Abstract
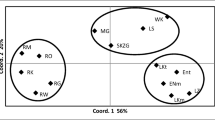
The variability of four microsatellite loci of chloroplast DNA was studied in 38 populations of Pinus sylvestris in the European part of Russia, in Siberia, Kazakhstan, Transbaikalia and Mongolia. High variability was observed in all regions. In total, 152 haplotypes were identified. The level of population differentiation R ST was 2.1%. The differentiation of three geographical groups of populations (European, Siberian-Kazakhstan and Transbaikalian-Mongolian) was insignificant (R CT = 0.004). At the same time, some rare haplotypes were found to be specific for a certain geographical region. Distribution of the rare haplotypes, which differentiated European populations from Asian populations and Mongolian and Transbaikalian populations from the Siberian, showed the independence of the history of these regions. This corresponds more to the hypothesis that the modern area of Pinus sylvestris originated via settlement from many origins than to the hypothesized single center of the post-glacial recolonization. The distribution of the pairwise differences between the individual specimens corresponded to the model of sudden population growth. The assessments of the age of this event for Pinus sylvestris (4.5–4.7 million years), which were obtained on the basis of this model, significantly exceeded the age of the Quaternary. Therefore, the revealed population growth is hardly due to the changes in flora related with the glaciation, but rather mirrors the moment of the species formation.
Similar content being viewed by others
References
Hewitt, G., The genetic legacy of the Quaternary ice ages, Nature, 2000, vol. 405, pp. 907–913.
Petit, R.J., Aguinagalde, I., de Beaulieu, J.L., et al., Glacial refugia: hotspot but not melting pots of genetic diversity, Science, 2003, vol. 300, pp. 1563–1565.
Lascoux, M., Palme, A.E., Cheddadi, R., and Latta, R.G., Impact of ice ages on the genetic structure of trees and shrubs, Philos. Trans. R. Soc., B, 2004, vol. 359, pp. 197–207.
Semerikov, V.L., Iroshnikov, A.I., and Lasko, M., Mitochondrial DNA variation pattern and postglacial history of the Siberian larch (Larix sibirica Ledeb.), Russ. J. Ecol., 2007, vol. 38, no. 3, pp. 147–154.
Semerikova, S.A. and Semerikov, V.L., Genetic variation and population differentiation in Siberian fir Abies sibirica Ledeb. inferred from allozyme markers, Russ. J. Genet., 2006, vol. 42, no. 6, pp. 636–644.
Semerikova, S.A. and Semerikov, V.L., The diversity of chloroplast microsatellite loci in Siberian fir (Abies sibirica Ledeb.) and two Far East fir species A. nephrolepis (Trautv.) Maxim. and A. sachalinensis Fr. Schmidt, Russ. J. Genet., 2007, vol. 43, no. 12, pp. 1373–1381.
Krutovskii, K. and Bergmann, F., Introgressive hybridization and phylogenetic relationships between Norway, Picea abies (L.) Karst., and Siberian, P. obovata Ledeb., spruce species studied by isozyme loci, Heredity, 1995, vol. 74, pp. 464–480.
Sannikov, S.N., Sannikova, N.S., and Petrova, I.V., Ocherk po teorii lesnoi populyatsionnoi biologii (Essay on the Theory of Forest Population Biology), Yekaterinburg: Ural. Otd. Ross. Akad. Nauk, 2012.
Neishtadt, M.I., Istoriya lesov i paleogeografiya SSSR v golotsene (History of forests and paleogeography of the Soviet Union in the Holocene), Moscow: Akad. Nauk, 1957.
Kremenetskii, K.V., Holocene history of distribution areas of principal conifer species in Siberia, in Korotkoperiodnye i rezkie landshaftno-klimaticheskie izmeneniya za poslednie 15000 let (Short-Term Periodic and Harsh Landscape and Climatic Changes over the Past 15000 Years), Moscow: Inst. Geografii Ross. Akad. Nauk, 1994, pp. 160–210.
Binney, A.H., Willis, K.J., Edwards, M.E., et al., The distribution of late-Quaternary woody taxa in northern Eurasia: evidence from a new macrofossil database, Quat. Sci. Rev., 2009, vol. 28, pp. 2445–2464.
Neale, D.B. and Sederoff, R.R., Paternal inheritance of chloroplast DNA and maternal inheritance of mitochondrial DNA in loblolly pine, Theor. Appl. Genet., 1989, vol. 77, pp. 212–216.
Naydenov, K., Senneville, S., Beaulieu, J., et al., Glacial vicariance in Eurasia: mitochondrial DNA evidence from Scots pine for complex heritage involving genetically distinct refugia at mid-northern latitudes and in Asia Minor, BMC Evol. Biol., 2007, vol. 22, pp. 7–233.
Pyhäjärvi, T., Salmela, M., and Savolainen, O., Colonization routes of Pinus sylvestris inferred from distribution of mitochondrial DNA variation, Tree Genet. Genom., 2008, no. 4, pp. 247–254.
Vidyakin, A.I., Semerikov, V.L., Polezhaeva, M.A., and Dymshakova, O.S., Spread of mitochondrial DNA haplotypes in population of Scots pine (Pinus sylvestris L.) in northern European Russia, Russ. J. Genet., 2012, vol. 48, no. 12, pp. 1267–1271.
Provan, J., Soranzo, N., Wilson, N.J., et al., A low mutation rate for chloroplast microsatellites, Genetics, 1999, vol. 153, pp. 953–947.
Walter, R. and Epperson, B.K., Geographic pattern of genetic variation in Pinus resinosa: area of greatest diversity is not the origin of postglacial populations, Mol. Ecol., 2001, vol. 10, pp. 103–111.
