Abstract
Background
The ecotoxicological risk posed by metals and their mixtures in sediments depends on their bioavailability. Many methods for evaluating the bioavailability of metals in sediments/soils are time-consuming and expensive, and frequently result in equivocal outcomes. The diffusive gradients in thin films (DGT) technique is a good measure of bioavailability for metals that can avoid the above drawbacks. Therefore, more effective approaches to this method should be developed that focus on metal bioavailability. No studies have been conducted using DGT to assess metal mixtures to aquatic biota in sediments. This study is therefore the first attempt to assess sediment toxicity of metals and their mixtures to aquatic biota based on the DGT technique. The intertidal zone of the Pearl River Estuary is selected as a case study.
Results
The bioavailable (DGT-labile) concentrations of metals range as follows (μg/L): Cd, 0.34–3.62; Pb, 1.35–1.92; Ni, 0.67–92.83; Cu, 0.74–10.30; Zn, 28.60–296.94; Co, 0.03–58.85; Fe, 7.23–4539.36; and Mn, 19.40–6626.83. The risk quotient (RQ), which is the ratio between the measured metal concentrations in the environment (MEC) and the predicted no-effect concentration (PNEC), is conducted to evaluate the single metal risk. The RQ based on summing up the MEC/PNEC ratios (RQMEC/PNEC) and the RQ based on sum of toxic units (RQSTU) are used to assess risk of metal mixture. TheRQ values of Cd, Pb, Ni, Cu, Zn, Fe, and Mn significantly exceed 1, indicating that the adverse effects of the metals are not negligible. Regarding the toxicity of metal mixtures, the values of RQMEC/PNEC and RQSTU are both between 62.45 and 743.48, revealing that the possible risk has already occurred in the study area.
Conclusions
The two methods of RQMEC/PNEC and RQSTU based on DGT-labile metal concentrations are effective and suitable to estimate the toxicity of metal mixtures in sediments.
Similar content being viewed by others
Background
The ecotoxicological hazard of metals depends on their bioavailability, which is impacted by environmental biogeochemical processes involving metal characteristics, sediment properties, and biological behavior and physiology [1, 2]. Many of the methods for evaluating metal bioavailability in sediments/soils are both time-consuming and expensive, and may repeatedly lead to uncertain outcomes such as sequential extraction, isotope dilution exchange method, and single-step chemical extractions [3,4,5]. These methods are impacted by physical and chemical factors [4, 5]. Therefore, more effective approaches are needed.
Most sediment quality guidelines (SQG) for metal evaluation are dependent upon empirical correlations of adverse biological effects and metal concentrations; ranking these has established the basis for empirical SQG in the initial tier of assessments [6, 7]. These SQG are set up mostly in terms of total metal concentrations. However, it is known that metal bioavailability in sediments relies heavily on the partitioning processes between the solid and liquid phases [8, 9].
The partitioning processes are strongly impacted by acid volatile sulfide (AVS), simultaneously extractable metals (SEM), organic carbon (OC), and the ions of iron and manganese [2, 10,11,12]. Based on several of these factors, some equilibrium partitioning (EqP) models, such as the AVS–SEM model, have been constructed to estimate metal bioavailability in sediments. These EqP models do not interpret metal exposure that may occur via ingestion of particles by deposit-feeding organisms [3].
Diffusive gradients in thin films (DGT) is a passive sampling technique that has been applied to evaluate labile metal concentrations in waters, soils, and sediments since 1994 [13,14,15,16], and has recently been used to predict the toxicity and bioavailability of metals to benthic organisms [3, 17, 18]. Yet, the DGT technique has rarely been used for environmental toxicology purposes to determinate the concentrations of an integrated pool of labile metals present in water and in weakened form in waters associated with sediments [3].
