Abstract
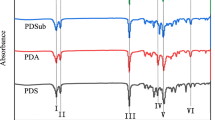
An aliphatic polyester based poly(ester amide)s (PEA) consisting of poly (L-lactic acid) and poly(butylene succinate) was successfully prepared via chain extension reaction of poly(L-lactic acid)-dicarboxylic acid (PLLA-COOH) and poly(butylene succinate)-dicarboxylic acid (PBS-COOH) using 2,2′-bis(2-oxazoline) as a chain extender. PLLA-COOH was obtained by direct polycondensation of L-lactic acid in the presence of 1, 4-succinic acid. PBS-COOH was synthesized by condensation polymerization of 1,4-butylene glycol with excessive succinic acid. The structures of PLLA-COOH, PBS-COOH, and PEAs were characterized by fourier transform infrared (FTIR) and 1H nuclear magnetic resonance (1H NMR). The molar masses were determined by gel permeation chromatography (GPC). The thermal properties of PLLA-COOH, PBS-COOH, and PEAs were characterized by thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC). The lattice parameters of PLLA-COOH, PBS-COOH, and PEAs were investigated by X-ray diffraction (XRD). Furthermore, The mechanical properties were characterized by tensile testing and notch Izod impact testing. The FTIR and 1H NMR results demonstrated the formation of PLLA-COOH, PBS-COOH, and PEAs. The GPC measurements showed that the molar masses of copolymer PEAs decreased with increasing PBS-COOH content. The TGA analysis confirmed that the introduction of PBS improved the thermal properties. DSC data indicated that the melting temperatures of the PEAs were lower than that of the prepolymers. The results of XRD suggested that the PLLA crystal structures was destroyed by the PBS units, and the crystallization of the PEAs mainly attributed to the PBS chain segments.The introduction of PBS units into the polymer structure improved the toughness of PLLA, which was detected in mechanical properties.

Similar content being viewed by others
References
J. Lunt, Polym Degrad. Stab., 59, 145 (1998).
M. J. Soares, P.-K. Dannecker, C. Vilela, J. Bastos, M. A. R. Meier, and A. F. Sousa, Eur. Polym. J., 90, 301 (2017).
K. M. Zia, A. Noreen, M. Zuber, S. Tabasum, and M. Mujahid, Int. J. Biol. Macromol., 82, 1028 (2016).
V. Nagarajan, K. Zhang, M. Misra, and A. K. Mohanty, ACS Appl. Mater. Interfaces, 7, 11203 (2015).
L. Sha, Z. Chen, Z. Chen, A. Zhang, and Z. Yang, Int. J. Polym. Sci., 2016, 1 (2016).
W. Zhong, J. Ge, and Z. Gu, J. Appl. Polym. Sci., 74, 2546 (2015).
R. M. Rasal, A. V. Janorkar, and D. E. Hirt, Prog. Polym. Sci., 35, 338 (2010).
L. Liu, J. Yu, L. Cheng, and W. Qu, Compos. Part A: Appl. Sci. Manuf., 40, 669 (2009).
R. Sinha, S. O. Kazuaki, and O. Masami, Macromolecules, 36, 2355 (2003).
N. Zhu, M. Ye, D. Shi, and M. Chen, Macromol. Res., 25, 165 (2017).
R. Supthanyakul, N. Kaabbuathong, and S. Chirachanchai, Polymer, 105, 1 (2016).
X. Zhang and Y. Zhang, Polymer, 140, 374 (2016).
J. B. Zeng, Y. D. Li, and W. D. Li, Ind. Eng. Chem. Res., 48, 1706 (2009).
J.-M. Raquez, Y. Habibi, M. Murariu, and P. Dubois, Prog. Polym. Sci., 38, 1504 (2013).
T. Gurunathan and S. K. Nayak, Polym. Adv. Technol., 27, 1484 (2016).
M. Pluta, J. Bojda, E. Piorkowska, M. Murariu, L. Bonnaud, and P. Dubois, Polym. Test., 61, 35 (2017).
Y. Zhou, L. Lei, B. Yang, J. Li, and J. Ren, Polym. Test., 60, 78 (2017).
M. Ajioka, K. Enomoto, and K. Suzuki, J. Environ. Polym. Degrad., 3, 225 (1995).
V. Sedlarik, P. Kucharczyk, V. Kasparkova, J. Drbohlav, A. Salakova, and P. Saha, J. Appl. Polym. Sci., 116, 1597 (2010).
