Abstract
Background
Transcription factors, including trihelix transcription factors, play vital roles in various growth and developmental processes and in abiotic stress responses in plants. The trihelix gene has been systematically studied in some dicots and monocots, including Arabidopsis, tomato, chrysanthemum, soybean, wheat, corn, rice, and buckwheat. However, there are no related studies on sorghum.
Results
In this study, a total of 40 sorghum trihelix (SbTH) genes were identified based on the sorghum genome, among which 34 were located in the nucleus, 5 in the chloroplast, 1 (SbTH38) in the cytoplasm, and 1 (SbTH23) in the extracellular membrane. Phylogenetic analysis of the SbTH genes and Arabidopsis and rice trihelix genes indicated that the genes were clustered into seven subfamilies: SIP1, GTγ, GT1, GT2, SH4, GTSb8, and orphan genes. The SbTH genes were located in nine chromosomes and none on chromosome 10. One pair of tandem duplication gene and seven pairs of segmental duplication genes were identified in the SbTH gene family. By qPCR, the expression of 14 SbTH members in different plant tissues and in plants exposed to six abiotic stresses at the seedling stage were quantified. Except for the leaves in which the genes were upregulated after only 2 h exposure to high temperature, the 12 SbTH genes were significantly upregulated in the stems of sorghum seedlings after 24 h under the other abiotic stress conditions. Among the selected genes, SbTH10/37/39 were significantly upregulated, whereas SbTH32 was significantly downregulated under different stress conditions.
Conclusions
In this study, we identified 40 trihelix genes in sorghum and found that gene duplication was the main force driving trihelix gene evolution in sorghum. The findings of our study serve as a basis for further investigation of the functions of SbTH genes and providing candidate genes for stress-resistant sorghum breeding programmes and increasing sorghum yield.
Similar content being viewed by others
Background
Abiotic stress can affect the growth process of plants considerably, reducing plant development and crop yield [1]. In view of this, plants have evolved a complex system to regulate their adaptability to stress signals [2, 3]. Transcription factors are ubiquitous in plants and play important roles in various growth and developmental processes and abiotic stress response [4]. More than 60 transcription factor families have been identified in plants [5, 6]. Nevertheless, the functions of several crucial transcription factor families have not been completely clarified. In the 1980s, the trihelix transcription factors exist only in plants and separated from the pea (Pisum sativum) for the first time [7]. They bind to the core sequence of 5 ‘-G-Pu- (T / A) -A- (T / A) -3 ‘of the promoter region of rbcS-3A gene to regulate light-dependent expression [8]. Trihelix transcription factors were initially called GT factors because they bind to photosensitive GT elements. The DNA-binding domain of GT factor has a typical helix-loop-helix-loop-helix structure, which is responsible for the name trihelix transcription factor. Studies have shown that the trihelix structure of GT factors is highly similar to the structure of Myb/SANT-LIKE DNA-binding domains [9]. GT factors evolved from Myb/SANT-LIKE proteins. The gaps between helix pairs result in different recognition sequences between GT factors and Myb/SANT-LIKE proteins [9, 10].
The trihelix gene has been systematically studied in some dicots and monocots, including Arabidopsis, tomato, chrysanthemum, soybean, wheat, corn, rice, and buckwheat. However, the trihelix family in sorghum have not been systematically studied. Because of the important functions of trihelix genes in tissue development, environmental adaptation and evolution, it is of great significance to systematically analyze the trihelix family members of sorghum. Presently, a total of 30 GT family members have been identified in Arabidopsis thaliana and were classified into GT-1, GT-2, GTγ, SH4, and SIP1 subfamilies, named after their founding members [11]. Similarly, 96 trihelix proteins have been identified in tomato and were classified into six subfamilies (GT-1, GT-2, SH4, SIP1, GTγ, and GTδ) [12]. The structures of most trihelix genes vary among plant species, especially at the C-terminal.
Some studies have reported the involvement of trihelix gene family in complex physiological functions. In Arabidopsis, GT1 subfamily genes may be involved in salt stress and pathogen infection response, and their expression was induced by light in 3-days-old seedlings [13]. Additionally, expression of RML1 of tomato GT-1 gene was inhibited by light in yellow seedlings [14]. Osmotic, salt, and cold stress induced the expression of trihelix transcription factors, GmGT-2A and GmGT-2B, in soybeans [15]. In Arabidopsis thaliana, GTL1 gene mutants can significantly reduce transpiration and improve drought tolerance [16].. The expression of GTγ evolution branch gene, OsGTγ-1, in tomato increased by 2.5 to 10 folds in response to salt stress, and the abscisic acid (ABA) treatment also upregulated its expression [17]. The Arabidopsis SIP1 genes, ASIL1 and ASIL2, downregulated the expression of LEA (rich in late embryogenesis) gene in Arabidopsis seedlings [11]. Trihelix genes play multiple functions during plant development. Therefore, it is necessary to clarify their roles and the molecular mechanisms involved in signal transduction pathways in different stress response.
Sorghum (Sorghum bicolor L. Moench) is an important food crop and is widely cultivated in different regions of the world, making it an ideal C4 plant for research. In this study, the trihelix gene family was identified in sorghum. The chromosomal distributions, protein characteristics, gene structures, and conserved motif compositions of the identified trihelix genes were analysed. We then identified orthology relations, analysed gene duplication events, and constructed phylogenetic trees of the identified trihelix genes. Additionally, we examined the expression pattern of selected sorghum trihelix genes under abiotic stresses.
Results
Identification of trihelix genes and analysis of their physicochemical properties in S. bicolor (L.)
The Hidden Markov Model (HMM) profile of trihelix domain (PF13837) was used to search the trihelix domain in the entire sorghum genome. Only genes with E value < 0.01 were classified as those of trihelix family. The Pfam and InterPro databases were used to confirm that the putative genes contained the Myb/SANT-LIKE domain. Finally, a total of 40 non-redundant trihelix genes were identified in sorghum. The sorghum trihelix genes were named from Sbtrihelix1-Sbtrihelix40 according to their positions on the chromosome. Sbtrihelix was abbreviated to SbTH. Table S1 contains a summary of the characteristics of SbTH, including gene ID, chromosome location, coding sequence (CDS) length and amino acid sequence, protein size, and isoelectric point (PI). SbTH5 encodes the smallest protein with 205 amino acids, whereas SbTH18 encodes the largest protein with 875 amino acids. The SbTH protein molecular weight (Mw) ranged from 22.68 kDa–96.29 kDa, while the predicted isoelectric point ranged from 4.42 (SbTH17) to 11.19 (SbTH31). The results of subcellular localization prediction of SbTH proteins showed that 34 SbTH genes were located in the nucleus, 5 in the chloroplast, and 1 (SbTH23) in the extracellular membrane. Among the 40 SbTH genes, 8 (20.0%) contained the GT1 domain, 23 (57.5%) contained the Myb_DNA-binding domain and 9 (22.5%) contained both GT1 domain and Myb DNA-binding domain. The ratio of SbTH genes to total genes in the S. bicolor genome was about 0.12% [18], which is similar to that of Arabidopsis (0.11%) [19, 20], soybean (0.14%) [21, 22], and rice (0.10%) [23, 24] but more that than of tomato (0.05%) [12, 25], chrysanthemum (0.04%) [26, 27], wheat (0.08%) [28, 29], and buckwheat (0.06%) [30, 31].
Phylogenetic analysis of trihelix genes in S. bicolor (L.)