Concentration addition (CA) and independent action (IA) are the two classic models for predicting mixture toxicity based on the toxicities of individual metals and their concentrations in the mixture [19]. CA assumes that metals share the same mechanism, while IA infers that constituents of a mixture do not share a similar toxicity mechanism [20]. Several studies have found that the real mixture toxicity is less than the mixture toxicity of CA predictions, but higher than the mixture toxicity of IA predictions [21,22,23]. The CA method considers a worst-case scenario, and is most widely used to evaluate the toxicity of mixtures.
An intertidal zone is the coastal area between the high tide and low tide [24]. An intertidal zone in good condition provides a comfortable habitat for benthic and epibenthic organisms, as well as playing an important role in supporting human well-being and the global geochemical cycle [25, 26]. As a buffer zone between sea and land, it has been affected by a high frequency of anthropogenic activities and is afflicted by the pressure of pollutants [27]. A massive of metals from land discharges into the coastal ecosystem and is deposited in the sediments. Until now, the information related to bioavailability and ecotoxicological risk of such metal mixtures based on DGT technique has not been available.
The Pearl River, which is the second largest river in China with respect to discharge amount, flows through 446,768 km2 of drainage area into the Pearl River Estuary (PRE) and finally reaches the South China Sea [28]. Unprecedented rates of development in the PRE region in the last few decades have led to the loss of 562 km2 of original intertidal marsh between 1978 and 2003 [29]. The intertidal zone ecosystem of PRE is now facing increasing pressures from heavy metal pollution due to the elevated metal discharges from different sources such as upstream discharges, domestic sewage, rock weathering or soil erosion on the mainland and runoff entering the waters [30, 31]. Accordingly, the intertidal zone of the PRE was chosen as the case study in this research.
At present, only one paper reported availability and ecotoxicological risk of one metal copper in coastal environment [16]. Therefore, to our knowledge, the bioavailability and ecotoxicological risk of metals based on DGT technique are currently scarce. In this study, we use DGT technique combined with a tiered approach, mainly based on the CA concept, to integrate the risk of metal mixtures and thus provide a novel method of contaminant evaluation for the sediments.
Materials and methods
Study area
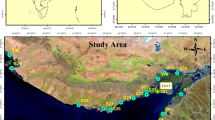
This study area is part of the Pearl River Estuary (PRE). The PRE is located in the south of the Tropic of Cancer, with a humid, hot and rainy tropical and subtropical monsoon climate. The annual mean temperature is 22.2 °C and the annual average precipitation is 1154.8–2702.2 mm [32]. PRE is a bell-shaped semi-enclosed sea on the Guangdong coast of South China, occupying an area of about 2500 km2 [28]. The average annual inflow of the RRE into the sea is 1.124 × 1011 m3 [32]. Water depths in the PRE vary from 2–5 m in the western region, to about 15 m in the eastern region [28]. The PRE is perennially affected by an irregular semidiurnal tide, with a tidal coefficient between 0.94 and 1.77 [32]. The ranges of water temperature, salinity, alkalinity and pH are 16.64–30.09 °C, 0–35‰, 1.80–2.40 meq/L and 6.5–8.58, respectively [32,33,34].
Sampling and analysis of sediment properties
Undisturbed surface sediments of 0–3 cm were carefully gathered from 21 sites (Fig. 1) during summer tide in June 2018 using a triangular plastic spade. Five subsamples were obtained at each site and these were thoroughly mixed to achieve a typical sample. The samples from each site were then divided into half, with each part put separately into a clean plastic bag. One half of each sample was then freeze-dried, cleaned of visible debris such as rock and plant fragments, ground in a clean mortar, passed through a 2000-μm stainless-steel sieve for homogenization, and kept at − 20 ℃ until analysis of DGT-labile metals, organic matter (OM), and inorganic carbonate (CaCO3). The other part of each sample was stored at − 4 ℃ for the determination of particle size. Details of how to determine sediment properties (OM, CaCO3, and particle size) are provided in the Additional file 1.
