Abstract
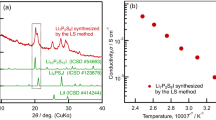
The compositions of nonstoichiometric phases Pb1 – xRxF2 + x (CaF2 type, R is a rare-earth element) have been optimized with respect to the ionic conductivity and thermal stability based on the temperature measurements of the electrical conductivity of single crystals and alloys, in dependence of their R3+ ionic radius and RF3 content, and on the analysis of the phase diagrams of the PbF2–RF3 systems. It is shown that Pb0.95Yb0.05F2.05 and Pb0.9Sc0.1F2.1 crystals have the highest conductivities among the Pb1 – xRxF2 + x phases (σ500 K = 9.3 × 10–3 and 2.0 × 10–2 S/cm and σ293 K = 1.4 × 10–6 and 1.5 × 10–4 S/cm, respectively). The conductivity σ293 K of the solid electrolyte Pb0.9Sc0.1F2.1 is sufficient for using it in room-temperature fluorine-ion sensors and current sources.



Similar content being viewed by others
REFERENCES
B. P. Sobolev and N. I. Sorokin, Crystallogr. Rep. 59 (6), 807 (2014).
N. I. Sorokin, A. M. Golubev, and B. P. Sobolev, Crystallogr. Rep. 59 (2), 248 (2014).
N. I. Sorokin and B. P. Sobolev, Crystallogr. Rep. 64 (5), 621 (2019).
A. K. Ivanov-Shits, N. I. Sorokin, P. P. Fedorov, and B. P. Sobolev, Solid State Ionics 31 (4), 253 (1989).
A. K. Ivanov-Shits, N. I. Sorokin, P. P. Fedorov, and B. P. Sobolev, Solid State Ionics 31 (4), 269 (1989).
A. K. Ivanov-Shits, N. I. Sorokin, P. P. Fedorov, and B. P. Sobolev, Solid State Ionics 37 (1–2), 125 (1990).
N. I. Sorokin, Elektrokhimiya 42 (7), 828 (2006).
B. P. Sobolev, N. I. Sorokin, and N. B. Bolotina, Photonic and Electronic Properties of Fluoride Materials, Vol. 1: Int. Collection “Progress in Fluorine Science,” Ed. by A. Tressaud and K. Poeppelmeier (Elsevier, Amsterdam, 2016), p. 465.
N. I. Sorokin, P. P. Fedorov, A. K. Ivanov-Shits, and B. P. Sobolev, Fiz. Tverd. Tela 30 (5), 1537 (1988).
N. I. Sorokin, E. A. Sul’yanova, I. I. Buchinskaya, and B. P. Sobolev, Crystallogr. Rep. 50 (4), 695 (2005).
N. I. Sorokin, B. P. Sobolev, and M. Breiter, Phys. Solid State 44 (8), 1579 (2002).
J. W. Fergus, Sens. Actuators B 42, 119 (1997).
N. I. Sorokin and B. P. Sobolev, Crystallogr. Rep. 52 (5), 842 (2007).
A. V. Chadwick, Solid State Ionics 8, 209 (1983).
N. I. Sorokin, Russ. J. Phys. Chem. 76 (3), 319 (2002).
B. P. Sobolev, The Rare Earth Trifluorides, Part 1: The High Temperature Chemistry of the Rare Earth Trifluorides (Institute of Crystallography, Moscow, 2000; Institut d’Estudis Catalans, Barcelona, 2000).
