Abstract
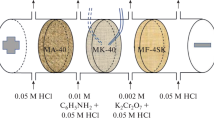
Surface-modified cation exchange materials are obtained based on industrial MK-40 heterogeneous and MF-4SK homogeneous cation-exchange membranes by in situ oxidative polymerization of aniline under electrodialysis conditions. The conduction and diffusion characteristics of the initial and modified membranes in solutions of sulfuric acid and nickel sulfate are studied. It is shown that the modification of the membranes with polyaniline leads to a decrease in their electrical conductivity and diffusion permeability without sacrificing high selectivity. The diffusion permeability of the cation-exchange membranes is higher in solutions of nickel sulfate in comparison with solutions of sulfuric acid, while an inverse dependence is found for anion-exchange membranes. The competitive transport of sulfuric acid and nickel sulfate during electrodialysis separation and concentration of their mixture using initial commercial and modified cation-exchange membranes paired with an MA-41 anion-exchange membrane is studied. It is shown that applying a layer of polyaniline with positively charged groups onto one of the surfaces of MK-40 or MF-4SK cation-exchange membranes leads to a decrease in the transport of a doubly charged nickel cation both in the separation and concentration modes over the entire range of current densities. The highest repulsion effect is observed in the case of the use of homogeneous modified membranes, where the selective permeability coefficient P(H2SO4/NiSO4) increases from 0.7–1.7 up to 32.5–19.7 depending on the current density. It is found that the use of surface-modified with polyaniline cation-exchange membranes makes it possible to concentrate a solution containing 0.1 mol-equiv/L (4.9 g/L) H2SO4 and 0.1 mol-equiv/L (7.7 g/L) NiSO4 with simultaneous separation to sulfuric acid with a concentration of about 2.4 mol-equiv/L (120 g/L) and a solution of nickel sulfate. Here, the concentration of nickel sulfate in the concentrate does not exceed 0.13 mol-equiv/L (10 g/L).














Similar content being viewed by others
REFERENCES
On the State and Use of Mineral Resources of the Russian Federation in 2020: State Report, Ed. by E. I. Petrov and D. D. Teten’ka (FGBU VIMS, 2021). Applied March 5, 2023. www.rosnedra.gov.ru/data/Files/File/7992.pdf.
