Abstract
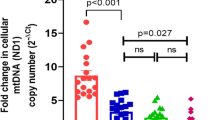
Inflammation and oxidative stress are intrinsically linked to early poor placentation, typical of pregnancies complicated by preeclampsia associated with intrauterine growth restriction (PE-IUGR). Low mitochondrial DNA copy number (mtDNAcn) in peripheral blood constitutes a good peripheral surrogate marker of inflammation and oxidative stress. On these basis, we explored a possible correlation between mtDNAcn in peripheral blood in the first trimester of pregnancy and the PE-IUGR onset. To shed light on this issue, we setup a nested case-control study from a prospective cohort of pregnant women undergoing firsttrimester aneuploidies screening. Two groups of patients affected by PE classified according to the clinical phenotype were identified: (1) patients who developed PE-IUGR and (2) patients who developed PE associated with appropriate for gestational age intrauterine fetal growth (PE-AGAf). Controls were women with a physiologic pregnancy matched to cases on the basis of age (+6 months, ratio 2:1). Mitochondrial DNA copy number was quantified using real-time polymerase chain reaction and normalized to nuclear DNA. The median (interquartile range) mtDNAcn in peripheral blood in patients with PE-IUGR (n = 12) and in patients with PE-AGAf (n = 16) was 70 (44–97) and 108 (95–145), respectively (P =.004). Both these values were significantly lower than that detected in the control group (161[133-183], P <.001). The area under the receiver-operator curve for PE-IUGR and PE-AGAf were 0.94 (95% confidence interval [CI]: 0.88-1.00, P <.001) and 0.81 (95%CI: 0.70-0.91, P <.001), respectively. In conclusion, MtDNAcn in peripheral blood resulted significantly lower both in patients affected by PE-IUGR and in those affected by PE-AGAf when compared to controls. The accuracy of this biomarker resulted particularly good in predicting PE-IUGR.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Myatt L, Redman CW, Staff AC, et al. Strategy for standardization of preeclampsia research study design. Hypertension. 2014;63(6):1293–1301.
Conde-Agudelo A, Belizán JM. Risk factors for pre-eclampsia in a large cohort of Latin American and Caribbean women. BJOG. 2000;107(1):75–83.
Ye C, Ruan Y, Zou L, et al. The 2011 survey on hypertensive disorders of pregnancy (HDP) in China: prevalence, risk factors, complications, pregnancy and perinatal outcomes. PLoS One. 2014;9(6):e100180.
Mol BWJ, Roberts CT, Thangaratinam S, Magee LA, de Groot CJM, Hofmeyr GJ. Pre-eclampsia. Lancet. 2016;387(10022):999–1011.
Levine RJ, Lam C, Qian C, et al. Soluble endoglin and other circulating antiangiogenic factors in preeclampsia. N Engl J Med. 2006;355(10):992–1005.
Chappell LC, Duckworth S, Seed PT, et al. Diagnostic accuracy of placental growth factor in women with suspected preeclampsia: a prospective multicenter study. Circulation. 2013;128(19):2121–2131.
Redman CW, Sargent IL, Staff AC. IFPA Senior Award Lecture: making sense of pre-eclampsia-two placental causes of preeclampsia? Placenta. 2014;35(suppl):S20–S25.
Ferrazzi E, Zullino S, Stampalija T, et al. Bedside diagnosis of two major clinical phenotypes of hypertensive disorders of pregnancy. Ultrasound Obstet Gynecol. 2016;48(2):224–231.
Thilaganathan B. Association of higher maternal blood pressure with lower infant birthweight: placental cause or cardiovascular effect? Hypertension. 2016;67(3):499–500.
Ferrazzi E, Stampalija T, Monasta L, Di Martino D, Vonck S, Gyselaers W. Maternal hemodynamics: a method to classify hypertensive disorders of pregnancy. Am J Obstet Gynecol. 2018;218(1):124.e1–124.e11.