To better understand the phylogenetic relationship of trihelix genes, we constructed a phylogenetic tree using the neighbour-joining (NJ) method with a bootstrap value of 1000 based on the amino acid sequences of 40 SbTH proteins, 27 Arabidopsis thaliana trihelix (AtTH), and 29 Oryza sativa trihelix (OsTH) proteins (Fig. 1, Additional file 1: Table S1). According to the topological structure of the tree and classification method proposed by Kaplan-Levy and Qin [32, 33], the 40 trihelix genes were clustered into six groups (SIP1, GTγ, GT1, GT2, SH4, GTSb8) and three ‘orphan genes’. Among the 40 trihelix genes, 32 SbTH genes were clustered into five subfamilies, which was consistent with the results of Arabidopsis and rice. Five genes (SbTH8, SbTH10, SbTH16, SbTH25, and SbTH38) formed an unknown significant branch. According to the classification characteristics of trihelix gene family, we named it GTSb8. This may represent a new evolutionary branch of the trihelix gene family in sorghum. Additionally, three genes (SbTH07/34/37) branched independently and were named ‘orphan genes’, suggesting that the three genes may have unique functions. The SIP1, GTγ, SH4, GT1, GT2, and GTSb8 subfamilies contained 12, 4, 6, 5, 5, and 5 SbTH genes, respectively. There were no differences in the sequences of the 32 proteins shared with Arabidopsis thaliana and rice during S. bicolor evolution, however, eight gene duplication events occurred in the sorghum genome.
Unrooted phylogenetic tree showing relationships among Trihelix domains of S. bicolor and Arabidopsis. The phylogenetic tree was derived using the NJ method in MEGA7.0. The tree shows the 24 phylogenetic subfamilies and 1 unclassified group (GTSb8) marked with red font on a white background. Trihelix proteins from Arabidopsis are marked with the prefix ‘At’
Gene structure and motif analysis of trihelix genes in S. bicolor (L.)
Structures and phases of introns/exons were determined by aligning the genomic DNA with full-length cDNAs of SbTH genes. Generally, trihelix members grouped in the same branch shared similar exons/introns organization based on the exon/intron number (Fig. 2a, b). Structural characteristics of the SbTH genes, including the number and distribution of exons and introns, are shown in Fig. 2b. The CDS of more than half (25,62%) of the trihelix genes were isolated by the introns. By analysing the gene structural characteristics, we determined that 15 (38%) of the SbTH genes had no intron, 11 (28%) had only 1 intron, 8 (20%) had 2 introns, 2 (5%) had 3 introns, and 2 (5%) had 4 introns, and SbTH27 (2.5%) and SbTH18 (2.5%) contained 5 and 16 introns, respectively. The number of exons in the SbTH family varied from 1 to 17, with the GT1 subfamily having 1–17 exons, SIP1 subfamily having 1–6, GTSb8 subfamily having 2–4 exons, three orphan genes having 1–3, GT2 and SH4 subfamilies having 2–3, while GTγ subfamily had only 1 exon and no intron. The GTγ subfamily contained the average lowest number of exons, whereas the GT1 family contained the highest.
Phylogenetic relationships, gene-structure analysis, and motif distributions of S. bicolor trihelix genes
a Phylogenetic tree was constructed using the NJ method with 1000 replicates on each node.
b Exons and introns are indicated by yellow rectangles and gray lines, respectively.
c Amino acid motifs in the Sbtrihelix proteins (1–10) are represented by coloured boxes. The black lines indicate relative protein lengths.
As shown in Fig. 2c, we used the MEME search tool (http:/meme.nbcr.net/meme/intro.html) to predict 10 conserved motifs (motif 1 to motif 10) of SbTH proteins to further analyze the diversity of sorghum trihelix genes. The lengths of the conserved motifs varied from 15 to 50 amino acids. The motif organizations of each SbTH protein are shown with the corresponding colour boxes in Fig. 2c. In Additional File 2 (Table S2), the detailed sequence of each motif is provided. Motif 1 and motif 2 exist in almost all SbTH proteins, and all SbTH proteins contain motif 2. Different groups shared similar motifs, suggesting that these conserved motifs might play significant roles in particular functions. Moreover, some SbTH contained more than one motif 2. For instance, SbTH02, SbTH06, SbTH23, and SbTH30, which are members of the GT2 subfamily, contained two motifs 2. However, SbTH15, which is also a member of GT2 subfamily, contained no motif 3 and only one motif 2. Most members of the SIP1 subfamily contained motifs 1, 2, 5, 7 and 8, except for SbTH27, SbTH31, and SbTH04. Members of the SH4 subfamily contained motifs 1, 2, and 9, SbTH33 and SbTH01 also contained motif 8, and SbTH17 contained motif 5. Members of the GTγ subfamily contained motifs 1, 2, 5, and 10, and SbTH24 contained two motifs 1. There were two motifs 1 at the same time in SbTH07 and SbTH34, which may be linked to their unique functions.
Chromosome distribution and synteny analysis of trihelix gene in S. bicolor (L.)
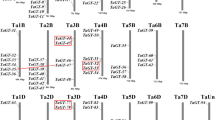
The chromosome positions of SbTH genes were extracted from the genome annotation files. As shown in Fig. S1, the 40 SbTH genes are unevenly and non-randomly distributed at precise positions on chromosome 1 to chromosome 9. The SbTH genes were named according to their physical positions on the S. bicolor chromosome from top to bottom. Chromosome 4 (Chr4) contained the largest number of SbTH genes (9, 22.5%), followed by Chr6 (8, 20%), Chr1 (6, 15%), Chr2 and Chr3 (4 genes each, 10%), Chr8 (3, 7.5%), and Chr5, Chr7, and Chr9 (2 genes each, 5%). SbTH04 and SbTH05 formed a tandem repeat at one end of Chr1 to form a gene cluster. Except for one SbTH gene in the middle of Chr2, the others were unevenly and non-randomly distributed on both ends of the chromosome. Additionally, there was a pair of tandem duplication gene (SbTH04/SbTH05, SIP1 subfamily gene) on chr1 and seven pairs of segmental duplication genes (Fig. 3, Additional File 3: Table S3). A chromosomal region within 200 kb exhibiting two or more identical genomic regions is defined as a tandem duplication event.
Schematic representation of the chromosomal distribution and interchromosomal relationships of S. bicolor trihelix genes. Coloured lines indicate all synteny blocks in the S. bicolor genome, and the red lines indicate duplicated trihelix gene pairs. Chromosome number is indicated at the bottom of each chromosome
As shown in Fig. 3, 13 (32.5%) paralogs were identified in the SbTH gene family, indicating an evolutionary relationship among these SbTH members. The SbTH genes were unevenly distributed in 10 S. bicolor linkage groups (LGs) (Fig. 3). Some LGs had more SbTH genes than others (LG1, LG4), with LG1 having the most SbTH genes (3). Further analysis of the subfamilies of these genes showed that all of them were linked within their subfamily. Among the six SbTH gene subfamilies, the SIP1 subfamily had the largest number of linked genes (6/13), whereas the GT2 and GTγ subfamilies had 3 linked genes each.
Evolutionary and synteny analyses between SbTH genes and those of several other species
To analyse the evolutionary relationship of the trihelix gene family between sorghum and five plants (Arabidopsis, wheat, rice, tomato, and buckwheat), an unrooted NJ tree with 10 conserved motifs was constructed using the NJ method of Geneious R11 according to the protein sequences of 40 SbTH genes and the trihelix genes of five other plants (Fig. S2, the detailed genetic correspondence can be found in Additional File 4: Table S4). The distribution of SbTH in the phylogenetic tree was relatively dispersed. Most members of the trihelix family from different species, shown in Fig. S2, shared motifs 2, and most trihelix family members contained motifs 1 and motifs 5. Generally, trihelix proteins in the same subfamily had similar motif compositions, and similar serial motifs tended to cluster in sorghum, wheat, and rice, indicating that SbTH proteins may be more closely related to those of rice and wheat than those of the other plants.