DGT experiment
Procedure of DGT extraction
The Chelex DGT device used in this study is composed of a binding gel (0.40 mm thickness), a diffusive gel (0.90 mm thickness), and a filter membrane (0.80 mm thickness) (EasySensor Ltd, www.easysensor.net). These three layers are assembled with the dual-mode holder, which consists of an “O-shape” ring, a recessed base, and a hollow base that offers structural support and accommodates the gels [35]. Target metals diffuse through the membrane filter and diffusive gel, and are accumulated on the binding gel [15]. Afterwards, the metals associated with the binding gel are extracted using acid solution for further analysis.
The DGT application procedure to measure homogenized sediments complies with the method proposed by Wang et al. [9]. Accordingly, the method involves three sequential steps: (1) pretreatment of samples; (2) DGT deployment on samples; and (3) DGT retrieval.
In initial step, sample of about 10 g was weighted, placed in a beaker, added deionized water to reach 70–80% water holding capacity, mixed thoroughly. The well stirred sediment was covered with plastic wrap to prevent water evaporation and incubated for 48 h at a constant ambient temperature.
In next step, a small amount of rehydrated sediments homogenized was added to the open cavity of the DGT device using a plastic spoon, shaken gently to ensure the sediment settles fully and was in contact with the surface of the filter membrane, with more sediment then added to completely fill the cavity. The loaded DGT device was then transferred into a plastic, semi-opened bag for incubation for 24 h at a constant ambient temperature, with 1–2 mL deionized water added to the bag to maintain moisture during deployment.
In last step, 24 h later, the DGT core was unscrewed and pulled out of the base using another base. The surface of the filter membrane was rinsed with deionized water and the binding gel retrieved and placed in a centrifugal tube. 1.8 mL 1 M HNO3 was added to the tube to immerse the gel and then the tube was closed and kept at 4 °C for 16 h. The elution was collected for analysis of DGT-labile metals. The concentrations of Cd, Pb, Ni, Cu, Zn, Co, Fe, and Mn were determined by inductively coupled plasma mass spectrometry (ICP-MS). Absolute difference between two independent determination results obtained under repeatability conditions not exceeded 10% of arithmetic mean value. The research work related to DGT was performed in the laboratory of EasySensor Ltd.
Models for calculating DGT-labile metal concentration
The target metal observes Fick’s 1st law of diffusion in the diffusive layer. The target metal concentration from the DGT extracting solution can be transformed to a mass of metal (M) through Eq. (1) as follows:
where Ce is the target metal concentration in the extracting solution; Ve is the volume (mL) of the extracting solution; Vg is the volume (mL) of gel (in this study, Vg is 0.2 mL); and fe is extraction rate, which is 0.938 for Cd, 0.955 for Pb, 1.05 for Ni, 1.03 for Cu, 0.88 for Zn, 0.975 for Co, 0.889 for Fe, and 0.967 for Mn [9]. The DGT-labile concentration is then calculated via Eq. (2):
where △g is the thickness of the diffusive layer and equal to 0.9 mm in this study; D is the diffusion coefficient of the target metal, which for the studied metals relies on the EasySensor user manual (http://www.easysensor.net/col.jsp?id=109); A is the exposed surface area of the DGT device (3.14 cm2); and t is the deployment time, which is equal to 48 h in this study.
Toxicity data assemblage
The acute toxic data relevant to Cd, Pb, Ni, Cu, Zn, Co, Fe, and Mn were retrieved from the USEPA ECOTOX database that includes algae, crustaceans, and fish (https://cfpub.epa.gov/ecotox/). These aquatic species are from freshwater and saltwater media types.
In order to reduce errors in any species with varying acute toxicity data, and to reflect the specific metal toxicity to aquatic biota, the median values of metals in algae, crustaceans, and fish species were calculated as shown in Table 1. For fish, the EC50 values of Cd, Pb, Cu, and Zn are used in this study. Due to the paucity of EC50 data for the other metals in fish species, the LC50 data of Ni, Co, Fe, and Mn are conducted in this study. Detailed information about the values of toxicity data and aquatic species are given in the Additional file 1.
