activation of NLRP3 and IL-1β [23]. Hyperuricemia was also shown to be an independent risk factor for chronic kidney disease via injury of the endothelial cells and release of the high mobility group box 1 protein (HMGB1), stimulating Toll-like receptors (TLR) to induce pro-inflammatory and chemotactic cytokines, vascular smooth muscle proliferation, and activation of the NLRP3 inflammasome [24]. Furthermore, uric acid may accumulate in the kidneys, resulting in stone formation [25].
Studies have reported that UA can act as a predictive factor in a number of diseases. Some of them suggest that in rhabdomyolysis patients, elevated UA levels may be associated with an increased risk of AKI. However, whether UA is a prognostic factor in rhabdomyolysis-induced AKI remains controversial [26,27,28,29,30].
Therefore, this study evaluated the value of uric acid in predicting traumatic rhabdomyolysis-induced AKI by meta-analyzing existing studies.
Method
Search strategy
The preferred reporting items for systematic reviews and meta-analysis (PRISMA) guideline was followed in the present study [31]. The keywords were selected using three strategies: MeSH terms, Emtree terms, and consultation with experts. We conducted a systematic search through PubMed/MEDLINE, Scopus, Web of Science (WOS) and Embase databases for relevant articles published since inception until November 1, 2023 (search strategy of all databases is reported in Supplementary Table 1).
Scopus was used to identify additional documents in the gray literature, such as conference abstracts. A hand search of the reference lists of relevant studies was conducted to find additional articles and unpublished data.
Our systematic review question was formulated as follows [32]: The population (P) were patients with traumatic-rhabdomyolysis, the index test (I) was serum uric acid level(mg/dl), the reference test (R) was any valid definition of AKI using serum creatinine level, glomerular filtration rate (GFR), Kidney Disease Improving Global Outcomes (KDIGO) guidelines or risk, injury, failure, loss of kidney function, and end-stage kidney disease (RIFLE) criteria.
A protocol for this study is registered in the International Prospective Register of Systematic Reviews (PROSPERO) with the identifier CRD42023460189.
Eligibility criteria and screening method
The inclusion criteria were as follows: traumatic-rhabdomyolysis patients as a population; measurement of serum uric acid level; prediction of AKI incidence using GFR, serum creatinine level, KDIGO or RIFLE criteria; and having a control group (without AKI). Analyses were conducted both retrospectively and prospectively. We excluded studies without a control group (non-AKI patients), in non-traumatic patients, without assessments of serum uric acid, animal studies, case reports, case series, reviews, systematic reviews, letters to editors, editorials and protocols. There was no restriction on the participants’ age. No language restriction is considered. The screening and selection process was carried out independently by two authors (M.G. and A.G). Disagreements were resolved either through discussion or by expert reviewers (M.Y. and S.S).
Data extraction and risk of bias assessment
Both authors (M.G and A.G.) independently extracted data, including the name of the first author, the publication year, the study design, the country, the age of the population, the definition of AKI, the total sample size, the number of participants with AKI and those without, and the mean and standard deviation (SD) of uric acid levels in AKI and non-AKI patients. The data extraction sheet is summarized in Table 1. Whenever data in an article could not be extracted, its corresponding author was contacted. Results presented as graphs were extracted using web plot digitizer [33].
The risk of bias was assessed by the two authors (M.G. and A.G) independently, using the QUADAS-2 checklist [34, 35]. The differences between the two were resolved through discussion or by expert reviewers (M.Y. and S.S).
The QUADAS-2 includes two sections: The risk of bias (ROB) and applicability. The risk of bias section includes four domains, such as patient selection, index test, reference standard and flow and timing. There are three domains in the applicability section, including patient selection, index test, and reference standard. All domains are judged as “low” for answer yes, “high” for answer no, or unclear. Then, studies were classified according to the following categories: low risk of bias and low concern regarding applicability (when all domains were low risk) and at risk of bias or as having concerns regarding applicability (when at least one domain was high risk or unclear).
Statistical analysis
The statistical analyses were conducted using Stata 17.0 (StataCorp LLC, College Station, TX, USA). We calculated a standardized mean difference (SMD) with a 95% confidence interval (95% CI) for each sample, and then pooled them to calculate an overall effect size. At least three separate analyses had to be reported for meta-analysis to be performed.
To compensate for methodological heterogeneities between studies, such as differences in population and methodology, a random-effects model was chosen. The I2 and chi-square tests were used to assess statistical heterogeneity between studies.