To examine the gene replication mechanism of sorghum trihelix family, we constructed six comparison system diagrams between sorghum and five representative species, including two dicotyledonous plants (Arabidopsis and tomato) and three monocotyledonous plants (buckwheat, wheat, and rice) (Fig. 4). From the details provided in Additional File 4 (Table S4), the number of collinear genes between sorghum and wheat, rice, tomato, and Arabidopsis were 27, 26, 5, and 4, forming 73, 35, 9, and 4 homologous gene pairs, respectively. By comparing the diagrams, we found that sorghum was the most similar with wheat and the least similar with buckwheat, which might be closely associated with the phylogenetic evolutionary relationship among them. SbTH03 gene showed collinearity in two monocots and two dicots, indicating that SbTH03 may be conserved in gene expansion induced by monocotyledon and dicotyledon differentiation, and play an important role in plant evolution and environmental adaptation. A total of 16 SbTH genes (SbTH01/12/13/15/ 17/20/22/23/24/27/28/30/32/33/36/39) were unique to monocots, indicating that these genes might have evolved after differentiation of monocots. Some SbTH genes were found to be associated with five synonymous gene pairs, including SbTH15/32/40. These genes may play a key role in the trihelix gene family during evolution. To better understand the evolutionary role of SbTH gene family, we performed Tajima D neutrality test (Additional File 5: Table S5). The results showed that the Tajima D value was far from 0, indicating that this gene family was strongly selected in the evolution of sorghum.
Expression patterns of SbTH genes in different tissues and organs
In the plants that have been studied and reported, the functional studies of many genes indicate that trihelix genes play a key role in crop growth and development [34]. To understand the physiological role of SbTH genes in sorghum growth and development, the expression levels of two selected genes from the seven subfamilies in different sorghum organs and tissues was examined using quantitative reverse transcription polymerase chain reaction (qPCR). The expression profiles of the SbTH genes in selected tissues, including of root, stem, leaf, pericarp, stamen, and pistil, are shown with histograms (Fig. 5a). The SbTH genes were highly expressed in specific tissues and organs, indicating that SbTH family members had multiple functions in the growth and developmental stages of sorghum. We observed that 10 SbTH genes (SbTH02/07/10/14/24/25/32/33/36/39) were relatively highly expressed in sorghum leaves, five (SbTH10/25/27/28/37) were relatively highly expressed in sorghum pericarps, and three (SbTH10/14/15) were relatively highly expressed in the stems,stamens and pistils. The relative expression of SbTH10 was the highest in leaves, pericarps, stamens and pistils, and SbTH15 was the highest in stems. Generally, the relative expression of the 14 SbTH genes in the sorghum seedling roots was low (Fig. 5a).
Tissue-specific gene expression and correlation between gene expression patterns of 14 sorghum trihelix genes a The expression patterns of 14 sorghum trihelix genes in the root (R), stem (SE), leaf (L), pericarp (PC), stamen (ST), and pistil (PS) tissues were examined by qPCR. Error bars were obtained from three measurements. Lowercase letter(s) above the bars indicate significant differences (α = 0.05, LSD) among the treatments. b The red round spot: positively correlated, the purple round spot: negatively correlated. The deepest and largest red round spot indicate a significant correlation at the 0.05 level.
Furthermore, we examined the correlation between the expression profiles of the 14 SbTH genes, and the result showed that majority of the SbTH genes were positively related, especially these SbTH genes (SbTH25/10/32/14/24/07/36/39) that were significantly correlated with several other SbTH genes. SbTH15 was negatively correlated with 12 SbTH genes except SbTH39, while SbTH28 was negatively correlated with SbTH32/14/24/36/39. Additionally, SbTH28, SbTH27, and SbTH30 were significantly positively correlated with one another. The correlation coefficient of SbTH24, SbTH07, and SbTH36 was 1 (Fig. 5b).
Expression patterns of SbTH genes in response to abiotic stress
To determine the role of SbTH genes in abiotic stress responses, we examined the expression profiles of 12 representative genes from the seven subfamilies under different abiotic stress conditions (high temperature, low temperature, osmotic, flooding, salt, and ultraviolet radiation) using qPCR. Figure 6 shows that most of the SbTH genes were expressed in different organs of sorghum after 2 h under high temperature, low temperature, and water flooding, whereas most of the SbTH genes were expressed after 24 h under osmotic, salt, and ultraviolet irradiation. There was a significant upregulation of the expression profiles of the 12 SbTH genes in the stems after 2 h under high temperature stress, however, SbTH37 expression was significantly upregulated in the roots after 24 h under high temperature stress. Most of the SbTH genes were highly expressed in the leaves after 2 h exposure to low temperature, while SbTH07 and SbTH37 were highly expressed in the stems and roots, respectively, after 24 h exposure to low temperature. Furthermore, most of the genes were upregulated in the roots after 24 h exposure to osmotic condition, however, SbTH25 and SbTH28 were relatively downregulated. Most of the genes (SbTH02/07/10/24/25/28) were upregulated in the leaves after 2 h exposure to flooding, while SbTH15, SbTH27, and SbTH32 were upregulated after 24 h exposure. Additionally, SbTH36 and SbTH37 were upregulated in the stems after 2 h exposure to flooding, while SbTH39 was upregulated in the roots after 24 h of exposure. Most of the genes (SbTH02/10/15/24/25/27/28) were upregulated in the leaves, while SbTH07, SbTH37, and SbTH39 were upregulated in the stems after 24 h exposure to ultraviolet radiation. However, SbTH32 was not upregulated in the roots, stems, and leaves. All the SbTH genes were highly expressed in leaves after 24 h of exposure to salt stress, in which the relative expression of SbTH10/24/37 reached hundreds or even thousands fold. Figure 7 shows the correlation coefficient diagram of 12 thrihelix genes transcriptional expression fold changes in response to abiotic stress. According to the analysis, there was a significant correlation between the relative expression of most genes at 2 h and 24 h, but no significant correlation between SbTH32 /37 and other genes. After 2 h of treatment, there was a negative correlation between SbTH32 and SbTH37, no significant correlation between SbTH37 and the other 11 thrihelix genes, between SbTH32 and SbTH39/24 was significant (Fig. 7a). After 24 h of treatment, the relative expression of SbTH37/07/32 was not significant compared with most other genes, between SbTH37 and SbTH07 was significant, between SbTH32 and SbTH37/07/25/36/10/24/15/27 was not significant, between SbTH07 and SbTH10/24/15/27/39/21 was not significant (Fig. 7b).
Correlation coefficient diagram of relative expression of 12 thrihelix genes in roots, stems and leaves of sorghum seedlings treated with 2 h (a) and 24 h (b) under different abiotic stress (FL: Flooding, HT: High temperature, LT: Low temperature, PEG: Osmotic, SA: Salt, UV: Ultraviolet radiation). The red round spot: positively correlated, the blue round spot: negatively correlated. The deepest and largest red round spot indicate a significant correlation at the 0.05 level
Expression patterns of 12 S. bicolor trihelix genes in roots, stems and leaves of sorghum seedlings treated with 2 h and 24 h under different abiotic stress (FL: Flooding, HT: High temperature, LT: Low temperature, PEG: Osmotic, SA: Salt, UV: Ultraviolet radiation) were examined by qPCR. Error bars were obtained from three measurements. Lowercase letter above the bar indicates significant difference (α = 0.05, LSD) among the treatments
Discussion
Sorghum is the dietary staple of over 500 million people in more than 30 countries in the tropics and semitropics [35]. Sorghum is a typical C4 crop and an important raw material for livestock nutrition and brewing industry. The sorghum reference genome was published in 2009 [36], however, whole genome studies of sorghum trihelix gene family have not been published. In the present study, 40 SbTH genes were identified in sorghum, similar to the number of SbTH genes in tomato and rice [12, 24]. Trihelix family genes were previously classified into three distinctive subfamilies (GTα, GTβ, and GTγ) [37]. Kaplan-Levy et al. classified trihelix genes from rice (Oryza sativa) and Arabidopsis into five clades namely GT-1, GT-2, SH4, SIP1, and GTγ [33]. Recently, a new subfamily, GTδ, was identified in tomato (Solanum lycopersicum) and rice [12, 24]. In the present study, phylogenetic analysis showed that sorghum trihelix genes were classified into six subfamilies (GT-1, GT-2, SH4, SIP1, GTγ, GTSb8) (Fig. 1). Based on the constructed phylogenetic tree, we identified at least one trihelix protein from S. bicolor in each subgroup of AtTHs and OsTHs [20, 24], indicating that the time of differentiation of the trihelix family may have been earlier than differentiation of monocotyledons and dicotyledons. The trihelix genes within the reported subfamilies may play a fundamental role in the tissue development, environmental adaptation and gene evolution in dissimilar plant species, including Arabidopsis thaliana [20], tartary buckwheat [30], Brachypodium distachyon [38], Moso bamboo [39], and wheat [29]. Compared to A. thaliana, the group SbSH4 (6, 15.0%) has more members, and indicates that those SbSH4 members may have undergone stronger partial differentiation in the long-term evolutionary process. A new subfamily (GTSb8) and three new ‘orphan genes’ were found in sorghum, suggesting the possibility of further differentiation of TH family in sorghum. This new cluster of ‘GTSb8’ indicates the complexity of genetic structure and physiological function of trihelix gene members. However, more evidence is needed to determine whether the new cluster is unique to C4 plants.