Ecotoxicological risk model
Single metals
The risk quotient (RQ), which is the ratio between the measured metal concentrations in the environment (MEC) and the predicted no-effect concentration (PNEC), is used to evaluate the ecotoxicological risk for each DGT-labile metal as given in Eq. (3):
Table 1 explains how to calculate the PNEC and gives the values obtained in this study. The RQ is considered a robust method for assessing single pollutant ecotoxicology risk and is widely used in environment studies [19, 36, 37]. An RQ value less than 1 indicates no potential ecotoxicological risk, while an RQ value higher than 1 suggests a potential ecological risk and that the environmental risk posed should not be excluded [28, 38]. A larger value indicates a greater potential risk.
Mixture of metals
The RQ for the mixture based on MEC/PNEC ratios (RQMEC/PNEC) and the RQ for the mixture based on toxic units (RQSTU) are acceptable and extensively used models [19, 38, 39]. The two models are as follows in Eqs. (4)–(5):
where AF is the assessment factor (= 1000) [38].
In both models, if the RQs exceeded 1, the environmental risk posed by the mixture should be considered. The risk assessment was constructed considering firstly all the metals, and later only the metals with RQ below 1, to evaluate the potential risk of this mixture too [33].
Statistical analysis
Prior to factor analysis (FA), the normal distribution of each sediment-property variable and DGT-labile metal concentration was tested, which showed that only OM fitted normality. Therefore, the contents of Cd, Ni, Cu, Co, Fe, and CaCO3 were log-transformed, and the concentrations of Pb, Zn, Mn, and the median grain size were transformed with the Box–Cox method. These transformed parameters then fitted normal distributions. The transformed parameters and OM were ultimately standardized for FA. Data transformations were conducted with StatSoft STATISTICA 8.0 and FA was implemented with IBM SPSS Statistics 19.0.
Results and discussion
Physicochemical properties of sediments
OM, CaCO3, and particle size composition were determined to find the general properties of sediments in this study; these are given in Additional file 1: Table S1. The OM content was between 1.44% and 14.91% with a mean of 7.42%, and the CaCO3 content ranged between 0.76% and 17.26% of the dry sediment weight with a mean of 2.68%. The median grain size (MZ) varied from 2.31 to 7.06 phi (Φ) with an average of 5.24 Φ.
It is well established that OM and particle size are crucial controlling factors that alter natural metal content sediments [40, 41]. In addition, CaCO3 plays a role in influencing natural metal distribution [42]. Sediment with a smaller grain diameter is prone to present relatively high metal contents, due in part to specific surface area. This enrichment is primarily ascribed to surface adsorption and ionic attraction [40]. Meanwhile, OM is frequently found on particles in smaller sediments, and the biofilm binds a range of trace metals [43]. The unitary linear regression relationships between MZ and OM and CaCO3 are shown in Fig. 2. Except at sites S3 and S16, OM content increases with an increase of Φ value (Fig. 2A). Sites S3 and S16 deviate from the linear trend, which could be due to localized sediment composition. For example, site S3 is located in mangrove forest, where sediment is composed mainly of sand (Fig. 2C). Mangrove forest can affect OM content in sediments. Site S16 is located in Spartina alterniflora wetland (Fig. 2D), where sediment has highest OM content among all the sampling sites (Fig. 2A). In the relationship between CaCO3 and MZ, CaCO3 content represents an increase with the increase of Φ value, except at site S21 which falls off the trendline, possibly due to its location in an area of shellfish aggregation (Fig. 2B).
