Abstract
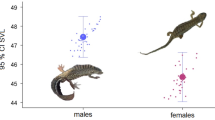
Males and females of the black forest–steppe adder (Pelias berus nikolskii) inhabiting Voronezh oblast, apart from differences in tail length, show external similarities. Individuals have a black body; females may have red–brown spots on their heads. We studied the differences in the ontogeny of 118 males and females caught between 2008 and 2017 in seven locations in Voronezh oblast. Seven traditional body measurements and one calculated trait of the overall head size were used for the analysis. Ontogenetic allometry was examined in both sexes. Allometric trajectories were constructed using reduced major axis (RMA, model II) regression. Bootstrap the residuals were used to test for the significance of the angles between the trajectories for the different sexes. A comparison of snake sizes at birth was conducted using a permutation test. To calculate both the sexual dimorphism index (SDI) and the allometry line of sexual size dimorphism (SSD), the residues obtained from the calculations of ontogenetic allometry trajectories were used, to which the predicted measurement values were added. The head size in relation to the body length was found to decrease with age. At birth, females were larger than males in the following traits: relative width of the head at eye level, relative length of the pileus, relative muzzle length, and overall head size. In the adult state, higher values of these traits were found in males. Males have longer tails. Differences in tail length appear at birth and persist throughout growth. Sex differences in growth changes in the traits studied, with the exception of the differences in head length, greatest width of the head, and tail length, are non-allometric in nature and, in addition to prenatal divergence, are caused by a greater growth rate in males. The growth of the tail in both sexes appears to be isometric. Rensch’s rule predicts that SSD will be positively correlated with size in taxa in which males are the larger sex, but negatively correlated with size in taxa in which the larger sex is the female. A study of SSD allometry showed that only width of the head at eye level, length of the pileus, muzzle length, and overall head size are consistent with this rule. It is therefore assumed that the larger head in males could have been formed under the action of sexual selection. This may be due to the need for them to participate in combat for access to females during the mating season.








Similar content being viewed by others
Notes
Ontogenesis of an individual includes two main processes: growth and development. Growth characterizes quantitative changes in individual development, and development characterizes qualitative changes.
Chronological age is the length of time an organism has elapsed since birth.
Biological age is the degree of morphological and physiological development of the organism.
REFERENCES
Abouheif, E. and Fairbairn, D.J., A comparative analysis of allometry for sexual size dimorphism: assessing Rensch’s rule, Am. Nat., 1997, vol. 149, no. 3, pp. 540–562. https://doi.org/10.1086/286004
Alberch, P., Gould, S.J., Oster, G.F., and Wake, D.B., Size and shape in ontogeny and phylogeny, Paleobiology, 1979, vol. 5, no. 3, pp. 296–317. https://doi.org/10.1017/S0094837300006588
Blanckenhorn, W.U., Dixon, A.F.G., Fairbairn, D.J., Foellmer, M.W., Gibert, P., Van Der Linde, K., Meier, R., Nylin, S., Pitnick, S., Schoff, C., Signorelli, M., Teder, T., and Wiklund, C., Proximate causes of Rensch’s rule: does sexual size dimorphism in arthropods result from sex differences in development time?, Am. Nat., 2007, vol. 169, no. 2, pp. 245–257. https://doi.org/10.1086/510597
Ceballos, C.P., Adams, D.C., Iverson, J.B., and Valenzuela, N., Phylogenetic patterns of sexual size dimorphism in turtles and their implications for Rensch’s rule, Evol. Biol., 2013, vol. 40, no. 2, pp. 194–208. https://doi.org/10.1007/s11692-012-9199-y
Clutton-Brock, T.H., Size, sexual dimorphism, and polygyny in primates, in Size and Scaling in Primate Biology, MA, Boston: Springer, 1985, pp. 51–60. https://doi.org/10.1007/978-1-4899-3647-9_4
Clutton-Brock, T.H., Harvey, P.H., and Rudder, B., Sexual dimorphism, socionomic sex ratio and body weight in primates, Nature, 1977, vol. 269, no. 5631, pp. 797–800. https://doi.org/10.1038/269797a0
Davison, A. and Hinkley, D., Bootstrap Methods and Their Application (Cambridge Series in Statistical and Probabilistic Mathematics), Cambridge: Cambridge University Press, 1997.
Dunaev, E.A. and Orlova, V.F., Zemnovodnye i presmykayushchiesya Rossii. Atlas-opredelitel’ (Amphibians and Reptiles of Russia: Atlas–Identification Guide), Moscow: Fiton+, 2012.
