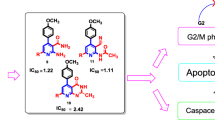
Abstract
A series of novel benzyl 5-(substitutedphenyl)-3-(2-oxo-2H-chromen-3-yl)-4,5-dihydro-1H-pyrazole-1-carbodithioate were obtained. The anti-cancer activity of synthesised compounds against lung cancer (A-549) and breast cancer cell lines (MDA-MB-231) was investigated by MTT assay. The most of the synthesised compounds were effective against both cancer cell lines and demonstrated significant cytotoxicity. Molecular docking investigation of the synthesised compounds and the cyclin dependent kinase-2 (CDK2) protein revealed that the synthesised compounds displayed greater C-score values, validating the findings.











Similar content being viewed by others
REFERENCES
Weir, H.K., Anderson, R.N., Coleman King, S.M., Soman, A., Thompson, T.D., Hong, Y., and Moller, B., Prev. Chronic. Dis., 2016, vol. 13, p. E157. https://doi.org/10.5888/pcd13.160211
Vallri, K.K., Nagaraju, P.V.V.S., Viswanath, I.V.K., and Singh, R.V., Russ. J. Org. Chem., 2020, vol. 56, no. 3, p. 524. https://doi.org/10.1134/S1070428020030252
Siegel, R.L., Miller, K.D., and Jemal, A., CA: A Cancer J. Clin., 2019, vol. 1, p. 7. https://doi.org/10.3322/caac.21551
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R.L., Torre, L.A., and Jemal, A., CA: Cancer J. Clin., 2018, vol. 68, p. 394. https://doi.org/10.3322/caac.21492
Sung, H., Ferlay, J., Siegel, R.L., Laversanne, M., Soerjomataram, I., Jemal, A., and Bray F., CA: Cancer J. Clin., 2021, vol. 71, p. 209. https://doi.org/10.3322/caac.21660
Zhang, J., Zhang, Z., Yang, B., Jing, X., Zhong, W., and Tang, H., Lat. Am. J. Pharm., 2016, vol. 35, p. 1719
Koleckar, V., Kubikova, K., Rehakova, Z., Kuca, K., Jun, D., Jahodar, L., and Opletal, L., Med. Chem., 2008, vol. 8, p. 436. https://doi.org/10.2174/138955708784223486
Dholaria, B., Hammond, W., Shreders, A., and Lou, Y., J. Hematol. Oncol., 2016, vol. 9, p. 138. https://doi.org/10.1186/s13045-016-0365-z
Hirsch, F.R., Scagliotti, G.V., Mulshine, J.L., Kwon, R., Curran, W.J., Wu, and Y.L., Paz-Ares, L., The Lancet., 2017, vol. 389, p. 299. https://doi.org/10.1016/S0140-6736(16)30958-8
Wood, S.L., Pernemalm, M., Crosbie, P.A., and Whetton, A.D., Cancer Treat. Rev., 2015, vol. 41, p. 361. https://doi.org/10.1016/j.ctrv.2015.02.008
Hill, A., Gotham, D., Fortunak, J., Meldrum, J., Erbacher, I., Martin, M., Shoman, H., Levi, J., Powderly, W.G., and Bower, M., BMJ Open., 2016, vol. 6, p. e009586-e009594. https://doi.org/10.1136/bmjopen-2015-009586
Zakowsky, M.F., Cancer Cytopathol., 2017, vol. 125, p. 470. https://doi.org/10.1002/cncy.21866
Sun, J.M., Hwang, D.W., Ahn, J.S., Ahn, M.J., and Park, K., Plos One., 2013, vol. 8, p. e64816-e64823. https://doi.org/10.1371/journal.pone.0064816
Gridelli, C. and Sacco, P.C., Curr. Opin. Oncol., 2016, vol. 28, p. 110. https://doi.org/10.1097/CCO.0000000000000266