The results of motif composition and gene structure analysis of the trihelix genes were consistent with the phylogenetic classification results. The SbTH genes in the GTSb8 and ‘orphan gene’ subfamilies had only the trihelix domain (GT domain), whereas all members of the SH4, SIP1, and GTγ subfamilies had MYB DNA-binding domains. The similarity of most members in the same subfamily indicate that the conserved motifs may play a critical role in the functions of specific groups. Sequence distribution indicated that genes with the same motif may be generated by gene amplification in the identical population, which is similar to the report in chrysanthemum [40]. Among the seven subfamilies, GT-1 and GT-2 have been examined in previous studies, and their homology is much higher than that of other subfamilies [41]. Gene duplication is one of the major evolutionary mechanisms for generating novel genes that help organisms adapt to different environments [42, 43]. Generally, gene families expand mainly by tandem and segmental duplications [42]. Tandem and segmental duplications are key factors in enriching protein function and promoting gene evolution and expansion [44]. Sorghum had fewer trihelix genes than did soybean (71), Populus trichocarpa (56), and Brassica napus (52) [45,46,47], but more trihelix genes than did chrysanthemum (20), buckwheat (31), and tomato (36) [12, 27, 30]. This difference may be due to whole-genome duplication event that occurred after the earliest ancestors of the other species diverged. It is assumed that the occurrence and evolution of some SbTH genes may be driven by these fragment duplication events, which is similar to the report on Populus trichocarpa [46]. Based on the results of chromosome distribution, there was no SbTH gene on chromosome 10, indicating that the SbTH gene family may have been affected by gene deletion during the evolutionary process [48]. A similar phenomenon was reported in the rice and soybean trihelix gene families, which contained only 6 and 13 pairs of duplicated genes among a total of 41 (29.3%) rice trihelix genes and 71 (36.6%) soybean trihelix genes, respectively [21, 24]. Some SbTH gene deletions can be attributed to dynamic changes after fragment duplication, which is consistent with the findings of Populus trichocarpa [46]. In this study, tandem repeat events (SbTH04 / SbTH05) contributed less to the increase of sorghum trihelix membership than segmental duplicated (12 trihelix genes, 30.0%). Further analysis of these Trihelix members revealed that they were all linked within subfamilies. Therefore, some Trihelix genes may be generated by some replication events, which further confirms that replication events may be an important mechanism for the rapid expansion of Trihelix family members in plants.
In addition, we analyzed the exon and intron structures of 40 identified SbTH genes (Fig. 2, Attached File 1: Table S1). The number of exons in each gene ranged from 1 to 17 (Fig. 2A/2B). The proportion of SbTH gene without introns (15, 37.5%) was close to that of rice [24]. Interestingly, most of the intron-free genes are distributed in the GTγ subfamily and the SIP1 subfamily, which are similar to Arabidopsis thaliana [20]. A certain number of introns can increase the length of genes and the frequency of recombination between genes, which is beneficial to the evolution of species. However, intron-free genes tend to respond quickly to changes in the environment [49]. The lowest average number of exons was observed in the GTγ subfamily, whereas the highest was observed in the GT1 subfamily, which is consistent with the result in wheat [29] and buckwheat [30]. As the largest subfamily, the motif compositions of the SIP1 subfamily (most of the members of this clade shared motifs 1, 2, 5, 7, and 8) were different from that of other subfamily members, whose motif compositions were similar to that in cabbage, chrysanthemum [27], wheat [29], and Medicago truncatula [50]. The SIP1 subfamily members may have more complex and diverse functions than other subfamily members in sorghum.
Previous studies have shown that trihelix TF family is widely involved in the development of plant organs [30]. The expression levels of trihelix gene in sorghum stem, root, leaf and flower were determined by qPCR. As shown in Fig. 5a, most of trihelix gene members showed significant differential expression (more than 2-fold difference). SbTH02, is classified into subfamily GT2, has the highest expression levels in leaves and pistils, which is similar to the expression pattern of homologous gene AT5G03680.1, which regulates collective leaf structure and inflorescence development in Arabidopsis [49, 51]. As expected, the subfamily GT1 members, SbTH32 and SbTH39, are highly expressed in leaves and stamens, which was consistent with the expression pattern of the homologous gene AT1G13450 [52]. In addition, the expression of SbTH07, SbTH10, SbTH14, SbTH25, SbTH33, and SbTH36 in leaves of sorghum were significantly higher than those in roots, stems and pericarps. These tissue-specific trihelix genes may play a role in the growth and differentiation of corresponding organs, but more experiments are needed to verify the function of these genes [53]. In addition, some SbTH genes showed significant positive correlation, such as SbTH27 and SbTH28 (Fig. 5b). For example, SbTH27, SbTH28 and AtTH13, both belonging to subgroup SIP1 and having similar motif components (Fig. 2). The expression of SbTH27 and SbTH28 in pericarp of millet were significantly higher than those in roots, stems and leaves, and their expression pattern is similar to that of AtTH13 [54]. Therefore, we can further verify the possible relationship between these genes and pericarp development through some experiments. The expression levels of some SbTH members were significantly positively correlated, indicating that they may play a synergistic effect in six sorghum organs (Fig. 5b).
To further explore the physiological role of the trihelix family in environmental adaptation, we systematically analyzed the expression of 12 SbTHs in sorghum seedlings under six stresses (Fig. 6). For example, under NaCl stress, the expression levels of 10 SbTH genes were significantly up-regulated in roots, 8 in stems, and 7 in leaves and which may help sorghum adapt to drought conditions. In this study, under UV, PEG and NaCl treatment, SbTH15 showed obvious induction effect at seedling stage, and its expression level in stems and leaves were significantly increased. AtTH26 and SbTH15, are the members of subfamily GT2, have similar motif composition. Previous studies have shown that AtTH26 (At5G28300) can be induced by NaCl, drought, cold, and abscisic acid, and highly expressed in Arabidopsis inflorescence and leaves to help improve its resistance to adversity [55]. ShCIGT, a cold-inducible gene isolated from wild tomato, contributes to the improvement of abiotic stress tolerance in tomato [56]. Similarly, the expression of SbTH39 was significantly up-regulated in almost all abiotic stresses, which may enhance the adaptability of sorghum to the environment in a similar pattern. In Arabidopsis, the GT1 cis element interacts with the GT-1-like transcription factor AtGT-3b in vitro and in the yeast system. Transcription of AtGT-3b was also rapidly induced within 30 min after sodium chloride treatment, thus helping to enhance its resistance to salt stress [57]. Yoo et al. [58] found that GT2-like 1 (GTL1) in Arabidopsis thaliana is a transcriptional suppressor for promoter of STOMATAL DENSITY AND DISTRIBUTION 1 (SDD1), which can negatively regulate stomatal development and transpiration [57, 59]. Interestingly, SbTH37 is highly expressed in response to almost all stresses in some tissues, suggesting that some new evolutionary directions in sorghum may be the result of multiple adaptations to the environment. In addition, many studies have shown that TH-TFs are not only involved in response to abiotic stress, but also in disease resistance [15]. After being infected by Magnaporthe grisea, the GT-1-like gene in rice, rml1, can be rapidly up-regulated in seedlings to reduce the damage of the pathogen [14]. The GTL1 gene plays a key role in the MPK4 pathway in Arabidopsis by regulating the balance of salicylic acid and acting as a bacteria-induced immune factor [60]. SbTH28, a member of the subfamily GTSb8, was significantly down-regulated in roots under six stresses. This shows that it may actively participate in the response to abiotic stress. In summary, the expression patterns of SbTH members of the six subfamilies show great differences, which indicates that different genes may play a role with unique physiological functions. These results indicate that the trihelix gene family may play an important role in the tissue development and abiotic stress of sorghum, which needs further experimental verification.