Vendramin, G.G., Fady, B., Gonzalez-Martinez, S.C., et al., Genetically depauperate but widespread: the case of an emblematic Mediterranean pine, Evolution, 2008, vol. 62, no. 3, pp. 680–688.
Devey, M.E., Bell, J.C., Smith, D.N., et al., A genetic linkage map for Pinus radiata based on RFLP, RAPD and microsatellite markers, Theor. Appl. Genet., 1996, vol. 92, pp. 673–679.
Vendramin, G.G., Lelli, L., Rossi, P., and Morgante, M., A set of primers for the amplification of 20 chloroplast microsatellites in Pinaceae, Mol. Ecol., 1996, vol. 5, pp. 111–114.
Nei, M., Molecular Evolutionary Genetics, New York: Columbia Univ. Press, 1987.
El Mousadik, A. and Petit, R.J., High level of genetic differentiation for allelic richness among populations of the argan tree (Argania spinosa (L.) Sceels) endemic to Morocco, Theor. App. Genet., 1996, vol. 92, pp. 832–839.
Excoffier, L., Laval, G., and Schneider, S., ARLEQUIN ver. 3.1: An Integrated Software Package for Population Genetics Data Analysis, Computational and Molecular Population Genetics Lab (CMPG). Institute Zoology, Univ. Bern, Bern, Switzerland, 2006.
Goldstein, D.B., Ruiz Linares, A., Cavalli-Sforza, L.L., Feldman, M.W., An evaluation of genetic distances for use with microsatellite loci, Genetics, 1995, vol. 139, pp. 463–471.
Pons, O. and Petit, R.J., Measuring and testing genetic differentiation with ordered versus unordered alleles, Genetics, 1996, vol. 144, pp. 1237–1245.
Rohlf E.J. Numerical Taxonomy and Multivariate Analysis System, New York: Exter Publ. LTD, 1988.
Dupanloup, I., Schneider, S., and Excoffier, L., A simulated annealing approach to define the genetic structure of populations, Mol. Ecol., 2002, vol. 11, pp. 2571–2581.
Fu, Y.X., Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection, Genetics, 1997, vol. 147, pp. 915–925.
Rogers, A.R. and Harpending, H., Population growth makes waves in the distribution of pairwise genetic differences, Mol. Biol. Evol., 1992, vol. 9, pp. 552–569.
Navascués, M., Vaxevanidou, Z., González-Martínez, S.C., et al., Chloroplast microsatellites reveal colonization and metapopulation dynamics in the Canary Island pine, Mol. Ecol., 2006, vol. 15, pp. 2691–2698.
Provan, J., Soranzo, N., Wilson, N.J., et al., Gene-pool variation in Caledonian and European Scots pine (Pinus sylvestris L.) revealed by chloroplast simple-sequence repeats, Proc. R. Soc. London, Ser. B, 1998, vol. 265, no. 1407, pp. 1697–1705.
Robledo-Arnuncio, J.J., Collada, C., Alia, R., and Gil, L., Genetic structure of montane isolates of Pinus sylvestris L. in a Mediterranean refugial area, J. Biogeogr., 2005, vol. 32, no. 4, pp. 595–605.
Bucci, G., Anzidei, M., Madaghiele, A., and Vendramin, G.G., Detection of haplotypic variation and natural hybridization in halepensis-complex pine species using chloroplast simple sequence repeat (SSR) markers, Mol. Ecol., 1998, vol. 7, no. 12, pp. 1633–1643.
Bucci, G., Gonzalez-Martinez, S.C., Le Provost, G., et al., Range-wide phylogeography and gene zones in Pinus pinaster Ait. revealed by chloroplast microsatellite markers, Mol. Ecol., 2007, vol. 16, no. 10, pp. 2137–2153.
Rodriguez-Banderas, A., Vargas-Mendoza, C.F., Buonamici, A., and Vendramin, G.G., Genetic diversity and phylogeographic analysis of Pinus leiophylla: a post-glacial range expansion, J. Biogeogr., 2009, vol. 36, no. 9, pp. 1807–1820.
Moreno-Letelier, A. and Pinero, D., Phylogeographic structure of Pinus strobiformis Engelm. across the Chihuahuan Desert filter-barrier, J. Biogeogr., 2009, vol. 36, no. 1, pp. 121–131.
Wang, X.-R., Szmidt, A., and Lindgren, D., Allozyme differentiation among populations of Pinus sylvestris (L.) from Sweden and China, Hereditas, 1991, vol. 114, pp. 219–226.
Semerikov, V.L., Podogas, A.V., and Shurkhal, A.V., Variability of allozyme loci in populations of common pine, Russian Journal of Ecology, 1993, vol. 24, no. 1, pp. 14–20.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.L. Semerikov, S.A. Semerikova, O.S. Dymshakova, K.G. Zatsepina, V.V. Tarakanov, I.V. Tikhonova, A.K. Ekart, A.I. Vidyakin, S. Jamiyansuren, R.V. Rogovtsev, L.I. Kalchenko, 2014, published in Genetika, 2014, Vol. 50, No. 6, pp. 660–669.
Rights and permissions
About this article
Cite this article
Semerikov, V.L., Semerikova, S.A., Dymshakova, O.S. et al. Microsatellite loci polymorphism of chloroplast DNA of scots pine (Pinus sylvestris L.) in Asia and eastern Europe. Russ J Genet 50, 577–585 (2014). https://doi.org/10.1134/S1022795414040127
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1022795414040127