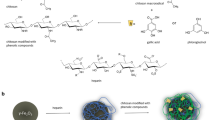
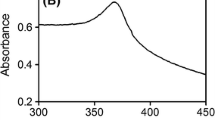
Abstract
The effect of nanodispersions with lipoic acid and its esters on the activity of lactate dehydrogenase in neutrophils and platelets has been studied. It has been shown that nanoparticles with lipoic acid and its esters decrease the activity of lactate dehydrogenase in neutrophils and platelets 1.5–2 times, which indicates that nanodispersions containing lipoic acid and its conjugates decrease the concentration of reactive oxygen species in activated neutrophils and platelets 2–3 times. Studies on the inhibition of free radical processes in neutrophils and platelets showed that the nanoforms with lipoic acid and its esters possess an antioxidant activity.





Similar content being viewed by others
REFERENCES
Kulesh, A.A., Demin, D.A., and Vinogradov, O.I., Cons. Med., 2021, vol. 23, pp. 792–799. https://doi.org/10.26442/20751753.2021.11.201153
Orellana-Urzúa, S., Rojas, I., Líbano, L., and Rodrigo, R., Curr. Pharm. Des., 2020, vol. 26, P. 4246–4260. https://doi.org/10.2174/1381612826666200708133912
Quinlan, C.L., Perevoshchikova, I.V., Hey-Mogensen, M., Orr, A.L., and Brand, M.D., Redox Biol., 2013, vol. 1, pp. 304–312. https://doi.org/10.1016/j.redox.2013.04.005
Maczurek, A., Hager, K., Kenklies, M., Sharman, M., Martins, R., Engel, J., Carlson, D.A., and Münch, G., Adv. Drug Deliv. Rev., 2008, vol. 60, pp. 1463–1470. https://doi.org/10.1016/j.addr.2008.04.015
Alban, F.T.E., Rowan, D.G., Golen, F., and Heger, M., The Liver. Oxidative Stress and Dietary Antioxidants, Patel, V.B., Rajendram, R., and Preedy, V.R., Eds., London: Academic, 2018, pp. 109–119. https://doi.org/10.1016/B978-0-12-803951-9.00010-0
Shay, K.P., Moreau, R.F., Smith, E.J., Smith, A.R., and Hagen, T.M., Biochim. Biophys. Acta, 2009, vol. 1790, pp. 1149–1160. https://doi.org/10.1016/j.bbagen.2009.07.026
Najafi, N., Mehri, S., Ghasemzadeh Rahbardar, M., and Hosseinzadeh, H., Phytother. Res., 2022, vol. 36, pp. 2300–2323. https://doi.org/10.1002/ptr.7406
Teoh, W.P., Resko, Z.J., Flury, S., and Alonzo, F., J. Bacteriol., 2019, vol. 201, pp. 1–14. https://doi.org/10.1128/JB.00446-19
Du, J., Li, X., Lin, C., and He, X., Inflammation, 2016, vol. 38, pp. 1458–1463. https://doi.org/10.1007/s10753-015-0120-6
Palaniappan, A.R. and Dai, A., Neurochem. Res., 2010, vol. 32, pp. 1552–1558. https://doi.org/10.1007/s11064-007-9355-4
Salehi, B., Berkay Yılmaz, Y., and Antika, G., Biomolecules, 2019, vol. 9, pp. 356–359. https://doi.org/10.3390/biom9080356
Brufani, M. and Figliola, R., Acta Biomed., 2014, vol. 85, pp. 108–115.
Gruzman, A., Hidmi, A., Katzhendler, J., Haj-Yehie, A., and Sasson, S., Bioorg. Med. Chem., 2004, vol. 12, pp. 1183–1190. https://doi.org/10.1016/j.bmc.2003.11.025
Xia, N., Liu, T., Wang, Q., Xia, Q., and Bian, X., J. Microencapsul., 2017, vol. 34, pp. 571–581. https://doi.org/10.1080/02652048.2017.1367852
Olie, R.H., van der Meijden, P.E.J., Spronk, H.M.H., and ten Cate, H., Handbook Exp. Pharmacol., 2022, vol. 270, pp. 103–130. https://doi.org/10.1007/164_2020_357
Suslina, Z.A., Erofeeva, A.V., Tanashyan, M.M., and Ionova, V.G., Neurolog. Psychiatry, 2011, vol. 19, pp. 562–573.