Conclusion
In summary, the study is the first genome-wide analysis of trihelix genes in sorghum. We identified 40 trihelix genes in sorghum, which were classified into seven subfamilies and distributed in nine chromosomes. Additionally, we identified one pair of tandem duplication gene and seven pairs of segmental duplication genes in the SbTH gene family, indicating that gene duplication is the main force driving trihelix gene evolution in sorghum. Based on the expression profiles of the SbTH genes in different organs and tissues of sorghum under different abiotic stress conditions, some of the key candidate genes were screened out. For example, SbTH10, SbTH37, and SbTH39 may play important roles in the tissues development and abiotic stresses of sorghum. The findings of our study serve as a basis for further investigation of the functions of SbTH genes and provide candidate genes for increasing sorghum yield.
Methods
Gene identification
We downloaded the complete S. bicolor genome sequence (Accession: GCA_000003195) from the Ensembl Genomes website (http://plants.ensembl.org/Sorghum_bicolor/Info/Index). The trihelix family members were identified by two BLASTp searches [61, 62]. First, all possible trihelix proteins with score value ≥100 and e-value ≤1− 10 were identified from the S. bicolor genome, referring to trihelix protein sequences of A. thaliana by BLASTp search. Second, the HMM profile consistent with the trihelix domain was obtained from the Pfam protein family database (http://www.pfam.sanger.ac.uk). Candidate SbTH proteins containing the trihelix were screened out using HMMER3.0 (default parameters) with a cutoff of 0.01 (www.plants.ensembl.org/hmmer/index.html) [63] and SMART http://www.smart.embl-heidelberg.de). [64, 65]. In addition, information on basic features of the trihelix proteins of the SbTH gene family, including coding sequence length, isoelectric point, protein molecular mass, and subcellular localization, was obtained from the ExPasy website (http://web.expasy.org/protparam/).
Trihelix gene structure
The trihelix domain sequences of the characterised SbTH proteins were used to create multiple protein sequence alignments using ClustalW with default parameters [66]. The deduced amino acid sequences in the trihelix domains were then adjusted manually using Mega 6.0 and GeneDoc 2.7. The exon/intron structures of the SbTH genes were generated by the Gene Structure Display Server (GSDS: http://GSDS.cbi.pku.edu.cn) [67]. To compare the differences in SbTH proteins, the conserved motifs of the trihelix proteins were determined. The analysis of the conserved protein motifs in SbTH proteins was performed with the protein conserved motif online search program MEME (http://meme-suite.org/tools/meme) [68, 69]. The optimization parameters were set to the maximum number of motifs of 10 and the motif breadth as 6 to 200 amino acid residues [62, 69, 70].
Chromosomal distribution and gene duplication
All SbTH genes were mapped to S. bicolor chromosomes based on physical location information from the database of the S. bicolor genome using Circos [71]. The detection and study of the gene duplication events in SbTH genes were performed using the multiple collinear scanning toolkits (MCScanX) with default parameters [72]. We analysed the homology of the trihelix genes between S. bicolor and five plants (A. thaliana, V. vinifera, S. lycopersicum, B. distachyon, O. sativa subsp. indica, and Z. mays) using Dual Synteny Plotter (https://github.com/CJ-Chen/TBtools). Non-synonymous (ka) and synonymous (ks) substitutions of each duplicated trihelix gene were calculated using Ka/Ks-Calculator 2.0 [73].
Phylogenetic analysis and classification of trihelix gene family
The Arabidopsis trihelix and SbTH protein sequences were used for multiple amino acid sequence alignments using MEGA X software, and we used the NJ method with a bootstrap value of 1000 replicates and default parameters to construct the unrooted phylogenetic tree. The full-length amino acid sequences of the trihelix proteins (Additional file 1: Table S1) of SbTH in A. thaliana, V. vinifera, S. lycopersicum, B. distachyon, O. sativa subsp. indica, and Z. mays were used to construct the phylogenetic trees. The trihelix protein sequences were obtained from the UniProt database (UniProthttps://www.uniprot.org/). The identified SbTH genes were classified into different subfamilies.
Plant materials, growth conditions, and abiotic stress in S. bicolor
Sorghum bicolor ‘Hongyingzi’ was used for this study. The sorghum plants have been under cultivation in the greenhouse of Guizhou University since 2019. The plants were grown in pots filled with soil and vermiculite (1:1) in a growth chamber under a 16 h/25 °C day and 8 h/20 °C night regime and 75% relative humidity. We collected the stems, roots, leaves, pericarp, stamen, and pistil from five healthy sorghum plants. The organs were quickly fixed in liquid nitrogen and stored at − 80 °C until further analysis. The expression profiles of selected SbTH genes in different organs of 21-d-old sorghum plants after 2 h and 24 h under different abiotic stress conditions were examined using qPCR analysis. The seedlings were subjected to salt (900 mM NaCl), water flooding (whole plant), osmotic (30% PEG6000) [74, 75] [1], UV exposure (70 μW/cm2, 220 V, 30 W), high temperature and low temperature stress conditions (The plants were placed in light incubators at 40 °C and 4 °C, with 80% light, 16 h during the day, 8 h at night and 75% humidity.). Each stress treatment was performed with five replicates. Sorghum plants used for later sampling were planted in the teaching experimental field of Guizhou University, and the cultivation and management measures were consistent with the field production.
Total RNA extraction, cDNA reverse transcription, and qPCR analysis
Total RNA of each sample was extracted using a plant RNA extraction kit (TIANGEN DP441), and the sequences were used for cDNA library construction. qPCR SYBR Green Premix (Vazyme, China) was used to conduct qPCR analysis in a CFX™ real-time PCR detection system (Bio-Rad, USA). The primer sequences used were designed by Primer 5.0 (Additional File 6: Table S6). We used the Actin gene, which was stably expressed at each growth stage in almost all tissues, as the internal control [76]. The ACTIN gene was used as calibration to detect three technical repeats of the three biological repeats, and 2-ΔΔCT method was used to analyze the expression [77].
Statistical analysis
Data obtained during the study were subjected to analysis of variance (ANOVA) using SPSS software (IBM Corporation). Mean values were compared using Fisher’s least significant difference (LSD) test at 0.05 significance level. The histograms were drawn using Origin 8.0 software (OriginLab Corporation, Northampton, Massachusetts, USA).
Availability of data and materials
The entire Sorghum bicolor genome sequence (Accession:GCA_000003195) information was obtained from the Ensembl Genomes website (http://ensemblgenomes.org/). The Sorghum bicolor materials (Hongyingzi) used in the experiment were supplied by Prof. Mingjian Ren of Guizhou University. This variety was approved in the 5th Crop Variety Approval Committee of Guizhou Province on June 19, 2008, the second chairperson meeting, and was stored in the grain crop germplasm bank of College of Agriculture, Guizhou University. It was numbered GZ234. The permission of this material has been approved by the Guizhou University. The datasets supporting the conclusions of this article are included in the article and its Additional files.
Abbreviations
- TH:
-
Trihelix
- SbTH:
-
Sorghum bicolor Trihelix
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- qPCR:
-
Quantitative real-time polymerase chain reaction
- TF:
-
Transcription factor
- AtTH:
-
Arabidopsis thaliana Trihelix
- HMM:
-
Hidden Markov Model
- PI:
-
Isoelectric point
- LG:
-
Linkage group
- Mw:
-
Molecular weight
- PSL:
-
Predicted Subcellular Location
- N:
-
Nucleus
- Ch:
-
Chloroplast
- Ec:
-
Extracellular
- Cy:
-
Cytoplasm
- PL:
-
Protein length
References
Vishwakarma K, Shweta UN, Singh J, Liu SL, Singh VP, Prasad SM, et al. Differential phytotoxic impact of plant mediated silver nanoparticles (AgNPs) and silver nitrate (AgNO3) on Brassica sp. Front Plant Sci. 2017;8:1501. https://doi.org/10.3389/fpls.2017.01501.