Gronberg, B.H., Sundstrom, S., Kaasa, S., Bremnes, R.M., Flotten, O., Amundsen, T., Hjelde, H.H., Plessen, C., and Jordhoy, M., Eur. J. Cancer., 2010, vol. 46, p. 2225. https://doi.org/10.1016/j.ejca.2010.04.009
Yang, L., Zhou, Y., Li, Y., Zhou, J., Wu, Y., Cui, Y., Yang, G., and Hong, Y., Cancer Lett., 2015, vol. 357, p. 520. https://doi.org/10.1016/j.canlet.2014.12.003
Akki, M., Reddy, D.S., Katagi, K.S., Kumar, A., Devarajegowda, H.C., Sunitha Kumari, M., Babagond, V., Mane, S., and Joshi, S. D., J. Mol. Struct., 2022, vol. 1266, p. 133452. https://doi.org/10.1016/j.molstruc.2022.133452
Krishna, C., Bhargavi, M.V., Rao, Y.J., and Krupadanam, G.L.D., Russ. J. Gen. Chem., 2017, vol. 87, p. 1857. https://doi.org/10.1134/S1070363217080345
Reddy, D.S., Hosamani, K.M., Devarajegowda, H.C., and Kurjogi, M.M., RSC Adv., 2015, vol. 5, p. 64566. https://doi.org/10.1039/C5RA09508E
Peng, X.M., Damu, G.L.V., and Zhou, C.H., Curr. Pharm. Des., 2013, vol. 19, p. 3884. https://doi.org/10.2174/1381612811319210013
Morita, H., Dota,T., and Kobayashi J., Bioorg. Med. Chem. Lett., 2004, vol. 14, p. 3665. https://doi.org/10.1016/j.bmcl.2004.05.015
Meggio, F., Pagano, M.A., Moro, S., Zagotto, G., Ruzzene, M., Sarno, S., Cozza, G., Bain, J., Elliott, M., Donella-Deana, A., Brunati, A.M., and Pinna, L.A., Biochemistry, 2004, vol. 43, p. 12931, https://doi.org/10.1021/bi048999g
Wu, X.-Q., Huang, C., Jia, Y.-M., Song, B.-A., Li, J., and Liu, X.-H., Eur. J. Med. Chem., 2014, vol. 74, p. 717. https://doi.org/10.1016/j.ejmech.2013.06.014
Chen, Y., Liu, H.R., Liu, H.S., Cheng, M., Xia, P., Qian, K., Wu, P.C., Lai, C.Y., Xia, Y., Yang, Z.Y., MorrisNatschke, S.L., and Lee, K.H., Eur. J. Med. Chem., 2012, vol. 49, p. 74. https://doi.org/10.1016/j.ejmech.2011.12.025
Chu, C.Y., Tsai, Y.Y., Wang, C.J., Lin, W.L., and Tseng, T.H., Eur. J. Pharmacol., 2001, vol. 416, p. 25. https://doi.org/10.1016/s0014-2999(01)00859-7
Ruiz, M.C., Chilpa, R.R., Estrada, E., Reyes, E.J., Fariña, G.G., and Fragoso, L.R.J., J. Pharm. Pharmacol., 2007, vol. 59, p. 719. https://doi.org/10.1211/jpp.59.5.0013
Bras, G.L., Radanyi, C., Peyrat, J.F., Brion, J.D., Alami, M., Marsaud, V., Stella, B., and Renoir, J.M., J. Med. Chem., 2007, vol. 50, p. 6189. https://doi.org/10.1021/jm0707774
Donnelly, A.C., Mays, J.R., Burlison, J.A., Nelson, J.T., Vielhauer, G., Holzbeierlein, J., and Blagg, B.S.J., J. Org. Chem., 2008, vol. 73, p. 8901. https://doi.org/10.1021/jo801312r
Dong, Y., Shi, Q., Liu, Y.-N., Wang, X., Bastow, K.F., and Lee, K.-H., J. Med. Chem., 2009, vol. 52, p. 3586. https://doi.org/10.1021/jm9001567
Wang, X., Nakagawa-Goto, K., Bastow, K.F., Don, M.J., Lin, Y.-L., Wu, T-S., and Lee, K.-H., J. Med. Chem., 2006, vol. 49, p. 5631. https://doi.org/10.1021/jm060184d
Stanway, S.J., Purohit, A., Woo, L.W., Sufi, S., Vigushin, D., Ward, R., Wilson, R.H., Stanczyk, F.Z., Dobbs, N., Kulinskaya, E., Elliott, M., Potter, B.V., Reed, M.J., and Coombes, R.C., Clin. Cancer Res., 2006, vol. 12, p. 1585. https://doi.org/10.1158/1078-0432.CCR-05-1996