DGT-labile concentration in sediments
The spatial distributions and concentrations of the studied metals in surface sediments using DGT technique are shown in Fig. 3 and Additional file 1: Table S1. The highest values of Cd, Ni, Zn, and Co are found at site S2; the highest values of Pb, Cu, Fe, and Mn are located at site S18, site S16, site S1, and site S7, respectively. The mean DGT-labile concentrations are as follows: Mn (2327.80 μg/L) > Fe (604.03 μg/L) > Zn (52.13 μg/L) > Ni (10.18 μg/L) > Co (8.04 μg/L) > Cu (2.90 μg/L) > Pb (1.46 μg/L) > Cd (0.79 μg/L). DGT-labile metal concentrations (μg/L) in surface sediments of this study area demonstrated wide variations with Cd, 0.34–3.62; Pb, 1.35–1.92; Ni, 0.67–92.83; Cu, 0.74–10.30; Zn, 28.60–296.94; Co, 0.03–58.85; Fe, 7.23–4539.36; and Mn, 19.40–6626.83. The use of plastic sieves for trace metal analysis is recommended by United Nations [44]. However, the stainless-steel sieve during the preparation of the sediment is used in this study. Generally, the stainless-steel sieve contains Ni, Fe and Co. Consequently, the Ni, Fe and Co data may be a little overestimate because of the contact of the sediment with the stainless-steel sieve during the preparation of the samples.
In order to identify possible relationships between sediment-property parameters and DGT-labile metals, FA with the principal component method was performed on the data. In this study, FA (VARIMAX rotation mode) was conducted to identify four factors (Fs) extracted in terms of eigenvalues (> 1), representing 85.88% of the data. The communalities interpreted by the variables (sediment-property parameters and DGT-labile metals), considering four Fs, vary from 77.6% for Pb to 95.1% for Cu. All variables are well represented by the four Fs (F1, F2, F3, and F4), accounting for 27.07%, 25.45%, 19.44%, and 13.92% of the data, respectively. The loadings of metals and the sediment-property parameters and factor scores for the investigated sites are tabulated in Table 2. F1 has strong positive loadings for Ni, Co, Fe, Mn, and OM (> 0.5), indicating that OM mainly influences the metals of Ni, Co, Fe, and Mn in the F1 sites that score > 0; F2 is loaded with Cd, Pb, Ni, Zn and Co (> 0.5), indicating that no sediment-property parameter influences the metals of Cd, Pb, Ni, Zn and Co in the F2 sites of score > 0; F3 represents heavy positive loading for OM, CaCO3, and MZ (> 0.5) and F4 has only heavy positive loading for Cu (> 0.5), suggesting that no sediment-property parameter influences the metals in the F3 sites (score > 0) and F4 sites (score > 0).
Ecotoxicological risk assessment
Risk for individual metals
The RQ values for each single metal in surface sediments are illustrated in Fig. 4 and Additional file 1: Table S2. The average values of RQ follow the sequence: Zn > Mn > Fe > Cu > Ni > Cd > Co > Pb. Based on the one-sample t-test (P < 0.05), the RQ values of Cd, Pb, Ni, Cu, Zn, Fe, and Mn are significantly above 1, suggesting that the adverse ecological effects of these metals are not negligible. In detail, the RQ values of Cd, Ni, Cu, Zn, Fe, and Mn at all sites are higher than the threshold value of 1; the RQ values of Pb and Co exceed the threshold value at three sites (14.29%) for Pb and six sites (28.57%) for Co, indicating that these metals may be originated from anthropogenic input of metals.
The overall results show that ecotoxicological risk cannot be eliminated in the study area because most metals in most sites exceed the threshold value of 1 (Fig. 3). Therefore, further detailed study should be implemented as soon as possible based on current directives.
Risk for metal mixtures
To achieve a greater predictable ecotoxicological risk assessment, we also evaluate the integrated risk of metal mixtures in surface sediments. As discussed, the RQ for the mixture based on MEC/PNEC ratios (RQMEC/PNEC) and the RQ for the mixture based on toxic units (RQSTU) are two methods widely used to estimate the combined toxicity of pollutant mixtures [18, 35].
Table 3 provides the STUs for aquatic organisms of three different trophic levels, as well as the RQs in terms of the MEC/PNEC ratios (RQMEC/PNEC) and the STUs (RQSTU). The values of RQMEC/PNEC and RQSTU are in the ranges of 70.77–704.83 and 62.45–743.48, respectively, indicating the possible risk. Generally, the RQs computed with the two methods were not different (Table 3). The risk assessment was computed firstly considering all the metals with MEC/PNEC higher than 1, and later considering only the metal with MEC/PNEC less than 1 when considered singly. In this study, the RQs were above 1 for the mixture, as expected. This highlights the importance of the risk assessment for mixtures of pollutants rather than just the single pollutants.
