Abstract
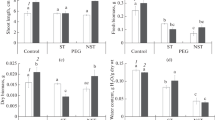
The plants from two populations (P1 and P2) of xero-halophyte Sedobassia sedoides (Pall.) Freitag & G. Kadereit (Chenopodiaceae) with С3–С4 intermediate type of photosynthesis were examined. Morphophysiological parameters were determined: dry biomass of the above-ground organs, maximum quantum yield of photosystem II (PSII), ultra- and mesostructure of the leaf, carbon isotope discrimination (δ13С) in plants grown under normal conditions and exposed to moderate salinization (0 and 200 mM NaCl). By the values of δ13С and efficiency of PSII, we did not detect significant differences between the populations. Under control conditions, the chloroplasts and mitochondria were located in Kranz-like cells of the bundle sheath of both populations in the region adjacent to vascular bundle, which is characteristic of С2 type of photosynthesis (with photorespiratory СО2 concentrating mechanism). P2 plants were notable for a greater volume of Kranz-like cells of the bundle sheath and more numerous, larger chloroplasts and mitochondria than in P1 plants. By structural leaf parameters, P1 plants may be attributed to a proto-Kranz type of photosynthesis (intermediate between С3 and С2 types) and P2 plants to С2 photosynthesis. Under salinization, the accumulation of dry biomass was reduced in both populations but more considerably in P1. The ultrastructure of organelles in both populations showed different responses to salinization, which was especially pronounced in Kranz-like cells of the bundle sheath. In P1 plants, the area of chloroplasts and mitochondria rose, whereas the area and number of chloroplasts under stress did not change in P2 plants but the area and number of mitochondria decreased. In the cells of plants from both populations, signs of degradation were observed (more pronounced in P1 plants); they were shown in chloroplasts twisting and a disturbance of their granal stacking. In P2 plants, Kranz-like cells of the bundle sheath preserved safer organelles but there occurred the cells with complete degradation of a vacuolar type. Thus, intraspecific ultra- and mesostructural differences were detected in plants from two populations of С3–С4 intermediate species S. sedoides, which reflect different stages of development of photorespiratory СО2 concentrating mechanism. Under salinization, the plants from two different populations showed unlike strategies of adaptation on the level of leaf ultra- and mesostructure.




Similar content being viewed by others
REFERENCES
Sage, R.F., Sage, T.L., and Kocacinar, F., Photorespiration and the evolution of C4 photosynthesis, Annu. Rev. Plant Biol., 2012, vol. 63, p. 19. https://doi.org/10.1146/annurev-arplant-042811-105511
Rakhmankulova, Z.F., Photorespiration: its role in the productive process and evolution of C4 plants, Russ. J. Plant Physiol., 2018, vol. 65, p. 303.
Rakhmankulova, Z.F., Shuiskaya, E.V., Voronin, P.Yu., Velivetskaya, T.A., Ignat’ev, A.V., and Usmanov, I.Yu., Comparative study on resistance of C3 and C4 xerohalophytes of the genus Atriplex to water deficit and salinity, Russ. J. Plant Physiol., 2018, vol. 65, p. 250.
Voznesenskaya, E.V., Koteyeva, N.K., Akhani, H., Roalson, E.H., and Edwards, G.E., Structural and physiological analyses in Salsoleae (Chenopodiaceae) indicate multiple transitions among C3, intermediate, and C4 photosynthesis, J. Exp. Bot., 2013, vol. 64, p. 3583. https://doi.org/10.1093/jxb/ert191
Sage, R.F., Khoshravesh, R., and Sage, T.L., From proto-Kranz to C4 Kranz: building the bridge to C4 photosynthesis, J. Exp. Bot., 2014, vol. 65, p. 3341. https://doi.org/10.1093/jxb/eru180
Bauwe, H., Photorespiration: the bridge to C4 photosynthesis, in C4photosynthesis and related CO2 concentrating mechanisms, Advances in Photosynthesis, Raghavendra, A.S. and Sage, R.F., Eds., Heidelberg-Berlin: Springer Verlag, 2011, vol. 32, p. 81.
