Abstract
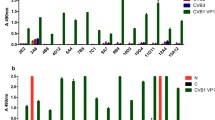
Hybridomas producing monoclonal antibodies (mABs) to the recombinant protein NS1 of the Zika virus synthesized in human embryonic kidney HEK293 (ZV293) cells have been obtained. The specificity of antibodies has been examined by the enzyme-linked immunosorbent assay (ELISA) using the similarly synthesized NS1 proteins of flaviviruses: the Dengue 1–4 serotype virus, Japanese encephalitis virus, yellow fever virus, tick-borne encephalitis virus, and the West Nile fever virus. Two types of hybridomas whose mABs interact with thermolabile (mABs 2F6 and 3F3) and thermostable epitopes (mABs 1B9, 6D7, and 6F2) of the NS1ZV protein have been selected. It has been shown that the 3F3 mAB, which is specific to the thermolabile determinant, can be used as both binding and detecting antibodies in the sandwich ELISA. Based on the 3F3 mAB, an experimental immunochromatographic test for the detection of the NS1ZV antigen has been developed. It has also been found that mABs 1B9, 6D7, and 6F2 against thermostable epitopes of the NS1 protein specifically recognize the NS1ZV protein after its heating for 15 min to 98°C.
Similar content being viewed by others
Abbreviations
- BSA:
-
bovine serum albumin
- ELISA:
-
enzymelinked immunosorbent assay
- IC test:
-
immunochromatographic test
- CG:
-
colloid gold
- mAB:
-
monoclonal antibody
- RT-PCR:
-
reverse transcription polymerase chain reaction
- PEG:
-
polyethylene glycol
- PBS–T:
-
0.1 M phosphate-buffered saline with 0.5% Twin-20
- HAT:
-
a mixture of hypoxanthine, aminopterin, and thymidine
- НТ:
-
a mixture of hypoxanthine and thymidine
- NR:
-
neutralization reaction: NS1ZV293, the recombinant protein NS1 of the Zika virus, synthesized in human embryonic kidney cell line HEK293
- NS1ZVE. сoli :
-
the recombinant protein NS1 of the Zika virus, synthesized in E. сoli
- ZVVero:
-
lysate of Vero cells infected with the Zika virus
References
Malone, R.W., Homan, J., Callahan, M.V., Glasspool-Malone, J., Damodaran, L., Schneider, A.D.B., Zimler, R., Talton, J., Cobb, R.R., Ruzic, I., Smith-Gagen, J., Janies, D., and Wilson, J., PLoS Negl. Trop. Dis., 2016, vol. 10, no. 3. e0004530. doi 10.1371/journal. pntd.0004530
World Health Organization. Zika Virus Disease, Interim case definition, World Health Organization, 2016. www.who.int/csr/disease/zika/case-definition/en/.
Bingham, A.M., Cone, M., Mock, V., Heberlein-Larson, L., Stanek, D., and Blackmore, C., MMWR Morb. Mortal Wkly Rep., 2016, vol. 65, no. 18, pp. 475–478.
Gourinat, A.C., O’Connor, O., Calvez, E., Goarant, C., and Dupont-Rouzeyrol, M., Emerg. Infect. Dis., 2015, vol. 21, no. 1, pp. 84–86.
Corbett, K.S., Katzelnick, L., Tissera, H., Amerasinghe, A., de Silva, A.D., and de Silva, A.M., J. Infect. Dis., 2015, vol. 211, no. 4, pp. 590–599.
Shan, C., Xie, X., Barrett, A.D.T., Garcia-Blanc, M.A., Tesh, R.B., Vasconcelos, P.F.D.C., Vasilakis, N., Weaver, S.C., and Shi, P.Y., ACS Infect. Dis., 2016, vol. 2, 2016, pp. 170–172.
Lanciotti, R.S., Kosoy, O.L., Laven, J.J., Velez, J.O., Lambert, A.J., Johnson, A.J., Stanfield, S.M., and Duffy, M.R., Emerg. Infect. Dis., 2008, vol. 14, no. 8, pp. 1232–1239.
Stettler, K., Beltramello, M., Espinosa, D.A., Graham, V., Cassotta, A., Bianchi, S., Vanzetta, F., Minola, A., Jaconi, S., Mele, F., Foglierini, M., Pedotti, M., Simonelli, L., Dowall, S., Atkinson, B., Percivalle, E., Simmons, C.P., Varani, L., Blum, J., Baldanti, F., Cameroni, E., Hewson, R., Harris, E., Lanzavecchia, A., Sallusto, F., and Corti, D., Science, 2016, vol. 353, pp. 823–826.
Calvet, G.A., Santos, F.B., and Sequeira, P.C., Curr. Opin. Infect. Dis., 2016, vol. 29, pp. 446–459.