In addition, the unitary linear regression analysis between RQMEC/PNEC and RQSTU shows that RQMEC/PNEC content increases with an increase of RQSTU, indicating that the two methods are effective and reliable in assessing the toxicity of metal mixtures (Fig. 5). Accordingly, further study should be carried out in the Pearl River intertidal zone.
Conclusion
Concentrations of eight metals (Cd, Pb, Ni, Cu, Zn, Co, Fe, and Mn) from surface sediment samples from the Pearl River intertidal zone are analyzed based on DGT technique. The results show that DGT-labile concentrations demonstrate crucial spatial variation in both the single metal toxicity and integrated toxicity of metal mixtures to aquatic organisms from three trophic levels (algae, crustaceans, and fish species). The RQ for single metals indicates that Cd, Pb, Ni, Cu, Zn, Fe, and Mn are significantly above 1 and that their ecological adverse effects are therefore not negligible. Two different methods to estimate toxicity in metal mixtures—RQMEC/PNEC and RQSTU—demonstrate that the possible risk has already occurred in the study area. Linear-regression analysis between RQMEC/PNEC and RQSTU shows that the two methods are effective and suitable to estimate the toxicity of metal mixtures. Therefore, the ecological risk based on DGT technique should be fully taken into consideration to protect aquatic ecosystems.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request
Abbreviations
- DGT:
-
Diffusive gradients in thin films
- RQ:
-
Risk quotient
- MEC:
-
Measured metal concentrations in the environment
- PNEC:
-
Predicted no-effect concentration
- RQMEC/PNEC :
-
The RQ for the mixture based on MEC/PNEC ratios
- RQSTU :
-
RQ for the mixture based on toxic units
- SQG:
-
Sediment quality guidelines
- AVS:
-
Acid volatile sulfide
- SEM:
-
Simultaneously extractable metals
- OC:
-
Organic carbon
- EqP:
-
Equilibrium partitioning
- CA:
-
Concentration addition
- IA:
-
Independent action
- PRE:
-
Pearl River Estuary
- OM:
-
Organic matter
- CaCO3 :
-
Inorganic carbonate
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- PNEC:
-
Predicted no-effect concentration
- FA:
-
Factor analysis
- MZ:
-
Median grain size
References
Warren LA, Haack EA (2001) Biogeochemical controls on metal behaviour in freshwater environments. Earth Sci Rev 54:261–320
Jonge DM, Teuchies J, Meire P, Blust R, Bervoets L (2012) The impact of increased oxygen conditions on metal-contaminated sediments part I: effects on redox status, sediment geochemistry and metal bioavailability. Water Res 46:2205–2214
Amato ED, Simpson SL, Jarolimek CV, Jolley DF (2014) Diffusive gradients in thin films technique provide robust prediction of metal bioavailability and toxicity in estuarine sediments. Environ Sci Technol 48:4485–4494
Dai YC, Nasir M, Zhang YL, Wu HM, Guo HH, Lv J (2017) Comparison of DGT with traditional methods for assessing cadmium bioavailability to Brassica chinensis in different soils. Sci Rep 7:14206
Kim R-Y, Yoon J-K, Kim T-S, Yang JE, Owens G, Kim K-R (2015) Bioavailability of heavy metals in soils: definitions and practical implementation—a critical review Environ Geochem Health 37:1041–1061
Long E, Macdonald D, Smith S, Calder F (1995) Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ Manage 19:81–97