Lai, Y.S., Shih, C.Y., Huang, Y.F., and Chou, T.C., J. Agric. Food Chem., 2010, vol. 58, pp. 8596–8603. https://doi.org/10.1021/jf101518p
Shchelkonogov, V.A., Shipelova, A.V., Sinebryukhova, A.M., Shastina, N.S., Baranova, O.A., Chekanov, A.V., Solov’eva, E.Yu., and Fedin’ A.I., Biotechnology: Status and Development Prospects. Trudy Mezhdunarodnogo Kongressa (Proc. Int. Congress), Moscow, 2020, pp. 461–464. https://www.elibrary.ru/item.asp?selid=44375061&id=44374820
Shchelkonogov, V.A., Sorokoumova, G.M., Baranova, O.A., Chekanov, A.V., Klochkova, A.V., Kazarinov, K.D., Solov’eva, E.Yu., Fedin, A.I., and Shvets, V.I., Biomed. Khim., 2016, vol. 62, pp. 577–583. https://doi.org/10.18097/PBMC20166205577
Shchelkonogov, V.A., Inshakova, A.M., Shipelova, A.V., Baranova, O.A., Chekanov, A.V., Shastina, N.S., Solov’eva, E.Yu., and Fedin, A.I., Mendeleev Commun., 2021, vol. 31, pp. 507–508. https://doi.org/10.1016/j.mencom.2021.07.023
Farhana, A. and Lappin, S.L., Biochemistry, Lactate Dehydrogenase, StatPearls (Internet), 2022. https://www.ncbi.nlm.nih.gov/books/NBK557536
Meroni, S.B., Riera, M.F., Pellizzari, E.H., Schteingart, H.F., and Cigorraga, S.B., Int. J. Androl., 2003, vol. 26, pp. 310–317. https://doi.org/10.1046/j.1365-2605.2003.00432.x
Awasthi, D., Nagarkoti, S., Sadaf, S., Chandra, T., Kumar, S., and Dikshit, M., Biochim. Biophys. Acta Mol. Basis Dis., 2019, vol. 1865, pp. 165542. https://doi.org/10.1016/j.bbadis.2019.165542
Jones, W., Li, X., Qu, Z., Perriott, L., Whitesell, R.R., and May, J.M., Free Radical Biol. Med., 2002, vol. 33, pp. 83–93. https://doi.org/10.1016/s0891-5849(02)00862-6
Gomes, A., Fernandes, E., and Lima, J.C., J. Biochem. Biophys., 2005, vol. 65, pp. 48–80. Epub 2005 Nov 4. https://doi.org/10.1016/j.jbbm.2005.10.003
Miyake, M., Kakizawa, Y., Tobori, N., Kurioka, M., Tabuchi, N., Kon, R., Shimokawa, N., Tsujino, Y., and Takagi, M., Colloids Surf. B, 2018, vol. 169, pp. 444–452. https://doi.org/10.13140/RG.2.1.3892.5280
Mikheev, A.A., Shmendel’, E.V., Zhestovskaya, E.S., Nazarov, G.V., and Maslov, M.A., Tonkie Khim. Tekhnol., 2020, vol. 15, pp. 7–27. https://doi.org/10.32362/2410-6593-2020-15-1-7-27
Eqbal, A., Ansari, V. A., Hafeez, A., Ahsan, F., Imran, M., and Tanweer, S., Res. J. Pharm. Technol., 2021, vol. 14, pp. 2852–2858. https://doi.org/10.52711/0974-360X.2021.00502
Chekanov, A.V., Cand. Sci. (Biol.) Dissertation, Moscow, 2004.
ACKNOWLEDGMENTS
The authors express their gratitude to A.V. Romanenko for providing biomaterial for research, A.V. Symon for providing phosphatidylcholine Lipoid S-100, K.D. Kazarinov for providing arachidonic acid, as well as to the staff of the MIREA Center for Collective Use for providing equipment for research.
Funding
The work was carried out within the framework of state assignments for the RNRMU (no. 122051600109-5) and for the IRE RAS (no. 122041900109-3), and with financial support of the “Bazis” Foundation for the Development of Theoretical Physics and Mathematics (grant no. 22-1-1-28-1).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by VAS, AMI, ESD, AVS, AVC, and NSS. Conceptualization: AVC; Methodology: AMI; Writing—original draft preparation: VAS; Writing—review and editing: AIF; Supervision: EYS. The first draft of the manuscript was written by OAB and NSS, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The data that support the findings of this study are available from the corresponding author upon reasonable request. The use of biomaterial (venous blood samples) obtained from a group of apparently healthy individuals in this work and its subsequent use by A.V. Romanenko in the work on the thesis “Features of Vascular Inflammation and Hemostasis in the Acute Period of Ischemic and Hemorrhagic Strokes” were approved by the Committee for Biomedical Ethics of the RNRMU (partial record of the minutes of the RNIMU no. 204 of February 1, 2021). All volunteers gave voluntary consent to taking biomaterial. No conflict of interest was declared by the authors.
Additional information
Publisher's Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations: AA, arachidonic acid; ROS, reactive oxygen species; DHLA, dihydrolipoic acid; LDH, lactate dehydrogenase; LA, lipoic acid; LK2Hx, 1,6-di-O-lipoyloxyhexane; LA3Gro, 1,2,3-tri-O-lipoylglycerol; LADPAP, 2-O-lipoyloxy-1,3-(dipalmitoylamino)propane; PMNs, neutrophils; OG, oligoglycerol; PRP, platelet-rich plasma; LPO, lipid peroxidation; Pt, platelets; PBS, phosphate buffer solution (pH 7.4); PMA, phorbol-12-myristate-13-acetate; PC, phosphatidylcholine; DCFH-DA, 2,7-dichlorodihydrofluorescein diacetate; F-68, pluronic F-68.
Rights and permissions
About this article
Cite this article
Shchelkonogov, V.A., Inshakova, A.M., Darnotuk, E.S. et al. Effect of Nanodispersions with Lipoic Acid and Its Esters on the Functional Activity of Neutrophils and Platelets. Russ J Bioorg Chem 49, 1319–1327 (2023). https://doi.org/10.1134/S1068162023060122
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023060122