Abstract
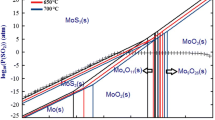
The results of the thermodynamic modeling are presented for oxidizing roasting of refractory molybdenite (MoS2) with magnesite (MgCO3). The phase and chemical compositions of the MoS2–MgCO3–O2 system and its changes are determined depending on the temperature and quantity of the additive agent. The modeling reveals feasibility of thermochemical decomposition of molybdenite with the formation of waterand soda-soluble compounds MgMoO4 and MgSO4, which is an evidence of efficiency of magnesium carbonate in the capacity of an additive agent. The optimal conditions of the thermochemical decomposition of molybdenite determined theoretically from thermodynamic calculations are proved experimentally.
Similar content being viewed by others
References
Zelikman, A.N. and Meerson, G.A., Metallurgiya redkikh metallov (Rare Metal Metallurgy), Moscow: Metallurgiya, 1973.
Liu W., Xu H., Yang X., and Shi X., Extraction of Molybdenum from Low-Grade Ni-Mo Ore in Sodium Hypochlorite Solution under Mechanical Activation, Minerals Engineering, 2011, vol. 24, no. 14, pp. 1580–1585.
Amer, A.M., Hydrometallurgical Recovery of Molybdenum from Egyptian Qattar Molybdenite Concentrate, Physicochem. Probl. Min. Proc., 2011, vol. 47, no. 14, pp. 105–112.
Aleksandrov, P.V., Medvedev, A.S., Milovanov, M.F., Imideev, V.A., Kotova, S.A., and Moskovskikh, D.O., Molybdenum Recovery from Molybdenite Concentrates by Low-Temperature Roasting with Sodium Chloride, Int. J. Min. Proc., 2017, vol. 161, pp. 13–20.
Vatolin, N.A., Khalezov, B.D., Kharin, E.I., and Zelenin, E.A., Review of Ways to Process Molybdenite Concentrates and Search for an Ecologically Friendly Process, GIAB, 2011, no. 12, pp. 170–175.
Khalezov, B.D., Kharin, E.I., Vatolin, N.A., and Zelenin, R.A. RF Patent no. 2536615, Byull Izobret., 2014, no. 36, p. 7.
Saily, A., Khurana, U., Yadav, S.K., and Tandon S.N. Thiophosphinic Acids as Selective Extractants for Molybdenum Recovery from a Low Grade Ore and Spent Catalysts, Hydrometallurgy, 1996, vol. 41, pp. 99–105.
Olson, G.J. and Clark, T.R., Bioleaching of Molybdenite, Hydrometallurgy, 2008, vol. 93, pp. 10–15.
Abdollahi, H., Noaparast, M., Shafaei, S.Z., Manafi, Z., Munoz, J.A., and Tuovinen, O.H., Silver-Catalyzed Bioleaching of Copper, Molybdenum and Rhenium from a Chalcopyrite–Molybdenite Concentrate, Int. Biodeterioration & Biodegradation, 2015, vol. 104, no. 1–2, pp. 194–200.
Lasheen, T.A., El-Ahmady, M.E., Hassib, H.B., and Helal, A.S., Molybdenum Metallurgy Review: Hydrometallurgical Routes to Recovery of Molybdenum from Ores and Mineral Raw Materials, Min. Proc. Extr. Metall. Rev., 2015, vol. 36, no. 3, pp. 145–173.
Vladimirov, L.P., Termodinamicheskie raschety ravnovesiya metallurgicheskikh reaktsii (Thermodynamic Calculations of Metallurgical Reaction Equilibrium), Moscow: Metallurgiya, 1970.
Belov, E.G. and Trusov, B.G., Termodinamicheskoe modelirovanie khimicheski reagiruyushchikh system (Thermodynamic Modeling of Chemically Reacting Systems), Moscow: MGTU, 2013.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D.P. Khomoksonova, E.S. Kashkak, I.G. Antropova, 2018, published in Fiziko-Tekhnicheskie Problemy Razrabotki Poleznykh Iskopaemykh, 2018, No. 2, pp. 130–135.
Rights and permissions
About this article
Cite this article
Khomoksonova, D.P., Kashkak, E.S. & Antropova, I.G. Improvement of Oxidizing Roasting of Molybdenite Concentrate by Addition of Magnesite. J Min Sci 54, 300–305 (2018). https://doi.org/10.1134/S1062739118023665
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062739118023665