Abstract.
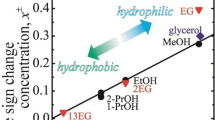
Recent experiments for various amides and sugars showed a clear correlation of the temperature dependence of the Soret coefficient with the hydrophilicity, quantitatively described by the logarithm of the 1-octanol/water partition coefficient log P . This coefficient is a measure for the hydrophilicity/hydrophobicity balance of a solute and is often used to model the transport of a compound in the environment or to screen for potential pharmaceutical compounds. In order to validate whether this concept works also for other water soluble molecules we investigated systematically the thermophoresis of mono- and polyhydric alcohols. As experimental method we use a holographic grating technique called infrared Thermal Diffusion Forced Rayleigh Scattering (IR-TDFRS). Experiments showed that the temperature dependence of the Soret coefficient of polyhydric alcohols also correlates with log P and lies on the same master plot as amides and sugars.
Graphical abstract

Similar content being viewed by others
References
M. Jerabek-Willemsen, T. Andre, R. Wanner, H.M. Roth, S. Duhr, P. Baaske, D. Breitsprecher, J. Mol. Struct. 1077, 101 (2014)
R. Piazza, S. Iacopini, B. Triulzia, Phys. Chem. Chem. Phys. 6, 1616 (2004)
S. Duhr, D. Braun, Proc. Natl. Acad. Sci. U.S.A. 103, 19678 (2006)
S. Iacopini, R. Piazza, Europhys. Lett. 63, 247 (2003)
Doreen Niether, Mona Sarter, Bernd König, Michaela Zamponi, Jörg Fitter, Andreas Stadler, Simone Wiegand, AIP Conf. Proc. 1929, 020001 (2018)
S. Alves, F.L.S. Cuppo, A. Bourdon, A.M. Figueiredo, J. Opt. Soc. Am. B 23, 2328 (2006)
M.P. Santos, S.L. Gomez, E. Bringuier, A.M.F. Neto, Phys. Rev. E 77, 011403 (2008)
B. Arlt, S. Datta, T. Sottmann, S. Wiegand, J. Phys. Chem. B 114, 2118 (2010)
Ph. Naumann, S. Datta, T. Sottmann, B. Arlt, H. Frielinghaus, S. Wiegand, J. Phys. Chem. B 118, 3451 (2014)
K. Maeda, N. Shinyashiki, S. Yagihara, S. Wiegand, R. Kita, J. Chem. Phys. 143, 124504 (2015)
Kazuya Eguchi, Doreen Niether, Simone Wiegand, Rio Kita, Eur. Phys. J. E 39, 86 (2016)
E. Lapeira, M.M. Bou-Ali, J.A. Madariaga, C. Santamaria, Microgravity Sci. Technol. 28, 553 (2016)
Doreen Niether, Silvia Di Lecce, Fernando Bresme, Simone Wiegand, Phys. Chem. Chem. Phys. 20, 1012 (2018)
D. Niether, H. Kriegs, J.K.G. Dhont, S. Wiegand, J. Chem. Phys. 149, 044506 (2018)
André, J. Phys. Chem. B 122, 4093 (2018)
Y. Kishikawa, S. Wiegand, R. Kita, Biomacromolecules 11, 740 (2010)
A. Königer, B. Meier, W. Köhler, Philos. Mag. 89, 907 (2009)
P. Polyakov, S. Wiegand, J. Chem. Phys. 128, 034505 (2008)
R. Sugaya, B.A. Wolf, R. Kita, Biomacromolecules 7, 435 (2006)
Doreen Niether, Dzmitry Afanasenkau, Jan K.G. Dhont, Simone Wiegand, Proc. Natl. Acad. Sci. U.S.A. 113, 4272 (2016)
D. Niether, T. Kawaguchi, J. Hovancova, K. Eguchi, J.K.G. Dhont, R. Kita, S. Wiegand, Langmuir 33, 8483 (2017)