Fairbairn, D.J., Allometry for sexual size dimorphism: pattern and process in the coevolution of body size in males and females, Annu. Rev. Ecol. Syst., 1997, vol. 28, no. 1, pp. 659–687. https://doi.org/10.1146/annurev.ecolsys.28.1.659
Fairbairn, D.J. and Preziosi, R.F., Sexual selection and the evolution of allometry for sexual size dimorphism in the water strider, Aquarius remigis, Am. Nat., 1994, vol. 144, no. 1, pp. 101–118. https://doi.org/10.1086/285663
Fuiman, L.A., Growth gradients in fish larvae, J. Fish Biol., 1983, vol. 23, no. 1, pp. 117–123. https://doi.org/10.1111/j.1095-8649.1983.tb02886.x
Geodakyan, V.A., On the structure of evolving systems, in Problemy kibernetiki (Problems of Cybernetics), Moscow: Nauka, 1972, no. 25, pp. 81–91.
Geodakyan, V.A., The role of sexes in the transmission and transformation of genetic information, Probl. Peredachi Inf., 1965, vol. 1, no. 1, pp. 105–112.
Gregory, P., Reptiles sexual dimorphism and allometric size variation in a population of grass snakes (Natrix natrix) in Southern England, J. Herpetol., 2004, vol. 38, no. 2, pp. 231–240. https://doi.org/10.1670/122-03A
Huxley, J.S., Problems of Relative Growth, New York: The Dial Press, MacVeagh, 1932.
King, R.B., Sexual dimorphism in snake tail length: sexual selection, natural selection, or morphological constraint?, Biol. J. Linn. Soc., 1989, vol. 38, no. 2, pp. 133–154. https://doi.org/10.1111/j.1095-8312.1989.tb01570.x
King, R.B., Bittner, T.D., Queral-Regil, A., and Cline, J.H., Sexual dimorphism in neonate and adult snakes, J. Zool., 1999, vol. 247, no. 1, pp. 19–28. https://doi.org/10.1111/j.1469-7998.1999.tb00189.x
LaBarbera, M., Analyzing body size as a factor in ecology and evolution, Annu. Rev. Ecol. Syst., 1989, vol. 20, no. 1, pp. 97–117. https://doi.org/10.1146/annurev.es.20.110189.000525
Lawing, A.M., Head, J.J., and Polly, P.D., The ecology of morphology: the ecometrics of locomotion and macroenvironment in North American snakes, in Paleontology in Ecology and Conservation, Berlin: Springer, 2012, pp. 117–146. https://doi.org/10.1007/978-3-642-25038-5_7
Leutenegger, W., Scaling of sexual dimorphism in body size and breeding system in primates, Nature, 1978, vol. 272, no. 5654, pp. 610–611. https://doi.org/10.1038/272610a0
Liao, W.B. and Chen, W., Inverse Rensch’s rule in a frog with female-biased sexual size dimorphism, Naturwissenschaften, 2012, vol. 99, no. 5, pp. 427–431. https://doi.org/10.1007%2Fs00114-012-0913-5
Lovich, J.E. and Gibbons, J.W., A review of techniques for quantifying sexual size dimorphism, Growth Dev. Aging, 1992, vol. 56, no. 4, pp. 269–269.
Lucchini, N., Kaliontzopoulou, A., Val, G.A., and Martínez-Freiría, F., Sources of intraspecific morphological variation in Vipera seoanei: allometry, sex, and colour phenotype, Amphibia–Reptilia, 2020, no. 1, pp. 1–16. https://doi.org/10.1163/15685381-bja10024
McArdle, B.H., The structural relationship: regression in biology, Can. J. Zool., 1988, vol. 66, no. 11, pp. 2329–2339. https://doi.org/10.1139/z88-348
Mosimann, J.E., Size allometry: size and shape variables with characterizations of the lognormal and generalized gamma distributions, J. Am. Stat. Assoc., 1970, vol. 65, no. 330, pp. 930–945. https://doi.org/10.1080/01621459.1970.10481136
Rensch, B., Die Abhängigkeit der relativen Sexualdifferenz von der Körpergrösse, Bonner Zool. Beitr., 1950, no. 1, pp. 58–69.