Mizoi J, Shinozaki K, Yamaguchi-Shinozaki K. AP2/ERF family transcription factors in plant abiotic stress responses. Bba-Gene Regul Mech. 2012;1819(2):86–96.
Chinnusamy V, Schumaker K, Zhu JK. Molecular genetic perspectives on cross-talk and specificity in abiotic stress signalling in plants. J Exp Bot. 2004;55(395):225–36. https://doi.org/10.1093/jxb/erh005.
Lindemose S, O'Shea C, Jensen MK, Skriver K. Structure, function and networks of transcription factors involved in abiotic stress responses. Int J Mol Sci. 2013;14(3):5842–78. https://doi.org/10.3390/ijms14035842.
Jin JP, Tian F, Yang DC, Meng YQ, Kong L, Luo JC, et al. PlantTFDB 4.0: toward a central hub for transcription factors and regulatory interactions in plants. Nucleic Acids Res. 2017;45(D1):D1040–5. https://doi.org/10.1093/nar/gkw982.
Riechmann JL, Heard J, Martin G, Reuber L, Jiang C, Keddie J, et al. Arabidopsis transcription factors: genome-wide comparative analysis among eukaryotes. Science (New York, NY). 2000;290(5499):2105–10.
Green PJ, Yong MH, Cuozzo M, Kano-Murakami Y, Silverstein P, Chua NH. Binding site requirements for pea nuclear protein factor GT-1 correlate with sequences required for light-dependent transcriptional activation of the rbcS-3A gene. EMBO J. 1988;7(13):4305–044. https://doi.org/10.1002/j.1460-2075.1988.tb03297.x.
Green PJ, Kay SA, Chua NH. Sequence-specific interactions of a pea nuclear factor with light-responsive elements upstream of the rbcS-3A gene. EMBO J. 1987;6(9):2543–9. https://doi.org/10.1002/j.1460-2075.1987.tb02542.x.
Nagano Y. Several features of the GT-factor Trihelix domain resemble those of the Myb DNA-binding domain. Plant Physiol. 2000;124(2):491–3. https://doi.org/10.1104/pp.124.2.491.
Yao Q, Xin M, Guanghui Y, Qi W, Liang W, Lingrang K, et al. Evolutionary history of Trihelix family and their functional diversification. Narnia. 2014;21(5):499–510. https://doi.org/10.1093/dnares/dsu016.
Gao MJ, Lydiate DJ, Li X, Lui H, Gjetvaj B, Hegedus DD, et al. Repression of seed maturation genes by a Trihelix transcriptional repressor in Arabidopsis seedlings. Plant Cell. 2009;21(1):54–71. https://doi.org/10.1105/tpc.108.061309.
Yu C, Cai X, Ye Z, Li H. Genome-wide identification and expression profiling analysis of trihelix gene family in tomato. Biochem Biophys Res Commun. 2015;468(4):653–9. https://doi.org/10.1016/j.bbrc.2015.11.010.
Murata J, Takase H, Hiratsuka K. Characterization of a novel GT-box binding protein from Arabidopsis. Plant Biotechnol. 2002;19(2):103–12. https://doi.org/10.5511/plantbiotechnology.19.103.
Wang R, Hong G, Han B. Transcript abundance of rml1, encoding a putative GT1-like factor in rice, is up-regulated by Magnaporthe grisea and down-regulated by light. Gene. 2004;324:105–15. https://doi.org/10.1016/j.gene.2003.09.008.
Xie Z, Zou H, Lei G, Wei W, Zhou Q, Niu C, et al. Soybean Trihelix transcription factors GmGT-2A and GmGT-2B improve plant tolerance to abiotic stresses in transgenic Arabidopsis. PLoS One. 2009;4(9):1–14. https://doi.org/10.1371/journal.pone.0006898.
Yoo CY, Pence HE, Jin JB, Miura K, Gosney MJ, Hasegawa PM, et al. The Arabidopsis GTL1 transcription factor regulates water use efficiency and drought tolerance by modulating stomatal density via Transrepression of SDD1. Plant Cell. 2010;22(12):4128–41. https://doi.org/10.1105/tpc.110.078691.
Fang Y, Xie K, Hou X, Hu H, Xiong L. Systematic analysis of GT factor family of rice reveals a novel subfamily involved in stress responses. Mol Gen Genomics. 2010;283(2):157–69. https://doi.org/10.1007/s00438-009-0507-x.
Deschamps S, Zhang Y, Llaca V, Ye L, Sanyal A, King M, et al. A chromosome-scale assembly of the sorghum genome using nanopore sequencing and optical mapping. Nat Commun. 2018;9(1):1–10. https://doi.org/10.1038/s41467-018-07271-1.
Kaul S, Koo H, Jenkins J, Rizzo M, Rooney T. Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature: International Weekly Journal of Science. 2000;408(6814):796–815. https://doi.org/10.1038/35048692.
Yasmeen E, Riaz M, Sultan S, Azeem F, Abbas A, Riaz K, et al. Genome-wide analysis of trihelix transcription factor gene family in Arabidopsis thaliana. Pak J Agric Sci. 2016;53(2):439–48. https://doi.org/10.21162/PAKJAS/16.3347.
Liu W, Zhang Y, Li W, Lin Y, Wang C, Xu R, et al. Genome-wide characterization and expression analysis of soybean trihelix gene family. PeerJ. 2020;8:e8753. https://doi.org/10.7717/peerj.8753.
Shen Y, Liu J, Geng H, Zhang J, Liu Y, Zhang H, et al. De novo assembly of a Chinese soybean genome. Sci China Life Sci. 2018;61(8):871–84. https://doi.org/10.1007/s11427-018-9360-0.
Tanaka T, Nishijima R, Teramoto S, Kitomi Y, Hayashi T, Uga Y, et al. De novo genome assembly of the indica rice variety IR64 using linked-read sequencing and nanopore sequencing. G3-Genes Genom Genet. 2020;10(5):1495–501.
Li J, Zhang M, Sun J, Mao X, Wang J, Wang J, et al. Genome-Wide Characterization and Identification of Trihelix Transcription Factor and Expression Profiling in Response to Abiotic Stresses in Rice (Oryza sativa L.). Int J Mol Sci. 2019;20(2):251.
Takei H, Shirasawa K, Kuwabara K, Toyoda A, Matsuzawa Y, Iioka S, et al. De novo genome assembly of two tomato ancestors, Solanum pimpinellifolium and Solanum lycopersicum var. cerasiforme, by long-read sequencing. DNA Res. 2021;28(1):1–9. https://doi.org/10.1093/dnares/dsaa029.
Song C, Liu YF, Song AP, Dong GQ, Zhao HB, Sun W, et al. The Chrysanthemum nankingense genome provides insights into the evolution and diversification of Chrysanthemum flowers and medicinal traits. Mol Plant. 2018;11(12):1482–91. https://doi.org/10.1016/j.molp.2018.10.003.
Song A, Wu D, Fan Q, Tian C, Chen S, Guan Z, et al. Transcriptome-wide identification and expression profiling analysis of Chrysanthemum Trihelix transcription factors. IJMS. 2016;17(2):198. https://doi.org/10.3390/ijms17020198.
Walkowiak S, Gao LL, Monat C, Haberer G, Kassa MT, Brinton J, et al. Multiple wheat genomes reveal global variation in modern breeding. Nature. 2020;588(7837):277–83. https://doi.org/10.1038/s41586-020-2961-x.
Xiao J, Hu R, Gu T, Han JP, Qiu D, Su PP, et al. Genome-wide identification and expression profiling of trihelix gene family under abiotic stresses in wheat. BMC Genomics. 2019;20(1):287. https://doi.org/10.1186/s12864-019-5632-2.
Ma ZT, Liu MY, Sun WJ, Huang L, Wu Q, Bu TL, et al. Genome-wide identification and expression analysis of the trihelix transcription factor family in tartary buckwheat (Fagopyrum tataricum). BMC Plant Biol. 2019;19(1):344. https://doi.org/10.1186/s12870-019-1957-x.
