Abstract
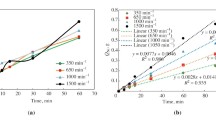
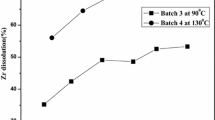
The dissolution of uranium alloys with Mo, Zr, Si, and other alloying elements in nitric acid has been studied. Data on the dissolution rate vs. the concentration of nitric acid and temperature are reported. The temperature coefficients of the reaction and the values of the effective activation energy of the interaction of alloys with an acid are calculated. For most of the samples, the obtained values are comparable with the dissolution rate of undoped uranium. In the case of introducing alloying elements Zr and Nb into metallic uranium, a decrease in the rate of the process occurred, and the rate of dissolution of uranium silicide was 5–10 times higher than that of undoped uranium. Upon dissolution, insoluble precipitates formed, their yield reached 8–10%. A relationship was established between the composition of inclusions present in the alloy and the composition of the precipitate after dissolution.




Similar content being viewed by others
REFERENCES
Sokurskii, Yu.N., Sterlin, Ya.M., and Fedorchenko, V.A., Uran i ego splavy (Uranium and Its Alloys), Moscow: Atomizdat, 1976.
Reshetnikov, F.G., Razrabotka tekhnologii polucheniya metallicheskogo urana i splavov na ego osnove, VNIINM – 50 let, Moscow, 1995.
Solonin, M.I., Vatulin, A.V., and Stetskii, Yu.A., Abstracts of Papers, XII ezhegodnoi konf. Yadernogo obshchestva Rossii “Issledovatel’skie reaktory: nauka i vysokie tekhnologii” 25–29 iyunya 2001 g. (12th Annual Conf. of Nuclear Society of Russia on Research Reactors: Science and High Technologies, June 25–29, 2001), 2001.
Zhirnov, A.D., Sirotkin, A.P., Bryunin, S.V., Pushkarev, S.V., and Runin, V.I., Atom. Energiya, 1973, vol. 34, no. 6, pp. 479–481.
Sirotkin, A.P., Atom. Tekhnika za Rubezhom, 1984, no. 3, pp. 3–13.
Kim, Sang-Ji, Kim, Young-Jin, Kim, Young-H, and Park, Chang-Kue, J. Nucl. Sci. Technol., 1999, vol. 36, no. 5, pp. 459–469.
Krasnorutskii, V.S. and Tatarinov, V.R., Vopr. Atom. Nauki i Tekhniki. Ser.: Fizika Radiatsionnykh Povrezhdenii i Radiatsionnoe Materialovedenie, 1999, nos. 1–2(73–74), pp. 87–94.
Vozvrashchenie reaktorov maloi moshchnosti (Return of Low Power Reactors), Atom. tekhnika za rubezhom. 2003, no. 3, pp. 25–28
Ferris, L.M., J. Chem. Eng. Data, 1961, vol. 6, pp. 600–603.
Meerson, G.A. and Mikhailova, V.G., Zn. Neorg. Khim., 1967, vol. 12, pp. 1615–1618.
Doucet, F.J., Goddard, D.T., Taylor, C.M., et al., Phys. Chem. Chem. Phys., 2002, vol. 4, pp. 3491–3499.
Akhmatov, A.A., Zil’berman, B.Ya., Fedorov, Yu.S., et al., Radiochemistry, 2003, vol. 45, no. 6, pp. 523–531. https://doi.org/10.1023/B:RACH.0000015756.24385.6d
Magnaldo, A., Masson, M., and Champion, R., Chem. Eng. Sci., 2007, vol. 62, pp. 766–774.
Maslennikov, A.G., Doctoral Sci. (Chem.) Dissertation, Moscow, 2008.
Khonina, I.V., Lumpov, A.A., Shadrin, A.Yu., Zil’berman, B.Ya., and Kravchenko, N.G., Radiochemistry, 2010, vol. 52, no. 2, pp. 151–154. https://doi.org/10.1134/S1066362210020104
Burakov B.E, Pokhitonov Yu.A, Ryazantsev, V.I., Savin, R.A., Saprykina,, V.F., and Rens, P.D., Radiochemistry, 2010, vol. 52, no. 4, pp. 403–407. https://doi.org/10.1134/S1066362210040144
Belash, N.N., Tatarinov, V.R., Ragulina, N.I., Semenov, N.A., and Danilova, O.V., Vopr. Atom. Nauki i Tekhniki. Ser.: Fizika Radiatsionnykh Povrezhdenii i Radiatsionnoe Materialovedenie, 2002, no. 3(81), pp. 88–93.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated from Radiokhimiya, No. 5, pp. 447–457, April, 2021 https://doi.org/10.31857/S003383112105004X
Supplementary information
Rights and permissions
About this article
Cite this article
Kolobov, E.A., Kirshin, M.Y. & Pokhitonov, Y.A. Dissolution of Uranium Alloys in Nitric Acid. Radiochemistry 63, 572–582 (2021). https://doi.org/10.1134/S1066362221050040
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362221050040