Long ER, MacDonald DD (1998) Recommended uses of empirically derived, sediment quality guidelines for marine and estuarine ecosystems. Hum Ecol Risk Assess 4:1019–1039
Kumar M, Gogoi A, Mukherjee S (2020) Metal removal, partitioning and phase distributions in the wastewater and sludge: performance evaluation of conventional, upflow anaerobic sludge blanket and downflow hanging sponge treatment systems. J Clean Prod 249:119426
Wang Y, Ding SM, Gong MD, Xu SW, Xu WM, Zhang CS (2016) Diffusion characteristics of agarose hydrogel used in diffusive gradients in thin films for measurements of cations and anions. Anal Chim Acta 945:47–56
Teuchies J, De Jonge M, Meire P, Blust R, Bervoets L (2012) Can acid volatile sulfides (AVS) influence metal concentrations in the Macrophyte Myriophyllum aquaticum? Environ Sci Technol 46:9129–9137
Di Toro DM et al (2005) Predicting sediment metal toxicity using a sediment biotic ligand model: methodology and initial application. Environ Toxicol Chem 24:2410–2427
Nealson KH (2010) Geomicrobiology: sediment reactions defy dogma. Nature 463:1033–1034
Gao L, Gao B, Xu DY, Liu LH (2020) DGT: A promising technology for in-situ measurement of metal speciation in the environment. Sci Total Environ 715:136810
Davison W, Zhang H (1994) In situ speciation measurements of trace components in natural waters using thin-film gels. Nature 367:546–548
Zhang CS, Ding SM, Xu D, Tang Y, Wong MH (2014) Bioavailability assessment of phosphorus and metals in soils and sediments: a review of diffusive gradients in thin films (DGT). Environ Monit Assess 186:7367–7378
Umbría-Salinas K, Valero A, Martins SE, Wallner-Kersanach M (2021) Copper ecological risk assessment using DGT technique and PNEC: a case study in the Brazilian coast. J Hazard Mater 403:123918
Koppel DJ, King CK, Brown KE, Price GAV, Adams MS, Jolley DF (2020) Assessing the risk of metals and their mixtures in the Antarctic nearshore marine environment with diffusive gradients in thin-films. Environ Sci Technol 54:306–315
Amato ED, Marasinghe Wadige CPM, Taylor AM, Maher WA, Simpson SL, Jolley DF (2018) Field and laboratory evaluation of DGT for predicting metal bioaccumulation and toxicity in the freshwater bivalve Hyridella australis exposed to contaminated sediments. Environ Pollut 243:862–871
Riva F, Zuccato E, Davoli E, Fattore E, Castiglioni S (2019) Risk assessment of a mixture of emerging contaminants in surface water in a highly urbanized area in Italy. J Hazard Mater 361:103–110
Nys C, Van Regenmortel T, Janssen CR, Oorts K, Smolders E, De Schamphelaere KAC (2018) A framework for ecological risk assessment of metal mixtures in aquatic systems. Environ Toxicol Chem 37:623–642
Walter H, Consolaro F, Gramatica P, Scholze M, Altenburger R (2002) Mixture toxicity of priority pollutants at no observed effect concentrations (NOECs) Ecotoxicology 11:299–310
Junghans M, Backhaus T, Faust M, Scholze M, Grimme LH (2006) Application and validation of approaches for the predictive hazard assessment of realistic pesticide mixtures. Aquat Toxicol 76:93–110
Hasenbein S, Lawler SP, Geist J, Connon RE (2015) The use of growth and behavioral endpoints to assess the effects of pesticide mixtures upon aquatic organisms. Ecotoxicology 24:746–759
NOAA (National Oceanic and Atmospheric Administration US) (2020) What is the intertidal zone? https://oceanservice.noaa.gov/facts/intertidal-zone.html.