McCarthy C, Kenny LC. Therapeutically targeting mitochondrial redox signalling alleviates endothelial dysfunction in preeclampsia. Sci Rep. 2016;6:32683.
Bilodeau JF. Review: maternal and placental antioxidant response to preeclampsia-impact on vasoactive eicosanoids. Placenta. 2014;35(suppl):S32–S38.
Burton GJ, Woods AW, Jauniaux E, Kingdom JC. Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta. 2009;30(6):473–482.
Rolnik DL, Wright D, Poon LC, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377(7):613–622.
Stampalija T, Monasta L, Di Martino DD, et al. The association of first trimester uterine arteries doppler velocimetry with different clinical phenotypes of hypertensive disorders of pregnancy: a longitudinal study. J Matern Fetal Neonatal Med. 2017;20:1–9.
Knez J, Marrachelli VG, Cauwenberghs N, et al. Peripheral blood mitochondrial DNA content in relation to circulating metabolites and inflammatory markers: a population study. PLoS One. 2017; 12(7):e0181036.
Lee J, Lee D, Im J, Lee J. Mitochondrial DNA copy number in peripheral blood is independently associated with visceral fat accumulation in healthy young adults. Int J Endocrinol. 2014;2014:586017.
Knez J, Winckelmans E, Plusquin M, et al. Correlates of peripheral blood mitochondrial DNA content in a general population. Am J Epidemiol. 2016;183(2):138–146.
Ballinger SW, Patterson C, Knight-Lozano CA, et al. Mitochondrial integrity and function in atherogenesis. Circulation. 2002;106(5):544–549.
Yakes F, Van Houten B. Mitochondrial DNA damage is more extensive and persists longer than nuclear DNA damage in human cells following oxidative stress. Proc Natl Acad Sci USA. 1997;94(2):514–519.
Liu LP, Cheng K, Ning MA, et al. Association between peripheral blood cells mitochondrial DNA content and severity of coronary heart disease. Atherosclerosis. 2017;261:105–110.
Ashar FN, Zhang Y, Longchamps RJ, et al. Association of mitochondrial DNA copy number with cardiovascular disease. JAMA Cardiol. 2017;2(11):1247–1255.
Kim JY, Choi JR, Park IH, et al. A prospective study of leucocyte mitochondrial DNA content and deletion in association with the metabolic syndrome. Diabetes Metab. 2017;43(3):280–283.
Révész D, Verhoeven JE, Picard M, et al. Associations between cellular aging markers and metabolic syndrome: findings from the CARDIA study. J Clin Endocrinol Metab. 2018;103(1):148–157.
Huang CH, Su SL, Hsieh MC, et al. Depleted leukocyte mitochondrial DNA copy number in metabolic syndrome. J Atheroscler Thromb. 2011;18(10):867–873.
Tranquilli AL, Dekker G, Magee L, et al. The classification, diagnosis and management of the hypertensive disorders of pregnancy: a revised statement from the ISSHP. Pregnancy Hypertens. 2014;4(2):97–104.
Società Italiana di Ecografia Ostetrica e Ginecologica (SIEOG). CIC, Editor Guidelines on Ultrasound Screening in Obstetrics and Gynecology. Rome, Italy: Internazionali; 1996.
Colleoni F, Lattuada D, Garretto A, et al. Maternal blood mitochondrial DNA content during normal and intrauterine growth restricted (IUGR) pregnancy. Am J Obstet Gynecol. 2010;203(4):365.e1–365.e6.
Unal I. Defining an optimal cut-point value in ROC analysis: an alternative approach. Comput Math Methods Med. 2017;2017:3762651.
Steegers EA, Dadelszen von P, Duvekot JJ, Pijnenborg R. Preeclampsia. Lancet. 2010;376(9741):631–644.
Erez O, Romero R, Maymon E, et al. The prediction of late-onset preeclampsia: results from a longitudinal proteomics study. PLoS One. 2017;12(7):e0181468.