Results
Study characteristics
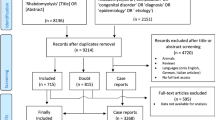
The study flowchart is shown in Fig. 1. After removing duplicates from 1441 publications, 689 titles and abstracts were screened, and 44 potentially relevant articles were selected for full-text evaluation based on the inclusion and exclusion criteria.
Ultimately, six observational studies published between 2007 and 2021 were eligible for inclusion. Through forwarding and backward citation tracking of eligible studies, no additional articles were found. Table 1 summarizes the main characteristics of the included studies.
The studies were all cross-sectional. Four studies were conducted in Asia and two in Europe. In all studies, both sexes were included. The etiology of rhabdomyolysis was trauma in all studies. Three of the studies involved earthquake victims. AKI was defined in one study using RIFLE criteria, one using KIDGO criteria, two studies using creatinine values greater than 1.6 mg/dL in the first three days after admission, and in one study AKI definition was not mentioned.
In all six studies, mean serum uric acid levels were compared between participants with and without AKI [13, 15, 29, 30, 36, 37]. One study presents its findings as an OR, two studies present their findings as ROC curves [13, 15, 30]. There was no presentation of sensitivity or specificity in any of the studies. The meta-analyses were therefore limited to standardized mean differences.
Comparing serum uric acid levels of patients with and without AKI
In these studies, 526 AKI + cases and 2404 AKI- cases were included. The average age of these patients was 46.03 ± 19.37 years, and 68.8% of them were male.
Comparing pooled standardized mean differences (SMD) showed a significant correlation between serum uric acid level and AKI occurrence in traumatic rhabdomyolysis patients (SMD = 1.61, 95% CI: 0.69 to 2.54, I2 = 96.94%; p value = 0.001). the mean and standard deviation of uric acid level in AKI and non-AKI group was 7.03 ± 2.12 and 4.25 ± 1.11 respectively. The result of the meta-analysis is presented in Fig. 2.
Risk of bias assessment
Based on QUADAS-2, five studies were categorized as “high” and one as “low” risk of bias (Table 2). Two studies included did not use random selection for participants [36, 38]. Reference standard appropriate for correctly classifying target conditions in all studies. As well, index test results were interpreted without knowing the reference standard results.
Publication bias
Both funnel plots and Eggers tests were used to assess publication bias (Fig. 3). There was an assymetria in funnel plot, however, the egger test did not detect any significant publication bias (p = 0.2766).
Discussion
This meta-analysis was the first to examine the association between serum uric acid levels and acute kidney injury following traumatic rhabdomyolysis. Our results showed a significant correlation between serum uric acid levels and AKI occurrence. This suggests that uric acid levels may be a potential biomarker for predicting AKI following traumatic rhabdomyolysis. In order to identify the proper cutoff point, further research is needed. Furthermore, it is crucial for researchers to report sensitivity, specificity, OR, and AUC in order to facilitate meta-analyses of these items.
Acute kidney injury (AKI) is an important complication of RM, which is associated with high morbidity and mortality. Through early and aggressive fluid therapy, AKI can be prevented in this condition. Even so, logistics supplies are often insufficient for fluid therapy in disaster situations. To manage AKI successfully, patients at risk must be distinguished from those not at risk [39].
Items that can help predict or identify patients at risk for AKI, or rules of thumb that can identify those who are at risk, would be very helpful. To make these items easy to use in emergency situations, simple clinical or biochemical parameters should be used. Several laboratory values have already been tested to identify a possible association with AKI [40,41,42].
When interpreting our results, it is important to keep the following in mind:
An important limitation of this study is the meta-analysis of observational studies. Observational designs are limited in nature and cannot assess all confounding factors, and therefore cannot prove causal relationships effectively.
Second, a crude SMD was used in this study due to insufficient data regarding adjusted effect sizes. Hence, various uncontrolled factors such as age, sex, comorbidities, duration of trauma to admission and unknown factors may confound the observed positive relationship between uric acid and AKI.
Third, we were unable to conduct meta-analyses on OR, sensitivity, specificity, and AUC due to insufficient data.
Although efforts were made to make the studies similar from a methodology perspective, there was heterogeneity between the studies. There was some variation between articles regarding the definition of AKI and the cut-point for creatin kinase for RM. In addition, Yoo et al. study was conducted on children (aged one month to 18 years) [30].
In addition, given the low number of eligible studies, subgroup analysis was not possible in terms of methodology.
Regarding the asymmetry in our funnel plots, Egger et al. have mentioned that asymmetry in funnel plots can be caused by several factors: publication bias, poor methodological quality, the presence of substantial heterogeneity, selective outcome reporting, sampling variations, and by chance. Consequently, publication bias might not be the only reason for the observed asymmetry [43].
