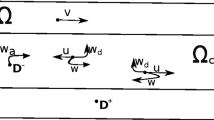
Abstract
Contemporary cell investigation methods permit us to describe the molecular transport mechanisms of intracellular substances and cell components. Analysis of the completeness and the consistency of the existing transport network data and their utilization is an issue for mathematical modeling. Self-organization of the cell transport network and transport of the cargo and organelles upon it are modeled in this work by examples of the mitotic spindle formation, retrograde axonal transport, and lipid transport.
Similar content being viewed by others
References
K. Takahashi, S. N. Arjunan, and M. Tomita, “Space in systems biology of signaling pathways–to-wards intracellular molecular crowding in silico,” FEBS Lett. 579, 1783–1788 (2005).
U. F. Greber and M. Way, “A superhighway to virus infection,” Cell 124, 741–754 (2006).
T. Hirase and K. Node, “Endothelial dysfunction as a cellular mechanism for vascular failure,” Am. J. Physiol. Heart Circ. Physiol. 303, 499–505 (2012).
E. A. Perez, “Microtubule inhibitors: Differentiating tubulin-inhibiting agents based on mecha-nisms of action, clinical activity, and resistance,” Mol. Cancer Therap. 8, 2086–2095 (2009).
J. Avila, J. J. Lucas, M. Perez, and F. Hernandez, “Role of tau protein in both physiological and pathological conditions,” Physiol. Rev. 84, 361–384 (2004).
I. Vorobjev, V. Malikov, and V. Rodionov, “Self-organization of a radial microtubule array by dynein-dependent nucleation of microtubules,” Proc. Natl. Acad. Sci. USA 98, 10160–10165 (2001).
V. Rodionov, E. N. Cytrynbaum, and A. Mogilner, “Computational model of dynein dependent self-organization of microtubule asters,” J. Cell Sci. 117, 1381–1397 (2004).
N. A. Kolchanov, Computational System Biology (Sib. Otdel. RAN, Novosibirsk, 2008) [in Russian].
B. N. Kholodenko, “Cell-signalling dynamics in time and space,” Nature Rev. Mol. Cell Biol. 7, 165–176 (2006).
A. Cangiani and R. Natalini, “A spatial model of cellular molecular trafficking including active transport along microtubules,” J. Theor. Biol. 267, 614–625 (2010).
C. Lemerle, B. di Ventura, and L. Serrano, “Space as the final frontier in stochastic simulations of biological systems,” FEBS Lett. 579, 1789–1794 (2005).
A. V. Kuznetsov, “Numerical investigation of axonal cargo rerouting in a dendrite: A three kinetic state model,” Int. J. Numer. Methods Biomed. Eng. 29, 428–443 (2013).
M. Tomita, “Whole-cell simulation: a grand challenge of the 21st century,” Trends Biotechnol. 19, 205–210 (2001).
C. Collinet, M. Stöter, C. R. Bradshaw, N. Samusik, J. C. Rink, D. Kenski, B. Habermann, F. Buchholz, R. Henschel, M. S. Mueller, W. E. Nagel, E. Fava, Y. Kalaidzidis, and M. Zerial, “Systems survey of endocytosis by multiparametric image analysis,” Nature 464, 343–350 (2010).
B. Cui, C. Wu, L. Chen, A. Ramirez, E. L. Bearer, W. P. Li, W. C. Mobley, and S. Chu, “One at a time, live tracking of NGF axonal transport using quantum dots,” Proc. Natl. Acad. Sci. USA 104, 13666–13671 (2007).
H. Lodish, Molecular Cell Biology (W. H. Freeman, New York, 2007).
B. Alberts, Molecular Biology of the Cell (Garland Science, New York, 2002).
E. Schulze and M. Kirschner, “Dynamic and stable populations of microtubules in cells,” J. Cell Biol. 104, 277–288 (1987).
D. van Damme, K. van Poucke, E. Boutant, C. Ritzenthaler, D. Inzé, and D. Geelen, “In vivo dynamics and differential microtubule-binding activities of map65 proteins,” Plant Physiol. 136, 3956–3967 (2004).
H. Chen, J. Yang, P. S. Low, and J. X. Cheng, “Cholesterol level regulates endosome mobility via rab proteins,” Biophys. J. 94, 1508–1520 (2007).
J. Huotari and A. Helenius, “Endosome maturation,” The EMBO J. 30, 3481–3500 (2011).
J. A. Wattis, B. O’Malley, H. Blackburn, L. Pickersgill, J. Panovska, H. M. Byrne, and K. G. Jackson, “Mathematical model for low density lipoprotein (ldl) endocytosis by hepatocytes,” Bull. Math. Biol. 70, 2303–2333 (2008).
R. Paul, R. Wollman, W. T. Silkworth, I. K. Nardi, D. Cimini, and A. Mogilner, “Computer simulations predict that chromosome movements and rotations accelerate mitotic spindle assembly without compromising accuracy,” Proc. Natl. Acad. Sci. USA 106, 15708–15713 (2009).
T. E. Holy and S. Leibler, “Dynamic instability of microtubules as an efficient way to search in space,” Proc. Natl. Acad. Sci. USA 91, 5682–5685 (1994).
C. B. O’Connell and A. L. Khodjakov, “Cooperative mechanisms of mitotic spindle formation,” J. Cell Sci. 120, 1717–1722 (2007).
R. Wollman, E. N. Cytrynbaum, J. T. Jones, T. Meyer, J. M. Scholey, and A. Mogilner, “Efficient chromosome capture requires a bias in the 'search-and-capture' process during mitotic-spindle assembly,” Curr. Biol. 15, 828–832 (2005).
P. del Conte-Zerial, L. Brusch, J. C. Rink, C. Collinet, Y. Kalaidzidis, M. Zerial, and A. Deutsch, “Membrane identity and GTPase cascades regulated by toggle and cut-out switches,” Mol. Syst. Biol. 4, 206 (2008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © K.A. Novikov, A.A. Romanyukha, A.N. Gratchev, J.G. Kzhyshkowska, O.A. Melnichenko, 2015, published in Matematicheskoe Modelirovanie, 2015, Vol. 27, No. 3, pp. 49–62.
Rights and permissions
About this article
Cite this article
Novikov, K.A., Romanyukha, A.A., Gratchev, A.N. et al. Mathematical model of cellular transport network self-organization and functioning. Math Models Comput Simul 7, 475–484 (2015). https://doi.org/10.1134/S2070048215050099
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070048215050099