Q. Gao, P. Lan, and H. Shao, Polym. J., 34, 786 (2002).
A. Chuma, H. W. Horn, and W. C. Swope, J. Am. Chem. Soc., 130, 6749 (2008).
M. Ryner, S. A. Kajsa, and C. A. Albertsson, Macromolecules, 34, 3877 (2010).
M. S. And and A. Södergård, Macromolecules, 32, 6412 (2013).
Y. Zhao, X. Shuai, and C. A. Chuanfu, Chem. Mater., 15, 2836 (2003).
J. L. Eguiburu, J. Sanroman, and B. Mjf, Polymer, 36, 173 (1995).
A.-L. Goffin, E. Duquesne, S. Moins, M. Alexandre, and P. Dubois, Eur. Polym. J., 43, 4103 (2007).
M. Ju, F. Gong, S. Cheng, and Y. Gao, Int. J. Polym. Sci., 2011, 1 (2011).
J. Dai, H. Bai, Z. Liu, L. Chen, Q. Zhang, and Q. Fu, RSC Adv., 6, 17008 (2016).
X. Lu, L. Tang, L. Wang, J. Zhao, D. Li, Z. Wu, and P. Xiao, Polym. Test., 54, 90 (2016).
R. Ouhib, B. Renault, H. Mouaziz, C. Nouvel, E. Dellacherie, and J.-L. Six, Carbohydr. Polym., 77, 32 (2009).
K. W. Kim and S. I. Woo, Macromol. Chem. Phys., 203, 2245 (2002).
S. I. Moon, C. W. Lee, and I. Taniguchi, Polymer, 42, 5059 (2001).
C. M. Lee, H. S. Kim, and J. S. Yoon, J. Appl. Polym. Sci., 95, 1116 (2005).
Y.-P. Song, D.-Y. Wang, X.-L. Wang, L. Lin, and Y.-Z. Wang, Polym. Adv. Technol., 22, 2295 (2011).
C.-H. Ho, C.-H. Wang, C.-I. Lin, and Y.-D. Lee, Polymer, 49, 3902 (2008).
Y.-M. Corre, A. Maazouz, J. Reignier, and J. Duchet, Polym. Eng. Sci., 54, 616 (2014).
R. Zhang, K. Huang, S. Hu, Q. Liu, X. Zhao, and Y. Liu, Polym. Test., 63, 38 (2017).
I. Moura, R. Nogueira, V. Bounor-Legare, and A. V. Machado, Mater. Chem. Phys., 134, 103 (2012).
J. Tuominen and J. V. Seppala, Macromolecules, 33, 3530 (2000).
S. Liu, C. Li, J. Zhao, Z. Zhang, and W. Yang, Polymer, 52, 6046 (2011).
S. Ozlem, B. Iskin, G. Yilmaz, M. Kukut, J. Hacaloglu, and Y. Yagci, Eur. Polym. J., 48, 1755 (2012).
N. J. Sijbrandi, A. J. Kimenai, E. P. C. Mes, R. Broos, G. Bar, M. Rosenthal, Y. Odarchenko, D. A. Ivanov, P. J. Dijkstra, and J. Feijen, Macromolecules, 45, 3948 (2012).
Z. Ge, D. Wang, and Y. Zhou, Macromolecules, 42, 2903 (2009).
T. Yokohara and M. Yamaguchi, Eur. Polym. J., 44, 677 (2008).
L. Liu, J. Yu, L. Cheng, and X. Yang, Polym. Degrad. Stab., 94, 90 (2009).
S.-L. Li, F. Wu, Y. Yang, Y.-Z. Wang, and J.-B. Zeng, Polym. Adv. Technol., 26, 1003 (2015).
J. Zhou, Z. Jiang, Z. Wang, J. Zhang, J. Li, Y. Li, J. Zhang, P. Chen, and Q. Gu, RSC Adv., 3, 18464 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: This work was financially supported by the Opening Project of Key Laboratory for Palygorskite Science and Applied Technology of Jiangsu Province (HPK201605), Huaian Science and Technology project (HAS2015002), the Open Project Fund for Jiangsu Provincial Engineering Laboratory for Advanced Materials of Salt Chemical Industry (No. 2065031701) and A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Rights and permissions
About this article
Cite this article
Zou, J., Qi, Y., Su, L. et al. Synthesis and Characterization of Poly(ester amide)s Consisting of Poly(L-lactic acid) and Poly(butylene succinate) Segments with 2,2′-Bis(2-oxazoline) Chain Extending. Macromol. Res. 26, 1212–1218 (2018). https://doi.org/10.1007/s13233-019-7018-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-019-7018-3