Ricker, W.E., Computation and uses of central trend lines, Can. J. Zool., 1984, vol. 62, no. 10, pp. 1897–1905. https://doi.org/10.1139/z84-279
Shine, R., Olsson, M.M., Moore, I.T., LeMaster, M.P., and Mason, R.T., Why do male snakes have longer tails than females?, Proc. R. Soc. London, Ser. B, 1999, vol. 266, no. 1434, pp. 2147–2151. https://doi.org/10.1098/rspb.1999.0901
Shitikov, V.K. and Rozenberg, G.S., Randomizatsiya i butstrep: statisticheskii analiz v biologii i ekologii s ispol’zovaniem R (Randomization and Bootstrap: Statistical Analysis in Biology and Ecology Using R), Tolyatti: Kassandra, 2013.
Silva, F.M., Oliveira, L.S., Souza Nascimento, L.R., Machado, F.A., and Costa Prudente, A.L., Sexual dimorphism and ontogenetic changes of Amazonian pit vipers (Bothrops atrox), Zool. Anz., 2017, no. 271, pp. 15–24. https://doi.org/10.1016/j.jcz.2017.11.001
Smith, J.M., Parental investment: a prospective analysis, Anim. Behav., 1977, vol. 25, no. 1, pp. 1–9. https://doi.org/10.1016/0003-3472(77)90062-8
Sokal, R.R. and Rohlf, F.J., Biometry, in The Principles and Practice of Statistics in Biological Research, New York: W.H. Freeman and Co., 1995.
Storer, R.W., Sexual dimorphism and food habits in three north American accipiters, Auk, 1966, vol. 83, no. 3, pp. 423–436. https://doi.org/10.2307/4083053
Strauss, R.E., On allometry and relative growth in evolutionary studies, Syst. Zool., 1987, vol. 36, no. 1, pp. 72–75.https://doi.org/10.2307/2413309
Székely, T., Freckleton, R.P., and Reynolds, J.D., Sexual selection explains Rensch’s rule of size dimorphism in shorebirds, Proc. Natl. Acad. Sci. U. S. A., 2004, vol. 101, no. 33, pp. 12224–12227. https://doi.org/10.1073/pnas.0404503101
Tamagnini, D., Stephenson, J., Brown, R.P., and Meloro, C., Geometric morphometric analyses of sexual dimorphism and allometry in two sympatric snakes: Natrix helvetica (Natricidae) and Vipera berus (Viperidae), Zoology, 2018, no. 129, pp. 25–34. https://doi.org/10.1016/j.zool.2018.05.008
Tomović, L., Radojicić, J., Dzukić, G., and Kalezić, M.L., Sexual dimorphism of the sand viper (Vipera ammodytes L.) from the central part of Balkan Peninsula, Russ. J. Herpetol., 2002, vol. 9, no. 1, pp. 69–76. https://doi.org/10.30906/1026-2296-2002-9-1-69-76
Ushakov, M.V. and Zinenko, A.I., Subspecies of the common viper (Serpentes: Viperidae) from the Voronezh and Lipetsk regions, Vestn. Tambov. Univ., Ser. Estestv. Tekh. Nauki, Tambov, 2013, vol. 18, no. 6, pp. 3093–3097.
Ushakov, M.V., Klimov, A.S., and Tkachenko, A.V., To study the distribution of the eastern steppe viper, Vipera renardi (Christoph, 1861), in the Voronezh region, in Aktual’nye problemy gerpetologii i toksinologii. Sbornik nauchnykh trudov (Actual Problems of Herpetology and Toxinology. Collection of Scientific Papers), Tolyatti, 2006, no. 9, pp. 172–175.
Webb, T.J. and Freckleton, R.P., Only half right: species with female-biased sexual size dimorphism consistently break Rensch’s rule, PLoS One, 2007, vol. 2, no. 9, p. e897. https://doi.org/10.1371/journal.pone.0000897
Webster, M.S., Sexual dimorphism, mating system and body size in new world blackbirds (Icterinae), Evolution, 1992, vol. 46, no. 6, pp. 1621–1641. https://doi.org/10.1111/j.1558-5646.1992.tb01158.x
Zelditch, M.L., Swiderski, D.L., and Sheets, H.D., Geometric Morphometrics for Biologists: A Primer, Academic Press, 2012. https://doi.org/10.1016/C2010-0-66209-2
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest. The authors declare that they have no conflicts of interest.
Statement on the welfare of animals. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Ushakov, M.V., Frolova, E.N. Sexual Dimorphism in the Ontogeny of the Black Forest–Steppe Adder (Pelias berus nikolskii (Vedmederja, Grubant et Rudaeva 1986), Serpentes, Viperidae) from Voronezh Oblast, Based on Traditional Measurements. Biol Bull Russ Acad Sci 50, 1681–1694 (2023). https://doi.org/10.1134/S1062359023070300
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359023070300