Zhang LJ, Li XX, Ma B, Gao Q, Du HL, Han YH, et al. The Tartary buckwheat genome provides insights into Rutin biosynthesis and abiotic stress tolerance. Mol Plant. 2017;10(9):1224–37. https://doi.org/10.1016/j.molp.2017.08.013.
Qin Y, Ma X, Yu GH, Wang Q, Wang L, Kong LR, et al. Evolutionary history of Trihelix family and their functional diversification. DNA Res. 2014;21(5):499–510. https://doi.org/10.1093/dnares/dsu016.
Kaplan-Levy RN, Brewer PB, Quon T, Smyth DR. The trihelix family of transcription factors - light, stress and development. Trends Plant Sci. 2012;17(3):163–71. https://doi.org/10.1016/j.tplants.2011.12.002.
Smalle J, Kurepa J, Haegman M, Gielen J, Van Montagu M, Van Der Straeten D. The trihelix DNA-binding motif in higher plants is not restricted to the transcription factors GT-1 and GT-2. Proc Natl Acad Sci U S A. 1998;95(6):3318–22. https://doi.org/10.1073/pnas.95.6.3318.
Wu Y, Li X, Xiang W, Zhu C, Lin Z, Wu Y, et al. Presence of tannins in sorghum grains is conditioned by different natural alleles of tannin 1. Proc Natl Acad Sci U S A. 2012;109(26):10281–6. https://doi.org/10.1073/pnas.1201700109.
Paterson AH, Bowers JE, Bruggmann R, Dubchak I, Grimwood J, Gundlach H, et al. The Sorghum bicolor genome and the diversification of grasses. Nature. 2009;457(7229):551–6. https://doi.org/10.1038/nature07723.
Fang YJ, Xie KB, Hou X, Hu HH, Xiong LH. Systematic analysis of GT factor family of rice reveals a novel subfamily involved in stress responses. Mol Gen Genomics. 2010;283(2):157–69. https://doi.org/10.1007/s00438-009-0507-x.
Wang CW, Wang Y, Pan Q, Chen SK, Feng CZ, Hai JB, et al. Comparison of Trihelix transcription factors between wheat and Brachypodium distachyon at genome-wide. BMC Genomics. 2019;20(1):142. https://doi.org/10.1186/s12864-019-5494-7.
Cheng XR, Xiong R, Yan HW, Gao YM, Liu HL, Wu M, et al. The trihelix family of transcription factors: functional and evolutionary analysis in Moso bamboo (Phyllostachys edulis). BMC Plant Biol. 2019;19(1):154. https://doi.org/10.1186/s12870-019-1744-8.
Song A, Gao T, Wu D, Xin J, Chen S, Guan Z, et al. Transcriptome-wide identification and expression analysis of chrysanthemum SBP-like transcription factors. Plant Physiol Biochem. 2016;102:10–6. https://doi.org/10.1016/j.plaphy.2016.02.009.
Kuhn RM, Caspar T, Dehesh K, Quail PH. DNA binding factor GT-2 from Arabidopsis. Plant Mol Biol. 1993;23(2):337–48. https://doi.org/10.1007/BF00029009.
Kong H, Landherr LL, Frohlich MW, Leebens-Mack J. Ma H, dePamphilis CW: patterns of gene duplication in the plant SKP1 gene family in angiosperms: evidence for multiple mechanisms of rapid gene birth. Plant J. 2007;50(5):873–85. https://doi.org/10.1111/j.1365-313X.2007.03097.x.
Moore RC, Purugganan MD. The early stages of duplicate gene evolution. Proc Natl Acad Sci U S A. 2003;100(26):15682–7. https://doi.org/10.1073/pnas.2535513100.
Cannon SB, Mitra A, Baumgarten A, Young ND, May G. The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. BMC Plant Biol. 2004;4(1):10. https://doi.org/10.1186/1471-2229-4-10.
Wang W, Wu P, Liu T, Ren H, Li Y, Hou X. Genome-wide analysis and expression divergence of the Trihelix family in Brassica Rapa : insight into the evolutionary patterns in plants. Sci Rep-Uk. 2017;7(1):6463. https://doi.org/10.1038/s41598-017-06935-0.
Wang Z, Liu Q, Wang H, Zhang H, Xu X, Li C, et al. Comprehensive analysis of trihelix genes and their expression under biotic and abiotic stresses in Populus trichocarpa. Sci Rep-Uk. 2016;6(1):36274. https://doi.org/10.1038/srep36274.
Borges OM, Lauro BN, Graciela C, Carina TZA, Beatriz WS, Helena BZM, Márcia MP: Identification and in silico characterization of soybean trihelix-GT and bHLH transcription factors involved in stress responses. Genet Mol Biol 2012, 35(1 (suppl)):233–246.
Liu J, Chen N, Chen F, Cai B, Dal Santo S, Tornielli GB, et al. Genome-wide analysis and expression profile of the bZIP transcription factor gene family in grapevine (Vitis vinifera). BMC Genomics. 2014;15(1):281. https://doi.org/10.1186/1471-2164-15-281.
Shabalina SA, Ogurtsov AY, Spiridonov AN, Novichkov PS, Spiridonov NA, Koonin EV. Distinct patterns of expression and evolution of Intronless and intron-containing mammalian genes. Mol Biol Evol. 2010;27(8):1745–9. https://doi.org/10.1093/molbev/msq086.
Liu X, Zhang H, Ma L, Wang Z, Wang K. Genome-wide identification and expression profiling analysis of the Trihelix gene family under abiotic stresses in Medicago truncatula. Genes (Basel). 2020;11(11):1389. https://doi.org/10.3390/genes11111389.
Frerichs A, Thoma R, Abdallah AT, Frommolt P, Werr W, Chandler JW. The founder-cell transcriptome in the Arabidopsis apetala1 cauliflower inflorescence meristem. BMC Genomics. 2016;17(1):855. https://doi.org/10.1186/s12864-016-3189-x.
Tang XR, Hou AF, Babu M, Nguyen V, Hurtado L, Lu Q, et al. The Arabidopsis BRAHMA chromatin-remodeling ATPase is involved in repression of seed maturation genes in leaves. Plant Physiol. 2008;147(3):1143–57. https://doi.org/10.1104/pp.108.121996.
Song AP, Gao TW, Wu D, Xin JJ, Chen SM, Guan ZY, et al. Transcriptome-wide identification and expression analysis of chrysanthemum SBP-like transcription factors. Plant Physiol Bioch. 2016;102(15):10–6. https://doi.org/10.1021/ol901129v.
Krizek BA, Bantle AT, Heflin JM, Han H, Freese NH, Loraine AE. AINTEGUMENTA and AINTEGUMENTA-LIKE6 directly regulate floral homeotic, growth, and vascular development genes in young Arabidopsis flowers. J Exp Bot. 2021;72(15):5478–93. https://doi.org/10.1093/jxb/erab223.
Xi J, Qiu Y, Du L, Poovaiah BW. Plant-specific trihelix transcription factor AtGT2L interacts with calcium/calmodulin and responds to cold and salt stresses. Plant Sci. 2012;185-186:274–80. https://doi.org/10.1016/j.plantsci.2011.11.013.
Yu C, Song L, Song J, Ouyang B, Guo L, Shang L, et al. ShCIGT, a Trihelix family gene, mediates cold and drought tolerance by interacting with SnRK1 in tomato. Plant Sci. 2018;270:140–9. https://doi.org/10.1016/j.plantsci.2018.02.012.
Park HC, Kim ML, Kang YH, Jeon JM, Yoo JH, Kim MC, et al. Pathogen- and NaCl-induced expression of the SCaM-4 promoter is mediated in part by a GT-1 box that interacts with a GT-1-like transcription factor. Plant Physiol. 2004;135(4):2150–61. https://doi.org/10.1104/pp.104.041442.