In this meta-analysis, all six studies were in some way heterogeneous, so it was difficult to draw solid conclusions. This could be due to the limited relevant studies on this topic. Additionally, most of the studies didn’t report OR, sensitivity, specificity, and AUC, so meta-analysis was not possible on these items. Thus, to help reach a conclusive conclusion regarding uric acid’s ability to predict AKI caused by traumatic-rhabdomyolysis, more comprehensive and original research is needed.
Conclusion
This meta-analysis shows that uric acid levels could be considered as a predictor of AKI following traumatic rhabdomyolysis. The results of these studies provide evidence for other researchers and clinicians to pay closer attention to this item in patients with traumatic rhabdomyolysis in order to prevent AKI.
Data availability
No datasets were generated or analysed during the current study.
References
Kodadek L, Carmichael Ii SP, Seshadri A, Pathak A, Hoth J, Appelbaum R et al. Rhabdomyolysis: an American Association for the Surgery of Trauma Critical Care Committee Clinical Consensus Document. Trauma Surg acute care open [Internet]. 2022;7(1):e000836. Available from: http://www.ncbi.nlm.nih.gov/pubmed/35136842.
Chavez LO, Leon M, Einav S, Varon J. Beyond muscle destruction: a systematic review of rhabdomyolysis for clinical practice. Crit Care [Internet]. 2016;20(1):135. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27301374.
Long B, Koyfman A, Gottlieb M. An evidence-based narrative review of the emergency department evaluation and management of rhabdomyolysis. Am J Emerg Med [Internet]. 2019;37(3):518–23. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0735675718310398.
Bosch X, Poch E, Grau JM. Rhabdomyolysis and Acute Kidney Injury. N Engl J Med [Internet]. 2009;361(1):62–72. Available from: http://www.nejm.org/doi/abs/https://doi.org/10.1056/NEJMra0801327.
Yang J, Zhou J, Wang X, Wang S, Tang Y, Yang L. Risk factors for severe acute kidney injury among patients with rhabdomyolysis. BMC Nephrol [Internet]. 2020;21(1):498. Available from: https://bmcnephrol.biomedcentral.com/articles/https://doi.org/10.1186/s12882-020-02104-0.
Candela N, Silva S, Georges B, Cartery C, Robert T, Moussi-Frances J et al. Short- and long-term renal outcomes following severe rhabdomyolysis: a French multicenter retrospective study of 387 patients. Ann Intensive Care [Internet]. 2020;10(1):27. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32124091.
Harrois A, Soyer B, Gauss T, Hamada S, Raux M, Duranteau J et al. Prevalence and risk factors for acute kidney injury among trauma patients: a multicenter cohort study. Crit Care [Internet]. 2018;22(1):344. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30563549.
Rodríguez E, Soler MJ, Rap O, Barrios C, Orfila MA, Pascual J. Risk factors for acute kidney injury in severe rhabdomyolysis. PLoS One [Internet]. 2013;8(12):e82992. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24367578.
Chan JL, Imai T, Barmparas G, Lee JB, Lamb AW, Melo N et al. Rhabdomyolysis in obese trauma patients. Am Surg [Internet]. 2014;80(10):1012–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25264650.
Fernandez JJ, Smith SR. Traumatic Rhabdomyolysis: Crush Syndrome, Compartment Syndrome, and the ‘Found Down’ Patient. J Am Acad Orthop Surg [Internet]. 2023; Available from: https://journals.lww.com/https://doi.org/10.5435/JAAOS-D-23-00734.
Nance JR, Mammen AL. Diagnostic evaluation of rhabdomyolysis. Muscle Nerve [Internet]. 2015;51(6):793–810. Available from: https://onlinelibrary.wiley.com/doi/10.1002/mus.24606.
Boparai S, Lakra R, Dhaliwal L, Hansra RS, Bhuiyan MAN, Conrad SA et al. Therapeutic Plasma Exchange in Severe Rhabdomyolysis: A Case-Control Study. Cureus [Internet]. 2023; Available from: https://www.cureus.com/articles/131545-therapeutic-plasma-exchange-in-severe-rhabdomyolysis-a-case-control-study.
Omrani H, Naja I, Bahrami K, Naja F, Safari S. American Journal of Emergency Medicine Acute kidney injury following traumatic rhabdomyolysis in Kermanshah earthquake victims; A cross-sectional study. 2020;(xxxx):6–11.