Xu, X., Zhang, Y., Qu, D., Jiang, T., and Li, S., J. Exp. Clin. Cancer Res., 2011, vol. 30, p. 30. https://doi.org/10.1186/1756-9966-30-33
Zhang, W., Li, Z., Zhou, M., Wu, F., Hou, X., Luo, H., Liu, H., Han, X., Yan, G., Ding, Z., and Li, R., Bioorg. Med. Chem. Lett., 2014, vol. 24, p. 799. https://doi.org/10.1016/j.bmcl.2013.12.095
Ragavan, R.V., Vijayakumar, V., and Kumari, N.S., Pharmacol. Online, 2011, vol. 3, p. 1348
Tewari, A.K. and Mishra, A., Bioorg. Med. Chem., 2001, vol. 9, p. 715. https://doi.org/10.1016/s0968-0896(00)00285-6
El Shehry, M.F., Ewies, E.F. and Zayed, E.M., Russ. J. Gen. Chem., 2019, vol. 89, p. 492. https://doi.org/10.1134/S1070363219030216
Venkatesh, N., Sundergoud, S., Swamy, M.K., and Veerasomaiah, P., Russ. J. Org. Chem., 2020, vol. 56, p. 1635. https://doi.org/10.1134/S1070428020090213
Perez, A.G., Wester, R.T., Allen, M.C., Brown, J.A., Buchholz, A.R., Cook, E.R., Day, W.W., Hamanaka, E.S., Kennedy, S.P., Knight, D.R., Kowalczyk, P.J., Marala, R.B., Mularski, C.J., Novomisle, W.A., Ruggeri, R.B., Tracey, W.R., and Hill, R., Bioorg. Med. Chem. Lett., 2001, vol. 11, p. 803. https://doi.org/10.1016/S0960-894X(01)00059-2
Reddy, K.I., Aruna, C., Manisha, M., Srihari, K., Babu, K.S., Vijayakumar, V., Sarveswari, S., Priya, R., Amrita, A., and Siva, R., J. Photochem. Photobiol. B, 2017, vol. 168, p. 89. https://doi.org/10.1016/j.jphotobiol.2017.02.003
Camper, N.D., Gum, J.M., Lyles, D.E., Pennington, W.T., Beam, C.F., and Metz, C.R., J. S. C. Acad. Sci., 2017, vol. 15, p. 27.
Brighton, D., Johnston, S.R.D., and Wood, M., The Royal Marsden Hospital Handbook of Cancer Chemotherapy: A Guide for the Multidisciplinary Team, London: Elsevier Churchill Livingstone, 2005, p. 1–17
Reddy, T.S., Kulhari, V.H., Reddy, G., Bansal, V., Kamal, A., and Shukla, R., Eur. J. Med. Chem., 2015, vol. 101, p. 790. https://doi.org/10.1016/j.ejmech.2015.07.031
Swanepoel, B., Nitulescu, G.M., Olaru, O.T., Venables, L., and Venter, M.V.D., Int. J. Mol. Sci., 2019, vol. 20, p. 5559. https://doi.org/10.3390/ijms20225559
Schenone, S., Bruno, O., Ranise, A., Bondavalli, F., Brullo, C., Fossa, P., Mosti, L., Menozzi, G., Carraro, F., Naldini, A., Bernini, C., Manetti, F., and Botta, M., Bioorg. Med. Chem. Lett., 2004, vol. 14, p. 2511. https://doi.org/10.1016/j.bmcl.2004.03.013
Manetti, F., Brullo, C., Magnani, M., Mosci, F., Chelli, B., Crespan, E., Schenone, S., Naldini, A., Bruno, O., Trincavelli, M. L., Maga, G., Carraro, F., Martini, C., Bondavalli, F., and Botta, M., J. Med. Chem., 2008, vol. 51, p. 1252. https://doi.org/10.1021/jm701240c
Huang, S., Lin, R., Yu, Y., Lu, Y., Connolly, P.J., Chiu, G., Li, S., Emanuel, S.L., and Middleton, S.A., Bioorg. Med. Chem. Lett., 2007, vol. 17, p. 1243. https://doi.org/10.1016/j.bmcl.2006.12.031
Maciejewska, N., Olszewski, M., Jurasz, J., Serocki, M., Dzierzynska, M., Cekala, K., Wieczerzak, E., and Baginski, M., Scientific reports., 2022, vol. 12, p. 3703. https://doi.org/10.1038/s41598-022-07691-6