Lv M et al (2020) A national-scale characterization of organochlorine pesticides (OCPs) in intertidal sediment of China: Occurrence, fate and influential factors. Environ Pollut 257:113634
Halpern BS et al (2012) An index to assess the health and benefits of the global ocean. Nature 488:615–620
Halpern BS et al (2008) A global map of human impact on marine ecosystems. Science 319:948–952
Gu YG, Gao YP, Huang HH, Wu FX (2020) First attempt to assess ecotoxicological risk of fifteen rare earth elements and their mixtures in sediments with diffusive gradients in thin films. Water Res 185:116254
Bai JH et al (2011) Assessment of heavy metal pollution in wetland soils from the young and old reclaimed regions in the Pearl River Estuary. South China Environ Pollut 159:817–824
Zhang GL, Bai JH, Xiao R, Zhao QQ, Jia J, Cui BS, Liu XH (2017) Heavy metal fractions and ecological risk assessment in sediments from urban, rural and reclamation-affected rivers of the Pearl River Estuary, China. Chemosphere 184:278–288
Cheung KC, Poon BHT, Lan CY, Wong MH (2003) Assessment of metal and nutrient concentrations in river water and sediment collected from the cities in the Pearl River Delta. South China Chemosphere 52:1431–1440
China Bay Chronicles Compilation Committee (1998) Survey of China Bays: Key Estuaries, vol 14. China Ocean Press, Beijing, pp 239–298
Peng YH, Chen HR, Li SF (1991) pH and Alkalinity in the water body of Pearl River Mouth. Trop Oceanol 10:49–55
Chen MR, Si YY, Han LY, Liu X, Huang BQ, Kang CK (2021) Effect of prey selectivity and trophic cascades induced by mesozooplankton on the dynamics of phytoplankton. Mar Ecol Prog Ser 662:35–51
Ding SM, Wang Y, Zhang LP, Xu L, Gong MD, Zhang CS (2016) New holder configurations for use in the diffusive gradients in thin films (DGT) technique. RSC Adv 6:88143–88156
Ccanccapa A, Masiá A, Navarro-Ortega A, Picó Y, Barceló D (2016) Pesticides in the Ebro River basin: occurrence and risk assessment. Environ Pollut 211:414–424
Meng Y et al (2019) A review on occurrence and risk of polycyclic aromatic hydrocarbons (PAHs) in lakes of China. Sci Total Environ 651:2497–2506
Backhaus T, Faust M (2012) Predictive environmental risk assessment of chemical mixtures: a conceptual framework. Environ Sci Technol 46:2564–2573
Nika MC et al (2020) Wide-scope target analysis of emerging contaminants in landfill leachates and risk assessment using Risk Quotient methodology. J Hazard Mater 394:122493
Zhang LP et al (2007) Heavy metal contamination in western Xiamen Bay sediments and its vicinity, China. Mar Pollut Bull 54:974–982
Zhang WG, Feng H, Chang JN, Qu JG, Xie HX, Yu LZ (2009) Heavy metal contamination in surface sediments of Yangtze River intertidal zone: an assessment from different indexes. Environ Pollut 157:1533–1543
Gu YG (2018) Heavy metal fractionation and ecological risk implications in the intertidal surface sediments of Zhelin Bay, South China. Mar Pollut Bull 129:905–912
Wangersky PJ (1986) Biological control of trace metal residence time and speciation: a review and synthesis. Mar Chem 18:269–297
United Nations (2020) Agenda item 5: Parallel CORMON Sessions for Pollution, including Marine Litter and Biodiversity-Monitoring Guidelines/Protocols for Sampling and Sample Preservation of Sediment for IMAP Common Indicator 17: Heavy and Trace Elements and Organic Contaminant, UNEP/MED WG.482/11
Acknowledgements
Not applicable.
Funding
This study was funded by the National Key R&D Program of China (2019YFD0901105).
Author information
Authors and Affiliations
Contributions
Y-GG did all work including study design, investigation, experiments, data curation, validation, conceptualization, manuscript writing, correction and supervision. The author read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by the only author Yang-Guang Gu.
Consent for publication
The consent for publication is approved by Yang-Guang Gu.
Competing interests
Author Yang-Guang Gu declares that he has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Determination of sediment properties.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gu, YG. Risk assessment of eight metals and their mixtures to aquatic biota in sediments with diffusive gradients in thin films (DGT): a case study in Pearl River intertidal zone. Environ Sci Eur 33, 122 (2021). https://doi.org/10.1186/s12302-021-00564-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12302-021-00564-1