Sun K, Shi Z, Abudureheman Y, Liu Q, Zhao Y, Zhang X et al. Clinical and Epidemiological Characteristics of Rhabdomyolysis: A Retrospective Study. Seixas A, editor. Int J Clin Pract [Internet]. 2023;2023:1–11. Available from: https://www.hindawi.com/journals/ijclp/2023/6396576/.
Najafi I, Van Biesen W, Sharifi A, Hoseini M, Rashid Farokhi F, Sanadgol H et al. Early detection of patients at high risk for acute kidney injury during disasters: development of a scoring system based on the Bam earthquake experience. J Nephrol [Internet]. 2008;21(5):776–82. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18949734.
George C, Leslie SW, Minter DA. Hyperuricemia [Internet]. StatPearls. 2024. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31688126.
So A, Thorens B. Uric acid transport and disease. J Clin Invest [Internet]. 2010;120(6):1791–9. Available from: http://www.jci.org/articles/view/42344.
Wang H, Qiu F, Liu J, Luo C, Liu X. Elevated serum uric acid is associated with renal arteriolopathy and predict poor outcome in patients with lupus nephritis. Clin Exp Rheumatol [Internet]. 2023; Available from: https://www.clinexprheumatol.org/abstract.asp?a=19572.
Bardin T, Richette P. Definition of hyperuricemia and gouty conditions. Curr Opin Rheumatol [Internet]. 2014;26(2):186–91. Available from: https://journals.lww.com/00002281-201403000-00014.
Kimura Y, Tsukui D, Kono H. Uric Acid in Inflammation and the Pathogenesis of Atherosclerosis. Int J Mol Sci [Internet]. 2021;22(22):12394. Available from: https://www.mdpi.com/1422-0067/22/22/12394.
Xu X, Hu J, Song N, Chen R, Zhang T, Ding X. Hyperuricemia increases the risk of acute kidney injury: a systematic review and meta-analysis. BMC Nephrol [Internet]. 2017;18(1):27. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28095822.
Convento MS, Pessoa E, Dalboni MA, Borges FT, Schor N. Pro-inflammatory and oxidative effects of noncrystalline uric acid in human mesangial cells: contribution to hyperuricemic glomerular damage. Urol Res [Internet]. 2011;39(1):21–7. Available from: http://link.springer.com/https://doi.org/10.1007/s00240-010-0282-5.
Xiao J, Fu C, Zhang X, Zhu D, Chen W, Lu Y et al. Soluble monosodium urate, but not its crystal, induces toll like receptor 4-dependent immune activation in renal mesangial cells. Mol Immunol [Internet]. 2015;66(2):310–8. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0161589015003545.
Kim S-M, Lee S-H, Kim Y-G, Kim S-Y, Seo J-W, Choi Y-W et al. Hyperuricemia-induced NLRP3 activation of macrophages contributes to the progression of diabetic nephropathy. Am J Physiol Physiol [Internet]. 2015;308(9):F993–1003. Available from: https://www.physiology.org/doi/https://doi.org/10.1152/ajprenal.00637.2014.
Jalal DI. Hyperuricemia, the kidneys, and the spectrum of associated diseases: a narrative review. Curr Med Res Opin [Internet]. 2016;32(11):1863–9. Available from: https://www.tandfonline.com/doi/full/https://doi.org/10.1080/03007995.2016.1218840.
Li L, Tian X, Gao Y, Gu Y, Zhang K, Li L et al. Serum uric acid predicts the development of atherosclerosis in women but not in men: A ten-year cohort study in China. Nutr Metab Cardiovasc Dis [Internet]. 2023; Available from: https://linkinghub.elsevier.com/retrieve/pii/S0939475323004088.
Qin A, Yang D, Wang S, Dong L, Tan J, Tang Y et al. Uric acid-based ratios for predicting renal failure in Chinese IgA nephropathy patients. Int J Med Sci [Internet]. 2023;20(12):1584–91. Available from: https://www.medsci.org/v20p1584.htm.
Yan W, Tang H-Y, Yang Y-Q, He K-L. Serum uric acid and outcome in hospitalized elderly patients with chronic heart failure through the whole spectrum of ejection fraction phenotypes. BMC Cardiovasc Disord [Internet]. 2023;23(1):589. Available from: https://bmccardiovascdisord.biomedcentral.com/articles/https://doi.org/10.1186/s12872-023-03544-w.
Sever MS, Kellum J, Hoste E, Vanholder R. Application of the RIFLE criteria in patients with crush-related acute kidney injury after mass disasters. 2010;1370–6.
Yoo S, Cho MH, Baek HS, Song JY, Lee HS, Yang EM et al. Characteristics of pediatric rhabdomyolysis and the associated risk factors for acute kidney injury: a retrospective multicenter study in Korea. 2021;40(4):673–86.
Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ [Internet]. 2021;n160. Available from: https://www.bmj.com/lookup/doi/https://doi.org/10.1136/bmj.n160.
Pourahmadi M, Delavari S, Koes B, Keshtkar A, Nazemipour M, Mansournia MA. How to formulate appropriate review questions for systematic reviews in sports medicine and rehabilitation? Br J Sports Med [Internet]. 2021;55(22):1246–7. Available from: https://bjsm.bmj.com/lookup/doi/https://doi.org/10.1136/bjsports-2021-104315.
Ankit Rohatgi. WebPlotDigitizer [Internet]. Pacifica, California, USA; Available from: https://automeris.io/WebPlotDigitizer.
Whiting PF. QUADAS-2: A Revised Tool for the Quality Assessment of Diagnostic Accuracy Studies. Ann Intern Med [Internet]. 2011;155(8):529. Available from: http://annals.org/article.aspx?doi=10.7326/0003-4819-155-8-201110180-00009.
Whiting P, Rutjes AW, Reitsma JB, Bossuyt PM, Kleijnen J. The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC Med Res Methodol [Internet]. 2003;3(1):25. Available from: http://bmcmedresmethodol.biomedcentral.com/articles/https://doi.org/10.1186/1471-2288-3-25.
Mediterranea AM, Chemistry B, Chemistry M, Section MB-, Azzolina R. Clinicalstudyand laboratory outcomes on rhabdomyolysis in posttrauma- tic renal damage. 2007;69–71.
Galeiras R, Mourelo M, Pértega S, Lista A, Ferreiro ME, Salvador S et al. Rhabdomyolysis and acute kidney injury in patients with traumatic spinal cord injury. 2016;(16):504–12.
Sever MS, Kellum J, Hoste E, Vanholder R. Application of the RIFLE criteria in patients with crush-related acute kidney injury after mass disasters. Nephrol Dial Transplant [Internet]. 2011;26(2):515–24. Available from: https://academic.oup.com/ndt/article-lookup/doi/https://doi.org/10.1093/ndt/gfq426.
Abu-Zidan F, Idris K, Cevik A. Prehospital management of earthquake crush injuries: A collective review. Turkish J Emerg Med [Internet]. 2023;23(4):199. Available from: https://journals.lww.com/https://doi.org/10.4103/tjem.tjem_201_23.
Rodríguez-Capote K, Balion CM, Hill SA, Cleve R, Yang L, El Sharif A. Utility of Urine Myoglobin for the Prediction of Acute Renal Failure in Patients with Suspected Rhabdomyolysis: A Systematic Review. Clin Chem [Internet]. 2009;55(12):2190–7. Available from: https://academic.oup.com/clinchem/article/55/12/2190/5629443.
Fernandez WG, Hung O, Bruno GR, Galea S, Chiang WK. Factors predictive of acute renal failure and need for hemodialysis among ED patients with rhabdomyolysis. Am J Emerg Med [Internet]. 2005;23(1):1–7. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0735675704002633.
Muckart DJJ, Moodley M, Naidu AG, Reddy ADR, Meineke KR, Prediction of acute renal failure following soft-tissue injury, using the venous bicarbonate concentration. J Trauma Inj Infect Crit Care [Internet]. 1992;33(6):813–7. Available from: http://journals.lww.com/00005373-199212000-00003.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ [Internet]. 1997;315(7109):629–34. Available from: https://www.bmj.com/lookup/doi/https://doi.org/10.1136/bmj.315.7109.629.
Acknowledgements
The author acknowledges Men’s Health and Reproductive Health Research Center, Shahid Beheshti University of Medical Sciences for their support, which have been invaluable in the completion of this research.
Funding
There are no funds allocated for our project.
Author information
Authors and Affiliations
Contributions
Conceptualization: SS, MH, IN; Data curation: MG, AG, SS, MH; Formal analysis: MH; Methodology: SS, MH, IN, MG, AG; Project administration: SS, MH, IN; Visualization: MH, MG; Writing - original draft: MG, AG, SS, MH, IN; Writing - review & editing: MG, AG, SS, MH, IN. All authors read and approved the final version of manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
There are no ethical considerations associated with this review. Ethical approval is not required because this protocol does not involve any individuals directly.
Consent for publication
N/A.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Safari, S., Ghasemi, M., Yousefifard, M. et al. Uric acid in predicting the traumatic rhabdomyolysis induced acute kidney injury; a systematic review and meta-analysis. BMC Nephrol 25, 82 (2024). https://doi.org/10.1186/s12882-024-03509-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12882-024-03509-x