Abstract
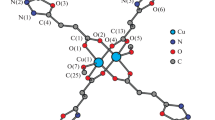
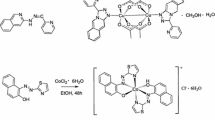
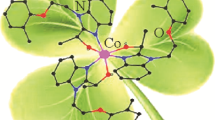
Azomethine HL was synthesized by the reaction of o-aminophenol with 2-acetylbenzimidazole and used to prepare the cobalt(III) complex [CoL2]2(ClO4)2⋅3H2O (I). The structure of complex I was established by NMR spectroscopy in solution and by single crystal X-ray diffraction (CIF file CCDC no. 2051279). The cobalt ion in the +3 oxidation state has an octahedral environment. The crystals are monoclinic, space group C2/c, a = 12.405(7), b = 13.946(11), c = 18.907(13) Å, α = 109.87(3)°, β = 94.534(12)°, γ = 104.564(19)°, V = 2928(3) Å3, ρ(calcd.) = 1.521 g/cm 3, Z = 2. The complex is diamagnetic. Study of the electrochemical behavior of I in acetonitrile showed that it can be reduced to neutral and radical anion forms.





Similar content being viewed by others
REFERENCES
Chattopadhyay, A., Meier, M., Ivaninskii, S., et al., Biochemistry, 2007, vol. 46, no. 28, p. 8315.
Montagner, C., de Souza, S.M., Groposo, C., et al., Z. Naturforsch., A: Phys. Sci., 2008, vol. 63, nos. 1–2, p. 21.
Hui, M.B.V., Lien, E.J., and Trousdale, M.D., Antivir. Res., 1994, vol. 24, no. 4, p. 261.
Krause, M., Rouleau, A., Stark, H., et al., J. Med. Chem., 1995, vol. 38, no. 20, p. 4070.
Lewinski, J., Zachara, J., Justyniak, I., and Dranka, M., Coord. Chem. Rev., 2005, vol. 249, nos. 11–12, p. 1185.
Kleij, A.W., Chem.-Eur. J., 2008, vol. 14, no. 34, p. 10520.
Garnovskii, A.D., Nivorozhkin, A.L., and Minkin, V.I., Coord. Chem. Rev., 1993, vol. 126, nos. 1–2, p. 1.
Garnovskii, A.D., Burlov, A.S., Vasil’chenko, I.S., et al., Russ. J. Coord. Chem., 2010, vol. 36, no. 2, p. 81. https://doi.org/10.1134/S1070328410020016
Harada, J., Uekusa, H., and Ohashi, Y., J. Am. Chem. Soc., 1999, vol. 121, no. 24, p. 5809.
Ohshima, A., Momotake, A., and Arai, T., J. Photochem. Photobiol., A, 2004, vol. 162, nos. 2–3, p. 473.
Uzhinov, B.M. and Khimich, M.N., Usp. Khim., 2011, vol. 80, no. 6, p. 580.
Minkin, V.I., Tsukanov, A.V., Dubonosov, A.D., and Bren, V.A., J. Mol. Struct., 2011, vol. 998, nos. 1–3, p. 179.
Tahlan, S., Ramasamy, K., Lim, S.M., et al., BMC Chem., 2019, vol. 13, no. 12, p. 1.
Tahlan, S., Ramasamy, K., Lim, S.M., et al., Chem. Sent. J., 2019, vol. 12, no. 139, p. 1.
Wang, X.J., Xi, M.Y., Fu, J.H., et al., Chin. Chem. Lett., 2012, vol. 23, no. 6, p. 707.
Desai, N.C., Shihory, N.R., Kotadiya, G.M., and Desai, P., Eur. J. Med. Chem., 2014, vol. 82, p. 480.
Camacho, J., Barazarte, A., Gamboa, N., et al., Bioorg. Med. Chem., 2011, vol. 19, no. 6, p. 2023.
Starcevic, K., Kralj, M., Ester, K., et al., Bioorg. Med. Chem., 2007, vol. 15, no. 13, p. 4419.