Alonso-Cantabrana, H. and von Caemmerer, S., Carbon isotope discrimination as a diagnostic tool for C4 photosynthesis in C3–C4 intermediate species, J. Exp. Bot., 2016, vol. 67, p. 3109. https://doi.org/10.1093/jxb/erv555
Nakamura, N., Iwano, M., Havaux, M., Yokota, A., and Munekage, Y.N., Promotion of cyclic electron transport around photosystem I during the evolution of NADP-malic enzyme-type C photosynthesis in the genus Flaveria, New Phytol., 2013, vol. 199, p. 832. https://doi.org/10.1111/nph.12296
Freitag, H. and Kadereit, G., C3 and C4 leaf anatomy types in Camphorosmeae (Camphorosmoideae, Chenopodiaceae), Plant Syst. Evol., 2014, vol. 300, p. 665. https://doi.org/10.1007/s00606-013-0912-9
Mokronosov, A.T., Ontogeneticheskii aspekt fotosinteza (The Ontogenetic Aspect of Photosynthesis), Moscow: Nauka, 1981.
Lundgren, M.R., Besnard, G., Ripley, B.S., Lehmann, C.E.R., Chatelet, D.S., Kynast, R.G., Namaganda, M., Vorontsova, M.S., Hall, R.C., Elia, J., Osborne, C.P., and Christin, P.A., Photosynthetic innovation broadens the niche within a single species, Ecol. Lett., 2015, vol. 18, p. 1021. https://doi.org/10.1111/ele.12484
Shuyskaya, E., Rakhmankulova, Z., Voronin, P., Kuznetsova, N., Biktimerova, G., and Usmanov, I., Salt and osmotic stress tolerance of the C3–C4 xero-halophyte Bassia sedoides from two populations differ in productivity and genetic polymorphism, Acta Physiol. Plant., 2015, vol. 37, p. 236. https://doi.org/10.1007/s11738-015-1981-x
Rakhmankulova, Z.F., Shuiskaya, E.V., Suyundukov, Ya.T., Usmanov, I.Yu., and Voronin, P.Yu., Different responses of two ecotypes of C3–C4 xero-halophyte Bassiasedoides to osmotic and ionic factors of salt stress, Russ. J. Plant Physiol., 2016, vol. 63, p. 349.
Gupta, B. and Huang, B., Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization, Int. J. Genomics, 2014, vol. 2014, p. 701596. https://doi.org/10.1155/2014/701596
Kirchhoff, H., Chloroplast ultrastructure in plants, New Phytol., 2019, vol. 223, p. 565. https://doi.org/10.1111/nph.15730
Kondadi, A.K., Anand, R., and Reichert, A.S., Functional interplay between cristae biogenesis, mitochondrial dynamics and mitochondrial DNA integrity, Int. J. Mol. Sci., 2019, vol. 20, p. 4311. https://doi.org/10.3390/ijms20174311
Bose, J., Munns, R., Shabala, S., Gilliham, M., Pogson, B., and Tyerman, S.D., Chloroplast function and ion regulation in plants growing on saline soils: lessons from halophytes, J. Exp. Bot., 2017, vol. 68, p. 3129. https://doi.org/10.1093/jxb/erx142
Hasan, R., Ohnuki, Y., Kawasaki, M., Taniguchi, M., and Miyake, H., Differential sensitivity of chloroplasts in mesophyll and bundle sheath cells in maize, an NADP-malic enzyme-type C4 plant, to salinity stress, Plant Prod. Sci., 2005, vol. 8, p. 567. https://doi.org/10.1626/pps.8.567
Mitsuya, S., Kawasaki, M., Taniguchi, M., and Miyake, H., Light dependency of salinity-induced chloroplast degradation, Plant Prod. Sci., 2003, vol. 6, p. 219. https://doi.org/10.1626/pps.6.219
Yamane, K., Oi, T., Enomoto, S., Nakao, T., Arai, S., Miyake, H., and Taniguchi, M., Three-dimensional ultrastructure of chloroplast pockets formed under salinity stress, Plant Cell Environ., 2018, vol. 41, p. 563. https://doi.org/10.1111/pce.13115
Goncharenko, G.G., Padutov, V.E., and Potenko, V.V., Rukovodstvo po issledovaniyu khvoinykh vidov metodom elektroforeticheskogo analiza izofermentov (Manual for the Study of Coniferous Species by Electrophoretic Analysis of Isoferments), Gomel’: Polespechat’, 1989.