Lees C, Ferrazzi E. Relevance of haemodynamics in treating pre-eclampsia. Curr Hypertens Rep. 2017;19(9):76.
Ferrazzi E. Is it the case to dismiss maternal metabolic syndrome as a key co-factor in pre-eclampsia occurring predominantly late in gestation? Placenta. 2015;36(4):467–468.
Poidatz D, Dos Santos E, Gronier H, et al. Trophoblast syncytialisation necessitates mitochondrial function through estrogenrelated receptor-g activation. Mol Hum Reprod. 2015;21(2):206–216.
Poidatz D, Dos Santos E, Duval F, et al. Involvement of estrogenrelated receptor-g and mitochondrial content in intrauterine growth restriction and preeclampsia. Fertil Steril. 2015;104(2):483–490.
Furui T, Kurauchi O, Tanaka M, Mizutani S, Ozawa T, Tomoda Y. Decrease in cytochrome c oxidase and cytochrome oxidase subunit I messenger RNA levels in preeclamptic pregnancies. Obstet Gynecol. 1994;84(2):283–288.
Torbergsen T, Oian P, Mathiesen E, Borud O. Pre-eclampsia-a mitochondrial disease? Acta Obstet Gynecol Scand. 1989;68(2):145–148.
Folgerø T, Storbakk N, Torbergsen T, Øian P. Mutations in mitochondrial transfer ribonucleic acid genes in preeclampsia. Am J Obstet Gynecol. 1996;174(5):1626–1630.
Vuorinen K, Remes A, Sormunen R, Tapanainen J, Hassinen IE. Placental mitochondrial DNA and respiratory chain enzymes in the etiology of preeclampsia. Obstet Gynecol. 1998;91(6):950–955.
Vishnyakova PA, Volodina MA, Tarasova NV, et al. Mitochondrial role in adaptive response to stress conditions in preeclampsia. Sci Rep. 2016;6:32410.
Mandò C, De Palma C, Stampalija T, et al. Placental mitochondrial content and function in intrauterine growth restriction and preeclampsia. Am J Physiol Endocrinol Metab. 2014;306(4):E404–E413.
Qiu C, Hevner K, Enquobahrie DA, Williams MA. A casecontrol study of maternal blood mitochondrial DNA copy number and preeclampsia risk. Int J Mol Epidemiol Genet. 2012;3(3):237–244.
Díaz M, Aragonés G, Sánchez-Infantes D, et al. Mitochondrial DNA in placenta: associations with fetal growth and superoxide dismutase activity. Horm Res Paediatr. 2014;82(5):303–309.
Fragouli E, Spath K, Alfarawati S, et al. Altered levels of mitochondrial DNA are associated with female age, aneuploidy, and provide an independent measure of embryonic implantation potential. PLoS Genet. 2015;11(6):e1005241.
Ravichandran K, McCaffrey C, Grifo J, et al. Mitochondrial DNA quantification as a tool for embryo viability assessment: retrospective analysis of data from single euploid blastocyst transfers. Hum Reprod. 2017;32(6):1282–1292.
Wang T, Zhang M, Jiang Z, Seli E. Mitochondrial dysfunction and ovarian aging. Am J Reprod Immunol. 2017;77(5):e12651.
Barnes FL, Victor AR, Zouves CG, Viotti M. Mitochondrial DNA quantitation-making sense of contradictory reports. Hum Reprod. 2017;32(10):2149–2150.
Wells D, Ravichandran K, McCaffrey C, et al. Reply: mitochondrial DNA quantification-the devil in the detail. Hum Reprod. 2017;32(10):2150–2151.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Busnelli, A., Lattuada, D., Ferrari, S. et al. Mitochondrial DNA Copy Number in Peripheral Blood in the First Trimester of Pregnancy and Different Preeclampsia Clinical Phenotypes Development: A Pilot Study. Reproductive Sciences 26, 1054–1061 (2019). https://doi.org/10.1177/1933719118804410
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719118804410