I. I. Buchinskaya and P. P. Fedorov, Usp. Khim. 73 (4), 404 (2004).
C. C. Liang and A. V. Joshi, J. Electrochem. Soc. 122, 466 (1975).
I. V. Murin, Izv. SO Akad. Nauk SSSR, Ser. Khim. Nauk, No. 2, Iss. 1, 53 (1984).
J. M. Reau, P. P. Fedorov, L. Rabardel, et al., Mater. Res. Bull. 18, 1235 (1983).
A. Rhandour, J. M. Reau, S. F. Matar, and P. Hagenmuller, J. Phys. Chem. Solids 47 (6), 587 (1986).
J. ten Eicken, W. Gunsser, S. V. Chernov, et al., Solid State Ionics 53–56, 843 (1992).
S. J. Patwe, P. Balaya, P. S. Goyal, and A. K. Tyagi, Mater. Res. Bull. 36, 1743 (2001).
N. I. Sorokin and B. P. Sobolev, Crystallogr. Rep. 61 (3), 469 (2016).
D. S. Stockbarger, J. Opt. Soc. Am. 39 (9), 31 (1949).
V. A. Arkhangel’skaya, V. N. Baklanova, I. A. Ivanova, et al., Trudy GOI (Gos. Opt. Inst.) 54 (188), 129 (1983).
Y. Ito and K. Koto, Solid State Ionics 18–19, 1202 (1986).
B. P. Sobolev, The Rare Earth Trifluorides, Part 2: Introduction to Materials Science of Multicomponent Metal Fluoride Crystals (Institute of Crystallography, Moscow, 2001; Institut d’Estudis Catalans, Barcelona, 2001).
N. I. Sorokin, P. P. Fedorov, and B. P. Sobolev, Inorg. Mater. 33 (1), 1 (1997).
N. I. Sorokin, G. A. Shchavlinskaya, I. I. Buchinskaya, and B. P. Sobolev, Elektrokhimiya 34 (9), 1031 (1998).
C. E. Derrington, A. Navrotsky, and M. O’Keeffe, Solid State Commun. 18, 47 (1976).
J. P. Goff, W. Hayes, S. Hull, and M. T. Hutchings, J. Phys.: Condens. Matter 3, 3677 (1991).
I. Kosacki, A. P. Litvinchuk, J. J. Tarasov, and M. Ya. Valakh, J. Phys.: Condens. Matter 1, 929 (1989).
L. M. Volodkovich, G. S. Petrov, R. A. Vecher, and A. A. Vecher, Termochim. Acta 88, 497 (1985).
H. W. den Hartog and. J. van der Veen, Phys. Rev. B 37 (4), 1807 (1988).
P. P. Fedorov, I. P. Zibrov, B. P. Sobolev, and I. V. Shishkin, Zh. Neorg. Khim. 32 (7), 1794 (1987).
P. P. Fedorov, I. P. Zibrov, E. V. Tarasova, et al., Zh. Neorg. Khim. 33 (12), 3222 (1988).
P. P. Fedorov, V. Trnovcova, V. A. Meleshina, et al., Neorg. Mater. 30 (3), 406 (1994).
W. Hume-Rothery and G. V. Raynor, The Structure of Metals and Alloys (The Institute of Metals, London, 1956).
J. ten Eicken, W. Gunsser, M. Karus, et al., Solid State Ionics 72, 7 (1994).
R. W. Bonne and J. Schoonman, J. Electrochem. Soc. 124, 28 (1977).
I. V. Murin, A. V. Glumov, and O. V. Glumov, Elektrokhimiya 15 (8), 1119 (1979).
W. Bollmann and R. Reimann, Phys. Status Solidi A 16, 187 (1973).
J. Schoonman and H. W. den Hartog, Solid State Ionics 7, 9 (1982).
D. R. Figueroa, A. V. Chadwick, and J. H. Strange, J. Phys. C 11, 55 (1978).
A. N. Aleinikov, N. N. Aleinikov, and N. N. Vershinin, Proc. IX All-Union Symp. on Chemistry of Inorganic Fluorides, Cherepovets, July 3–6,1990, p. 31.
J. H. Kennedy and J. C. Hunter, J. Electrochem. Soc. 123 (1), 10 (1976).
P. Hagenmuller, J. M. Reau, C. Lucat, et al., Solid State Ionics 3–4, 341 (1981).
I. Kosacki, Appl. Phys. A 49, 413 (1989).
R. N. Zakirov and A. S. Marinin, Proc. IX All-Union Symp. on Chemistry of Inorganic Fluorides, Cherepovets, July 3–6,1990, p. 136.
A. A. Potanin, Zh. Vseross. Khim. O-va im. D. I. Mendeleeva. 45 (5–6), 58 (2001).
Funding
This study was supported by the Ministry of Science and Higher Education of the Russian Federation within the State assignment for the Federal Scientific Research Centre “Crystallography and Photonics” of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by Yu. Sin’kov
Rights and permissions
About this article
Cite this article
Sorokin, N.I., Sobolev, B.P. Optimization of the Compositions of Solid Electrolytes Pb1 – xRxF2 + x with Fluorite-Type Structure in Conductivity and Thermal Stability. Crystallogr. Rep. 65, 98–105 (2020). https://doi.org/10.1134/S1063774520010241
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774520010241