Yeh, F.C., Yang, R.C., and Boyle, T., POPGEN, version 1.32. Microsoft Windows-Based Freeware for Population Genetic Analysis, Edmonton: Univ. Alberta/CIFOR, 1999.
Balnokin, Yu.V., Kurkova, E.B., Khalilova, L.A., Myasoedov, N.A., and Yusufov, A.G., Pinocytosis in the root cells of a salt-accumulating halophyte Suaedaa-ltissima and its possible involvement in chloride transport, Russ. J. Plant Physiol., 2007, vol. 54, p. 797.
Mokronosov, A.T., Mesostructure and functional activity of the photosynthetic apparatus, in Mezostruktura i funktsional’naya aktivnost' fotosinteticheskogo apparata (Mesostructure and Functional Activity of the Photosynthetic Apparatus), Sverdlovsk: Ural. Gos. Univ., 1978, p. 5.
Griffiths, H., Weller, G., Toy, L.F., and Dennis, R.J., You’re sove in: bundle sheath physiology, phylogeny and evolution in C3 and C4 plants, Plant Cell Environ., 2013, vol. 36, p. 249.
Wang, P., Khoshravesh, R., Karki, S., Tapia, R., Balahadia, C., Bandyopadhyay, A., Quick, W.L., Furbank, R., Sage, T.L., and Langdale, J., Re-creation of a key step in the evolutionary switch from C3 to C4 leaf anatomy, Curr. Biol., 2017, vol. 27, p. 3278. https://doi.org/10.1016/j.cub.2017.09.040
Osborne, C.P. and Sack, L., Evolution of C4 plants: a new hypothesis for an interaction of CO2 and water relations mediated by plant hydraulics, Philos. Trans. R. Soc. Lond. B., 2012, vol. 367, p. 583.
Gowik, U. and Westhoff, P., The path from C3 to C4 photosynthesis, Plant Physiol., 2011, vol. 155, p. 56. https://doi.org/10.1104/pp.110.165308
Muhaidat, R., Sage, T.L., Frohlich, M.W., Dangler, N.G., and Sage, R.F., Characterization of C3–C4 intermediate species in the genus Heliotropium L. (Boraginaceae): anatomy, ultrastructure and enzyme activity, Plant Cell Environ., 2011, vol. 34, p. 1723.
Manaa, A., Goussi, R., Derbali, W., Cantamessa, S., Abdelly, C., and Barbato, R., Salinity tolerance of quinoa (Chenopodium quinoa Willd) as assessed by chloroplast ultrastructure and photosynthetic performance, Environ. Exp. Bot., 2019, vol. 162, p. 103. https://doi.org/10.1016/j.envexpbot.2019.02.012
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies involving animals or human participants performed by any of the authors.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
Additional information
Translated by N. Balakshina
Rights and permissions
About this article
Cite this article
Rakhmankulova, Z.F., Shuyskaya, E.V., Khalilova, L.A. et al. Ultra- and Mesostructural Response to Salinization in Two Populations of С3–С4 Intermediate Species Sedobassia sedoides. Russ J Plant Physiol 67, 835–844 (2020). https://doi.org/10.1134/S1021443720040135
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443720040135