Abstract
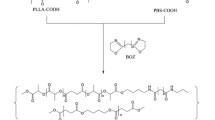
The paper describes some physical properties of poly(ether-amide)s (PEAs) prepared by solution polycondensation reaction of ether-amines such as 2,7-bis(4-aminophenoxy) naphthalene, 2,2-bis[4-(4-aminophenoxy)phenyl] propane, 1,4-bis(4-aminophenoxy methyl) cyclohexane and isophthalic acid. Resulting PEAs are high yield and have inherent viscosity ranging between 0.68–0.75 dL/g. The etheramines containing wholly, semi aromatic and aliphatic-aromatic segments were prepared by using two steps reaction. FTIR, 1H NMR, and elemental analyses were used for characterization of synthetic ether-amines and poly(ether-amide)s. PEAs films were prepared by solution casting technique then their thermal stability and mechanical properties were measured. TGA in nitrogen atmosphere shows that the PEAs are thermally stability, so 10% weight will be lost in the range of 335–358°C. Tensile strength and elongation at break of the PEA films ranged from 71.1–65.0 MPa and 6.48–8.41%, respectively.
Similar content being viewed by others
References
J. Lin and D. Sherrington, “Recent Developments in the Synthesis, Thermostability and Liquid Crystal Properties of Aromatic Polyamides”, in Polymer Synthesis. Advances in Polymer Science (Springer, Berlin, Heidelberg, 1994), Vol. 111, pp. 177–219.
P. E. Cassidy, Thermally Stable Polymers: Synthesis and Properties (Marcel Dekker, New York, 1980), pp. 110–116.
H. H Yang, Aromatic High-Strength Fibers (Wiley and Sons, New York, 1989).
R. B. Seymour and C. E. Carraher, Polymer Chemistry: an Introduction (Marcel Dekker, New York, 1981).
K. Faghihi, A. Raeisi, and H. Ardeshir Geravi, Fibers Polym. 18, 1 (2017).
C.-P. Yang and J.-H. Lin, J. Polym. Sci., Part A: Polym. Chem. 34, 341 (1996).
S. Mehdipour-Ataei and H. Heidari, Macromol. Symp. 193, 159 (2003).
S.-H. Hsiao and K.-H. Lin, Polymer 45, 7877 (2004).
H. S. Lee and S. Y. Kim, Macromol. Rapid Commun. 23, 665 (2002).
A. Shockravi, S. Mehdipour-Ataei, E. Abouzari-Lotf, and A. Yousefi, Eur. Polym. J. 42, 133 (2006).
K. Faghihi, M. Hajibeygi, and M. Shabanian, Macromol. Res. 17, 739 (2009).
K. Faghihi, M. Hajibeygi, and M. Shabanian, J. Macromol. Sci., Part A: Pure Appl. Chem. 47, 144 (2010).
C. D. Diakoumakos and J. A. Mikroyannidis, Polymer 35, 1986 (1994).
K. Faghihi, M. Hajibeygi, and M. Shabanian, Macromol. Res. 18, 421 (2010).
K. Faghihi, M. Shabanian, and M. Hajibeygi, Macromol. Res. 17, 912 (2009).
S. Mallakpour and E. Kowsari, Polym. Bull. 53, 169 (2005).
M. Shabanian, K. Faghihi, A. Raeisi, M. Varvanifarahani, H. A. Khonakdar, and U. Wagenknecht, J. Therm. Anal. Calorim. 117, 293 (2014).
N. Yamazaki and F. Higashi, J. Polym. Sci., Part C: Polym. Lett. 12, 185 (1974).
N. Yamazaki, M. Matsumoto, and F. Higashi, J. Polym. Sci., Polym. Chem. Ed. 13, 1373 (1975).
I. K. Varma, R. Kumar, and A. B. Bhattacharyya, J. Appl. Polym. Sci. 40, 531 (1990).
C. T. Vijayakumar, R. Surender, K. Rajakumar, and S. Alam, J. Therm. Anal. Calorim. 103, 693 (2011).
S. Maji and S. Banerjee, J. Membr. Sci. 349, 145 (2010).
A. Mirsamieia, K. Faghihia, and M. Shabanian, Polym. Adv. Technol. 28, 779 (2017).
D. H. Wang, J. J. Wie, K. M. Lee, T. J. White, and L.-S. Tan, Macromolecules 47, 659 (2014).
P. C. P. Watts, P. K. Fearon, W. K. Hsu, N. C. Billingham, H. W. Kroto, and D. R. M. Walton, J. Mater. Chem. 13, 491 (2003).
H. A. Shawky, J. Membr. Sci. 339, 209 (2009).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Mirsamiei, A., Faghihi, K. & Nanbedeh, S. Synthesis and Study of Poly(ether-amide)s Containing Aromatic and Aliphatic Ether-Amine Segment and Isophthalic Acid. Polym. Sci. Ser. B 60, 273–282 (2018). https://doi.org/10.1134/S1560090418030156
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090418030156