Farag, A.M., Mayhoub, A.S., Eldebss, T.M.A., Amr, A.-G.E., Ali, K.A.K., Abdel-Hafez, N.A., and Abdulla, M.M., Arch. Pharm. Chem. Life Sci.., 2010, vol. 343, p. 384. https://doi.org/10.1002/ardp.200900176
Husseiny, E.M., Bioorg. Chem., 2020, vol. 102, p. 104053. https://doi.org/10.1016/j.bioorg.2020.104053
Farag, A.M., Ali, K.A.K., El-Debss, T.M.A., Mayhoub, A.S., Amr, A.E., Abdel-Hafez, N.A., and Abdulla, M.M., Eur. J. Med. Chem., 2010, vol. 45, p. 5887. https://doi.org/10.1016/j.ejmech.2010.09.054
Shi, J.B., Tang, W.J., Bao, Qi.X., Li, J., Li, R., and Liu, X.H., Eur. J. Med. Chem., 2015, vol. 90, p. 889. https://doi.org/10.1016/j.ejmech.2014.12.013
Hassan, A.S., Moustafa, G.O., Awad, H.M., Nossier, E.S., and Mady, M.F., ACS Omega., 2021, vol. 6, p. 12361. https://doi.org/10.1021/acsomega.1c01604
Gupta, J.K., Sharma, P.K., Dudhe, R., Chaudhary, A., Singh, A., Verma, P.K., Mondal, S.C., Yadav, R.K., and Kashyap, S., Med. Chem. Res., 2012, vol. 21, p. 1625. https://doi.org/10.1007/s00044-011-9675-4
Ali, M.A., and Tarajder, M.T.H., J. Inorg. Nucl. Chem., 1977, vol. 39, p. 1785. https://doi.org/10.1016/0022-1902(77)80202-9
Manosroi, J., Dhumtanom, P., and Manosroi, A., Cancer Lett., 2006, vol. 235, p. 114. https://doi.org/10.1016/j.canlet.2005.04.021
Singh, V.K., Kadu, R., and Roy, H., Eur. J. Med. Chem., 2014, vol. 74, p. 552. https://doi.org/10.1016/j.ejmech.2013.12.035
Babić, S., Horvat, A.J., Pavlović, D.M., and Kaštelan-Macan, M., Trends Anal. Chem., 2007, vol. 26, p. 1043. https://doi.org/10.1016/j.trac.2007.09.004
Reddy, D.S., Kongot, M., Singh, V., Maurya, N., Patel, R., Singhal, N.K., Avecilla, F., and Kumar, A., Bioorg. Chem., 2019, vol. 92, p. 103212. https://doi.org/10.1016/j.bioorg.2019.103212
Manallack, D.T., Prankerd, R.J., Yuriev, E., Oprea, T.I., and Chalmers, D. K., Chem. Soc. Rev., 2013, vol. 42, p. 485. https://doi.org/10.1039/C2CS35348B
Fischer, H., Gottschlich, R., and Seelig, A., J. Membr. Biol., 1998, vol. 165, p. 201. https://doi.org/10.1007/s002329900434
Gasteiger, J., and Marsili, M., Tetrahedron,1980, vol. 36, p. 3219. https://doi.org/10.1016/0040-4020(80)80168-2
Tripos International Sybyl-X 2.0, Tripos International, St. Louis, MO, USA. 2012.
ACKNOWLEDGMENTS
The authors acknowledge the Department of Chemistry, Karnatak University’s Karnatak Science College, Dharwad and Seed money grant KUD for enabling research facilities. The authors would also like to acknowledge the SAIF and the Karnatak University Scientific Instrument Centre for their technical assistance for this investigation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Akki, M., Reddy, D.S., Katagi, K.S. et al. Coumarin-Pyrazole Linked Carbodithioates as Potential Anti-Сancer Agents: Design, Synthesis, Biological, and Molecular Docking Investigation. Russ J Gen Chem 92, 2092–2107 (2022). https://doi.org/10.1134/S1070363222100231
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222100231