A. Abidli, Y. Huang, Z. Ben Rejeb, et al., Chemosphere 292, 133102 (2022).
S. Rajoria, M. Vashishtha, and V. K. Sangal, Environ. Sci. Pollut. Res. 29, 72196 (2022).
A. Rawat, A. Srivastava, A. Bhatnagar, and A. K. Gupta, J. Cleaner Prod. 383, 135382 (2023).
X. Yu, Y. Hou, X. Ren, et al., J. Water Proc. Eng. 46, 102577 (2022).
S. Li, M. Dai, I. Ali, et al., Process Safety Environ. Prot. 172, 417 (2023).
C. Li, G. Dai, R. Liu, et al., Sep. Purif. Technol. 306, 122559 (2023).
L. Cassayre, B. Guzhov, M. Zielinski, and B. Biscans, Renewable Sustain. Energy Rev. 170, 112983 (2022).
K. Yan, P. Huang, M. Xia, et al., Sep. Purif. Technol. 295, 121283).
V. A. Shaposhnik and K. Kesore, J. Membr. Sci. 136, 35 (1997).
T. Xu, J. Membr. Sci. 263, 1.
A. Campione, L. Gurreri, M. Ciofalo, et al., Desalination 434, 121 (2018).
T. Benvenuti, M. A. S. Rodrigues, A. M. Bernardes, and J. Zoppas-Ferreira, J. Clean. Prod. 155, 130 (2017).
S. Melnikov, N. Sheldeshov, V. Zabolotsky, et al., Sep. Purif. Technol. 189, 74 (2017).
A. Achoh, I. Petriev, and S. Melnikov, Membranes 11, 980 (2021).
M. Sedighi, M. M. B. Usefi, A. F. Ismail, and M. Ghasemi, Desalination 549, 116319 (2023).
M. Reig, C. Valderrama, O. Gibert, and J. L. Cortina, Desalination 399, 88 (2016).
M. Ahmad, M. Ahmed, S. Hussain, et al., Desalination 545, 116159 (2023).
D. V. Golubenko, A. D. Manin, Y. Wang, T. Xu, A. B. Yaroslavtsev, Desalination 531, 115719 (2022).
S. Zhang, S. Wang, Z. Guo, et al., Sep. Purif. Technol. 300, 121926 (2022).
W. Wang, G. Hong, Y. Zhang, et al., J. Membr. Sci. 675, 121534 (2023).
J. Yan, H. Wang, H. Yan, et al., Desalination 554, 116513 (2023).
I. A. Stenina, P. A. Yurova, L. Novak, et al., Colloid Polym. Sci. 299, 719 (2021).
I. Stenina, P. Yurova, A. Achoh, et al., Polymers (Basel) 15, 647 (2023).
W. Zhang, M. Miao, and J. Pan, Desalination 411, 28 (2017).
V. I. Zabolotsky, A. R. Achoh, K. A. Lebedev, and S. S. Melnikov, J. Membr. Sci. 608, 118152 (2020).
V. D. Grebenyuk, B. K. Veisov, R. D. Chebotareva, et al., Zh. Prikl. Khim. 59, 916 (1986).
N. P. Gnusin and O. A. Demina, Theor. Found. Chem. Eng. 40, 27 (2006).
V. I. Zabolotskii, A. V. Demin, and O. A. Demina, Russ. J. Electrochem. 47, 327 (2011).
V. I. Zabolotskii, K. V. Protasov, and M. V. Sharafan, Russ. J. Electrochem. 46, 979 (2010).
V. I. Zabolotskii, V. F. Pis’menskii, O. A. Demina, and L. Novak, Russ. J. Electrochem. 49, 563 (2013).
S. S. Melnikov, O. A. Mugtamov, and V. I. Zabolotsky, Sep. Purif. Technol. 235, 116198 (2020).
A. V. Demin and V. I. Zabolotskii, Russ. J. Electrochem. 44, 1058 (2008).
E. V. Nazyrova, N. A. Kononenko, S. A. Shkirskaya, and O. A. Demina, Membr. Membr. Technol. 4, 145 (2022).
N. Berezina, N. Gnusin, O. Dyomina, and S. Timofeyev, J. Membr. Sci. 86, 207 (1994).
H. L. Yeager, B. O' Dell, and Z. Twardowski, J. Electrochem. Soc. 129, 85 (1982).
V. I. Zabolotskii, A. A. Shudrenko, and N. P. Gnusin, Elektrokhimiya 24, 744 (1988).
N. P. Berezina, S. A. Shkirskaya, M. V. Kolechko, et al., Russ. J. Electrochem. 47, 995 (2011).
K. V. Protasov, S. A. Shkirskaya, N. P. Berezina, and V. I. Zabolotskii, Russ. J. Electrochem. 46, 1131 (2010).
V. V. Kotov and V. A. Shaposhnik, Kolloid. Zh. 46, 1116 (1984).
T. Luo, S. Abdu, and M. Wessling, J. Membr. Sci. 555, 429 (2018).
I. Falina, N. Loza, S. Loza, et al., Membranes 11, 227 (2021).
T. Sata, T. Sata, and W. Yang, J. Membr. Sci. 206, 31 (2002).
H. Farrokhzad, S. Darvishmanesh, G. Genduso, et al., Electrochim. Acta 158, 64 (2015).
M. Kumar, M. A. Khan, AZ. Alothman, and M. R. Siddiqui, Desalination 325, 95 (2013).
M. Reig, H. Farrokhzad, B. Van der Bruggen, et al., Desalination 375, 1 (2015).
N. V. Loza, S. A. Loza, and N. A. Kononenko, RF Patent 2566415, 2015.
M. Andreeva, N. Loza, N. Kutenko, and N. Kononenko, J. Solid State Electrochem. 24, 101 (2020).
N. P. Berezina, N. A. Kononenko, O. A. Dyomina, and N. P. Gnusin, Adv. Colloid Interface Sci. 139, 3 (2008).
V. I. Zabolotsky, N. D. Pismenskaya, E. V. Laktionov, and V. V. Nikonenko, Desalination 107, 245 (1996).
V. I. Zabolotskii, S. S. Mel’nikov, and O. A. Demina, Russ. J. Electrochem. 50, 32 (2014).
M. A. Andreeva, N. V. Loza, N. D. Pis’menskaya, et al., Membranes 10, 145 (2020).
F. Li, Y. Jia, J. He, and M. Wang, J. Cleaner Prod. 320, 128760 (2021).
Y. Lorrain, G. Pourcelly, and C. Gavach, J. Membr. Sci. 110, 181 (1996).
M. Liu, J. Wang, J. Liu, et al., Polymer 268, 125721 (2023).
N. P. Gnusin, N. P. Berezina, N. A. Kononenko, et al., Russ. J. Phys. Chem. A 83, 107 (2009).
O. A. Demina, N. A. Kononenko, and I. V. Falina, Pet. Chem. 54, 515 (2014).
V. I. Zabolotskii and V. V. Nikonenko, Ion Transport in Membranes (Nauka, Moscow, 1996) [in Russian].
S. A. Shkirskaya, I. N. Senchikhin, N. A. Kononenko, and V. I. Roldugin, Russ. J. Electrochem. 53, 78 (2017).
N. V. Loza, S. V. Dolgopolov, N. A. Kononenko, M. A. Andreeva, and Yu. S. Korshikova, Russ. J. Electrochem. 51, 538 (2015).
M. A. Andreeva, N. V. Loza, N. D. Pis’menskaya, L. Dammak, and C. Larchet, Membranes 10, 145 (2020).
Funding
The study was financially supported by the Russian Foundation for Basic Research (grant no. 18-38-20069 mol_a_ved).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by E. Boltukhina
Rights and permissions
About this article
Cite this article
Loza, S.A., Romanyuk, N.A., Falina, I.V. et al. Electrodialysis Separation and Selective Concentration of Sulfuric Acid and Nickel Sulfate Using Membranes Modified with Polyaniline. Membr. Membr. Technol. 5, 236–256 (2023). https://doi.org/10.1134/S2517751623040030
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2517751623040030