Monforte, A.M., Ferro, S., Luca, L.D., et al., Bioorg. Med. Chem., 2014, vol. 22, no. 4, p. 1459.
Ghosh, K., Tarafdar, D., Samadder, A., and Khuda-Bukhsh, A.R., RSC Adv., 2014, vol. 4, no. 102, p. 58530.
Lee, D.Y., Singh, N., Satyender, A., and Jang, D.O., Tetrahedron Lett., 2011, vol. 52, no. 51, p. 6919.
Tikhomirova, K.S., Tolpygin, I.E., Starikov, A.G., and Kaz’mina, M.A., Khim. Geterotsikl. Comp., 2017, vol. 53, no. 2, p. 179.
Kulhanek, J. and Bures, F., Beilstein J. Org. Chem., 2012, vol. 8, p. 25.
Zhang, L., Wu, J., Shi, L., et al., Tetrahedron Lett., 2011, vol. 52, no. 30, p. 3897.
Liu, J.-C., Chen, L.-W., Song, X.-Y., et al., Chin. J. Struct. Chem., 2011, vol. 30, no. 5, p. 764.
Chang, H.C., Son, B.C., Song, G.Y., et al., Macromol. Res., 2013, vol. 21, no. 1, p. 118.
Du, J., Li, L.-J., and Li, Y., Inorg. Chem. Commun., 2005, vol. 8, no. 3, p. 246.
Lahneche, Y.D., Boulebd, H., Benslimane, M., et al., J. Coord. Chem., 2019, vol. 72, no. 18, p. 3156.
Lukov, V.V., Kogan, V.A., Levchenkov, S.I., et al., Russ. J. Coord. Chem., 2015, vol. 41, no. 1, p. 1. https://doi.org/10.1134/S1070328415010054
Popov, L.D., Levchenkov, S.I., Shcherbakov, I.N., et al., Russ. J. Gen. Chem., 2010, vol. 80, no. 12, p. 2501.
Pavlov, A.A., Denisov, G.L., Kiskin, M.A., et al., Inorg. Chem., 2017, vol. 56, p. 14759.
Tupolova, Y.P., Shcherbakov, I.N., Popov, L.D., et al., Dalton Trans., 2019, vol. 48, no. 20, p. 6960.
Tupolova, Y.P., Shcherbakov, I.N., Korchagin, D.V., et al., J. Phys. Chem., vol. 124, no. 47, p. 25957.
Cheeseman, W.H., J. Chem. Soc., 1964, vol. 11, p. 4695.
SMART (control) and SAINT (integration) Software. Version 5.0, Madison: Bruker AXS Inc., 1997.
Krause, L., Herbst-Irmer, R., Sheldrick, G.M., and Stalke, D., J. Appl. Crystallogr., 2015, vol. 48, p. 3.
Sheldrick, G., Acta Crystallogr., Sect. A: Found. Crystallogr., 2007, vol. 64, p. 112.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Lever, A., Inorganic Electronic Spectroscopy, New York: Elesvier, 1984.
Casellato, U., Fregona, D., Sitran, S., et al., Inorg. Chim. Acta, 1984, vol. 95, no. 6, p. 309.
ACKNOWLEDGMENTS
X-ray diffraction analysis and magnetic measurements were performed using equipment of the Center for Collective Use of Physical Methods of Investigation, Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences, which is supported by the state assignment for the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences, in the field of fundamental research.
Funding
NMR studies were carried out using the equipment of the Center for Molecular Structure Studies, Nesmeyanov Institute of Organoelement Compounds, Russian Academy of Sciences, and was supported by the Russian Science Foundation (grant no. 20-73-00194).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Rights and permissions
About this article
Cite this article
Popov, L.D., Borodkin, S.A., Kiskin, M.A. et al. Synthesis and Crystal Structure of Cobalt(III) Chelate with Tridentate Azomethine Ligand Containing a Benzimidazole Moiety. Russ J Coord Chem 48, 9–15 (2022). https://doi.org/10.1134/S1070328421110038
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328421110038