Yoo CY, Hasegawa PM, Mickelbart MV. Regulation of stomatal density by the GTL1 transcription factor for improving water use efficiency. Plant Signal Behav. 2011;6(7):1069–71. https://doi.org/10.4161/psb.6.7.15254.
Xi J, Qiu YJ, Du LQ, Poovaiah BW. Plant-specific trihelix transcription factor AtGT2L interacts with calcium/calmodulin and responds to cold and salt stresses. Plant Sci. 2012;185:274–80. https://doi.org/10.1016/j.plantsci.2011.11.013.
Volz R, Kim SK, Mi JN, Mariappan KG, Guo XJ, Bigeard J, et al. The Trihelix transcription factor GT2-like 1 (GTL1) promotes salicylic acid metabolism, and regulates bacterial-triggered immunity. PLoS Genet. 2018;14(10):14(10). https://doi.org/10.1371/journal.pgen.1007708.
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997;25(17):3389–402. https://doi.org/10.1093/nar/25.17.3389.
Liu M, Ma Z, Wang A, Zheng T, Huang L, Sun W, et al. Genome-wide investigation of the auxin response factor gene family in Tartary buckwheat (Fagopyrum tataricum). Int J Mol Sci. 2018;19(11):3526. https://doi.org/10.3390/ijms19113526.
Finn RD, Clements J, Eddy SR. HMMER web server: interactive sequence similarity searching. Nucleic Acids Res. 2011;39(suppl):W29–37. https://doi.org/10.1093/nar/gkr367.
El-Gebali S, Mistry J, Bateman A, Eddy SR, Luciani A, Potter SC, et al. The Pfam protein families database in 2019. Nucleic Acids Res. 2019;47(D1):D427–32. https://doi.org/10.1093/nar/gky995.
Letunic I, Bork P. 20 years of the SMART protein domain annotation resource. Nucleic Acids Res. 2018;46(D1):D493–6. https://doi.org/10.1093/nar/gkx922.
Thompson JD, Gibson TJ, Higgins DG. Multiple sequence alignment using ClustalW and ClustalX. Curr Protoc Bioinformatics. 2003;1(1):1–22. https://doi.org/10.1002/0471250953.bi0203s00.
Guo AY, Zhu QH, Chen X, Luo JC. GSDS: a gene structure display server. Yi Chuan. 2007;29(8):1023–6. https://doi.org/10.1360/yc-007-1023.
Bailey TL, Boden M, Buske FA, Frith M, Grant CE, Clementi L, et al. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 2009;37(Web Server):W202–8. https://doi.org/10.1093/nar/gkp335.
Xie T, Chen CJ, Li CH, Liu JR, Liu CY, He YH. Genome-wide investigation of WRKY gene family in pineapple: evolution and expression profiles during development and stress. BMC Genomics. 2018;19(1):490. https://doi.org/10.1186/s12864-018-4880-x.
Liu M, Ma Z, Sun W, Huang L, Wu Q, Tang Z, et al. Genome-wide analysis of the NAC transcription factor family in Tartary buckwheat (Fagopyrum tataricum). BMC Genomics. 2019;20(1):113. https://doi.org/10.1186/s12864-019-5500-0.
Krzywinski M, Schein J, Birol I, Connors J, Gascoyne R, Horsman D, et al. Circos: an information aesthetic for comparative genomics. Genome Res. 2009;19(9):1639–45. https://doi.org/10.1101/gr.092759.109.
Wang Y, Tang H, Debarry JD, Tan X, Li J, Wang X, Lee T-h, Jin H, Marler B, Guo H et al. MCScanX: a toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res 2012, 40(7):e49, DOI: https://doi.org/10.1093/nar/gkr1293.
Wang D, Zhang Y, Zhang Z, Zhu J, Yu J. KaKs_Calculator 2.0: a toolkit incorporating gamma-series methods and sliding window strategies. Genomics Proteomics Bioinformatics. 2010;8(1):77–80. https://doi.org/10.1016/S1672-0229(10)60008-3.
Wang H, Ao P, Yang S, Zou Z, Wang S, Gong M. Molecular cloning and expression analysis of the gene encoding proline dehydrogenase from Jatropha curcas L. Appl Biochem Biotechnol. 2015;175(5):2413–26. https://doi.org/10.1007/s12010-014-1441-x.
Hao GP, Wu ZY, Chen MS, Cao MQ, Pelletier G, Huang CL, et al. ATHK1 gene regulates signal transduction of osmotic stress in Arabidopsis thaliana. Zhi Wu Sheng Li Yu Fen Zi Sheng Wu Xue Xue Bao. 2004;30(5):553–60.
Reddy PS, Reddy DS, Sivasakthi K, Bhatnagar-Mathur P, Vadez V, Sharma KK. Evaluation of Sorghum [Sorghum bicolor (L.)] Reference Genes in Various Tissues and under Abiotic Stress Conditions for Quantitative Real-Time PCR Data Normalization. Front Plant Sci. 2016;7:529.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT Method. Methods (San Diego, Calif). 2001;25(4):402–8.
Acknowledgements
We thank all of the colleagues in our laboratory for useful discussions and technical assistance. We would like to thank Editage (www.editage.cn) for English language editing. We are very grateful to the editor and reviewers for critically evaluating the manuscript and providing constructive comments for its improvement.
Funding
This research was supported by Guizhou Science and Technology Plan Project (Qiankehe LH [2017] No. 7056), Growth Project of Young Scientific and Technological Talents in Colleges and Universities of Guizhou Province (KY [2017] No. 284), Special Project for the Construction of Modern Agricultural Industrial Technology System of Characteristic Miscellaneous Cereals in Guizhou Province (Qiancainong [2019] No. 15), Special Fund for Revitalization of Top Ten Industries (High quality Tobacco and Alcohol) Industry in Guizhou Province for ‘Research on breeding of New varieties of Sorghum’ (Guizhou Finance Industry [2020] No. 198).
Author information
Authors and Affiliations
Contributions
KYL planned and designed the research and analysed the data. KYL and YF wrote the manuscript. LLD, YBZ, and MXS studied gene expression by qPCR. SSC identified the S. bicolor trihelix gene family and analysed gene structure. MFY and YQD studied chromosome distribution and gene duplication and performed syntenic analysis of S. bicolor trihelix genes. YSP and YBD analysed the evolutionary relationship of trihelix genes in several different species. MJR supervised the research. ZHL, HY, and LYZ revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by the authors. These methods were carried out in accordance with relevant guidelines and regulations. We confirm that all experimental protocols were approved by Guizhou University.
Consent for publication
Not Applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1 Table S1.
List of the 40 SbTH genes identified in this study. (XLS 143 kb)
Additional file 2 Table S2.
Analysis and distribution of the conserved motifs in S. bicolor trihelix proteins. (XLS 22 kb)
Additional file 3 Table S3.
Seven pairs of segmental duplicated S. bicolor trihelix genes. (XLS 22 kb)
Additional file 4 Table S4.
One-to-one orthologous gene relationships between S. bicolor and other plants. (XLS 43 kb)
Additional file 5 Table S5.
Results of Tajima’s D neutrality test.
Additional file 6 Table S6.
Primer sequences for qPCR.
Additional file 7 Fig. S1.
Schematic representation of the chromosomal distribution of S. bicolor trihelix genes. Vertical bars represent the chromosomes of S. bicolor. The chromosome number is indicated to the left of each chromosome. The scale on the left represents chromosome length.
Additional file 8 Fig. S2.
Phylogenetic relationship and motif composition of the trihelix proteins of S. bicolor and five different plant species. Outer panel: An unrooted phylogenetic tree constructed using Geneious R11 with the NJ method. Inner panel: Distribution of the conserved motifs in trihelix proteins. The differently coloured boxes represent different motifs and their positions in each trihelix protein sequence. The sequence information for each motif is provided in Additional File 2: Table S2.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Li, K., Duan, L., Zhang, Y. et al. Genome-wide identification and expression profile analysis of trihelix transcription factor family genes in response to abiotic stress in sorghum [Sorghum bicolor (L.) Moench]. BMC Genomics 22, 738 (2021). https://doi.org/10.1186/s12864-021-08000-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12864-021-08000-7