I. Prigogine, L. Debrouckere, R. Amand, Physica 16, 851 (1950)
J. Luettmer-Strathmann, Int. J. Thermophys. 26, 1693 (2005)
B. Rousseau, C. Nieto-Draghi, J.B. Avalos, Europhys. Lett. 67, 976 (2004)
P.A. Artola, B. Rousseau, Phys. Rev. Lett. 98, 125901 (2007)
Robert Byron Bird, Warren E. Stewart, Edwin N. Lightfoot, Transport Phenomena, revised second edition (Wiley, New York, 2007)
C.R. Wilke, Pin Chang, AlChE J. 1, 264 (1955)
Myo T. Tyn, Waclaw F. Calus, J. Chem. Eng. Data 20, 310 (1975)
W. Hayduk, B.S. Minhas, Can. J. Chem. Eng. 60, 295 (1982)
Robert Evans, Zhaoxia Deng, Alexandria K. Rogerson, Andy S. McLachlan, Jeff J. Richards, Mathias Nilsson, Gareth A. Morris, Angew. Chem. Int. Ed. 52, 3199 (2013)
A. Gierer, K. Wirtz, Z. Naturforsch. A: Phys. Sci. 8, 532 (1953)
A. Becker, W. Köhler, B. Müller, Ber. Bunsenges. Phys. Chem. Chem. Phys. 99, 600 (1995)
S. Wiegand, H. Ning, H. Kriegs, J. Phys. Chem. B 111, 14169 (2007)
G. Wittko, W. Köhler, Philos. Mag. 83, 1973 (2003)
L.J. Tichacek, W.S. Kmak, H.G. Drickamer, J. Phys. Chem. 60, 660 (1956)
P. Kolodner, H. Williams, C. Moe, J. Chem. Phys. 88, 6512 (1988)
R. Kita, S. Wiegand, J. Luettmer-Strathmann, J. Chem. Phys. 121, 3874 (2004)
G.H. Großmann, K.H. Ebert, Ber. Bunsenges. Phys. Chem. Chem. Phys. 85, 1026 (1981)
W. Köhler, R. Schäfer, Polymer analysis by thermal-diffusion forced rayleigh scattering, in New Developments in Polymer Analytics II, edited by M. Schmidt, Advances in Polymer Science, Vol. 151 (Springer, Berlin, 2000) pp. 1--59
A. Bondi, J. Phys. Chem. 68, 441 (1964)
Gerardino D'Errico, Ornella Ortona, Fabio Capuano, Vincenzo Vitagliano, J. Chem. Eng. Data 49, 1665 (2004)
Jonathan T. Su, P. Brent Duncan, Amit Momaya, Arimatti Jutila, David Needham, J. Chem. Phys. 132, 044506 (2010)
John George, Nandhibatla V. Sastry, J. Chem. Eng. Data 48, 1529 (2003)
N.G. Tsierkezos, I.E. Molinou, J. Chem. Eng. Data 43, 989 (1998)
Fong-Meng Pang, Chye-Eng Seng, Tjoon-Tow Teng, M.H. Ibrahim, J. Mol. Liq. 136, 71 (2007)
C.A. Lipinski, F. Lombardo, B.W. Dominy, P.J. Feeney, Adv. Drug Deliv. Rev. 46, 3 (2001)
Vellarkad N. Viswanadhan, Arup K. Ghose, Ganapathi R. Revankar, Roland K. Robins, J. Chem. Inf. Comput. Sci. 29, 163 (1989)
Gilles Klopman, Ju-Yun Li, Shaomeng Wang, Mario Dimayuga, J. Chem. Inf. Comput. Sci. 34, 752 (1994)
Marvin 16.5.2.0, 2016, ChemAxon (Calculator Plugins for structure property prediction and calculation (of $\log P$)), http://www.chemaxon.com
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
The EPJ Publishers remain neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Bjelčić, M., Niether, D. & Wiegand, S. Correlation between thermophoretic behavior and hydrophilicity for various alcohols⋆. Eur. Phys. J. E 42, 68 (2019). https://doi.org/10.1140/epje/i2019-11831-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epje/i2019-11831-x