Rocha, L.B., Alves, R.P.S., Caetano, B.A., Pereira, L.R., Mitsunari, T., Amorim, J.H., Polatto, J.M., Botosso, V.F., Gallina, N.M.F., Palacios, R., Precioso, A.R., Granato, C.F.H., Oliveira, D.B.L., Da Silveira, V.B., Luz, D., Ferreira, L.C.S., and Piazza, R.M.F., Antibodies, 2017, vol. 6, p. 14. doi 10.3390/antib6040014
Okabayashi, T., Sasaki, T., Masrinoul, P., Chantawat, N., Yoksan, S., Nitatpattana, N., Chusri, S., Vargas, R.E.M., Grandadam, M., Brey, P.T., Soegijanto, S., Mulyantno, K.C., Churrotin, S., Kotaki, T., Faye, O., Faye, O., Sow, A., Sall, A.A., Puiprom, O., Chaichana, P., Kurosu, T., Kato, S., Kosaka, M., Ramasoota, P., and Ikuta, K., J. Clin. Microbiol., 2015, vol. 53, no. 2, pp. 382–388.
Hang, V.T., Nguyet, N.M., Trung, D.T., Tricou, V., Yoksan, S., Dung, N.M., Van Ngoc, T., Hien, T.T., Farrar, J., Wills, B., and Simmons, C.P., PLoS Negl. Trop. Dis., 2009, vol. 3, no. 1, p. e360. doi 10.1371/journal.pntd.0000360
Blacksell, S.D., Jarman, R.G., Bailey, M.S., Tanganuchitcharnchai, A., Jenjaroen, K., Gibbons, R.V., Paris, D.H., Premaratna, R., de Silva, H.J., Lalloo, D.G., and Day, N.P., Clin. Vaccine Immunol., 2011, vol. 18, no. 12, pp. 2095–2101.
Huhtamo, E., Hasu, E., Uzcategui, N.Y., Erra, E., Nikkari, S., Kantele, A., Vapalahti, O., and Piiparinen, H., J. Clin. Virol., 2010, vol. 47, no. 1, pp. 49–53.
Nowinski, R.C., Lostrom, M.E., Tam, M.R., Stone, M.R., and Burnette, W.N., Virology, 1979, vol. 93, no. 1, pp. 111–126.
Laemmli, U.K., Nature, 1970, vol. 227, pp. 680–685.
Handly, D.A., Methods for synthesis of colloidal gold, in Colloidal Gold, Hayat, M.A., Ed., London: Academic Press, 1989, vol. 1, pp. 14–32.
Hermanson, G.T., Bioconjugate Techniques, London: Elsevier, 2008, 2nd ed.
Oliver, C., Methods Mol. Biol., 1994, vol. 34, pp. 321–328.
Fedyukina, G.N., Vetchinin, S.S., Baranova, E.V., Rudnitskii, S.Yu., Solov’ev, P.V., Kolosova, N.V., and Biketov, S.F., Biotekhnologiya, 2015, no. 1, pp. 85–92.
Blitvich, B.J., Scanlon, D., Shiell, B.J., Mackenzie, J.S., Pham, K., and Hall, R.A., J. Gen. Virol., 2001, vol. 82, no. 9, pp. 2251–2256.
Lisowska, E., Cell. Mol. Life Sci., 2002, vol. 59, no. 3, pp. 445–455.
Dowling, W., Thompson, E., Badger, C., Mellquist, J.L., Garrison, A.R., Smith, J.M., Paragas, J., Hogan, R.J., and Schmaljohn, C., J. Virol., 2007, vol. 81, no. 4, pp. 1821–1837.
Gavrilov, B.K., Rogers, K., Fernandez-Sainz, I.J., Holinka, L.G., Borca, M.V., and Risatti, G.R., Virology, 2011, vol. 420, no. 2, pp. 135–145.
Bu, G., Zhang, N., and Chen, F., Food Res. Int., 2015, vol. 76, no. 3, pp. 511–517.
Sun, X., Jayaraman, A., Maniprasad, P., Raman, R., Houser, K.V., Pappas, C., Zeng, H., Sasisekharan, R., Katz, J.M., and Tumpey, T.M., J. Virol., 2013, vol. 87, no. 15, pp. 8756–8766.
Song, H., Qi, J., Haywood, J., Shi, Y., and Gao, G.F., Nat. Struct. Mol. Biol., 2016, vol. 23, no. 5, pp. 456–458.
Haddow, A.D., Nasar, F., Guzman, H., Ponlawat, A., Jarman, R.G., Tesh, R.B., and Weaver, S.C., PLoS Negl.Trop. Dis., 2016, vol. 10, no. 10, p. e0005083. doi 10.1371/journal.pntd.0005083
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vetchinin, S.S., Shevyakov, A.G., Fedyukina, G.N. et al. Generation of Hybridomas Producing Monoclonal Antibodies to the NS1 Protein of the Zika Virus. Russ J Bioorg Chem 44, 745–754 (2018). https://doi.org/10.1134/S1068162019010187
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162019010187