Abstract
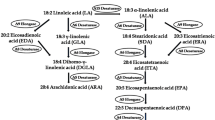
Cyanobacterium sp. IPPAS B-1200 is characterized by a high content of rare fatty acids (FAs), both myristic (14:0–30%) and myristoleic (14:1Δ9–10%) in the membrane lipids. Thus, short-chain FAs reach 40% of the sum of all FAs in cells, which is unusual for Cyanobacteria. Monounsaturated palmitoleic acids (16:1Δ9) also reach 40% of the sum of the FAs. We determined the complete nucleotide sequence of the genome of this cyanobacterium and found the only gene for the acyl-lipid Δ9-desaturase, desC1. We cloned this gene and characterized its specificity to the length of the substrate using heterologous expression in Escheriсhia coli. The results show that DesC1 nonspecifically generates olefin bond in FAs with a length of 14, 16, and 18 carbon atoms. This finding confirms that all monoesterifed FAs in Cyanobacterium sp. IPPAS B-1200 are generated by one acyl-lipid Δ9-desaturase.
Similar content being viewed by others
Abbreviations
- FA(s):
-
fatty acid(s)
- GC:
-
gas chromatography
- MS:
-
mass spectrometry
- PAGE:
-
polyacrylamide gel electrophoresis
- PCR:
-
polymerase chain reaction
References
Georgianna, D.R. and Mayfield, S.P., Exploiting diversity and synthetic biology for the production of algal biofuels, Nature, 2012, vol. 488, pp. 329–335.
Sarsekeyeva, F., Zayadan, B.K., Usserbaeva, A., Bedbenov, V.S., Sinetova, M.A., and Los, D.A., Cyanofuels—biofuels from cyanobacteria: reality and perspectives, Photosynth. Res., 2015, vol. 125, pp. 329–340.
Sarsekeyeva, F.K., Usserbaeva, A.A., Zayadan, B.K., Mironov, K.S., Sidorov, R.A., Kozlova, A.Yu., Kupriyanova, E.V., Sinetova, M.A., and Los, D.A., Isolation and characterization of a new cyanobacterial strain with a unique fatty acid composition, Adv. Microbiol., 2014, vol. 4, pp. 1033–1043.
Sinetova, M.A., Bolatkhan, K., Sidorov, R.A., Mironov, K.S., Skrypnik, A.N., Kupriyanova, E.V., Bolatkhan, K.Z., Shumskaya, M., and Los, D.A., Polyphasic characterization of the thermotolerant cyanobacterium Desertifilum sp. strain IPPAS B-1220, FEMS Microbiol. Lett., 2017, vol. 364. doi 10.1093/femsle/fnx027
Polashock, J.J., Chin, C.K., and Martin, C.E., Expression of the yeast Δ-9 fatty acid desaturase in Nicotiana tabacum, Plant Physiol., 1992, vol. 100, pp. 894–901.
Orlova, I.V., Serebriiskaya, T.S., Popov, V., Merkulova, N., Nosov, A.M., Trunova, T.I., Tsydendambaev, V.D., and Los, D.A., Transformation of tobacco with a gene for the thermophilic acyl–lipid desaturase enhances the chilling tolerance of plants, Plant Cell Physiol., 2003, vol. 44, pp. 447–450.
Liu, X., Sheng, J., and Curtiss, R., III, Fatty acid production in genetically modified cyanobacteria, Proc. Natl. Acad. Sci. USA, 2011, vol. 108, pp. 6899–6904.
Choi, Y.J. and Lee, S.Y., Microbial production of short-chain alkanes, Nature, 2013, vol. 502, pp. 571–574.
Los, D.A., Fatty Acid Desaturases, Moscow: Scientific World, 2014.
Amiri, R.M., Yur’eva, N.O., Shimshilashvili, K.R., Goldenkova-Pavlova, I.V., Pchelkin, V.P., Kuznitsova, E.I., Tsydendambaev, V.D., Trunova, T.I., Los, D.A., Jouzani, G.S., and Nosov, A.M., Expression of acyl-lipid Δ12-desaturase gene in prokaryotic and eukaryotic cells and its effect on cold stress tolerance of potato, J. Integr. Plant Biol., 2010, vol. 52, pp. 289–297.
Los, D.A. and Mironov, K.S., Modes of fatty acid desaturation in cyanobacteria: an update, Life, 2015, vol. 5, pp. 554–567.
Starikov, A.Y., Usserbaeva, A.A., Sinetova, M.A., Sarsekeyeva, F.K., Zayadan, B.K., Ustinova, V., Kupriyanova, E.V., Los, D.A., and Mironov, K.S., Draft genome sequence of Cyanobacterium sp. strain IPPAS B-1200 with a unique fatty acid composition, Genome Announc., 2016, vol. 4: e01306–16. doi 10.1128/genomeA.01306-16
Pereira, F.L., Soares, S.C., Dorella, F.A., Leal, C.A., and Figueiredo, H.C., Evaluating the efficacy of the new Ion PGM Hi-Q Sequencing Kit applied to bacterial genomes, Genomics, 2016, vol. 107, pp. 189–198.
Williams, J.G.K., Construction of specific mutations in photosystem II photosynthetic reaction center by genetic engineering methods in Synechocystis 6803, Methods Enzymol., 1988, vol. 167, pp. 766–778.
Maniatis, T., Sambrook, J., and Fritsch, E.F., Molecular Cloning: A Laboratory Manual, Cold Springer Harbor: Cold Springer Harbor Lab., 1989.
Bankevich, A., Nurk, S., Antipov, D., Gurevich, A.A., Dvorkin, M., Kulikov, A.S., Lesin, V.M., Nikolenko, S.I., Pham, S., Prjibelski, A.D., Pyshkin, A.V., Sirotkin, A.V., Vyahhi, N., Tesler, G., Alekseyev, M.A., and Pevzner, P.A., SPAdes: a new crossmark genome assembly algorithm and its applications to single-cell sequencing, J. Comput. Biol., 2012, vol. 19, pp. 455–477.
Edgar, R.C., MUSCLE: multiple sequence alignment with high accuracy and high throughput, Nucleic Acids Res., 2004, vol. 32, pp. 1792–1797.
McWilliam, H., Li, W., Uludag, M., Squizzato, S., Park, Y.M., Buso, N., Cowley, A.P., and Lopez, R., Analysis tool web services from the EMBL-EBI, Nucleic Acids Res., 2013, vol. 41: W597–600. doi 10.1093/nar/gkt376
Li, H. and Homer, N., A survey of sequence alignment algorithms for next-generation sequencing, Brief Bioinform., 2010, vol. 11, pp. 473–483.
Laemmli, U.K., Cleavage of structural proteins during the assembly of the head of bacteriophage T4, Nature, 1970, vol. 227, pp. 680–685.
Gurevich, A., Saveliev, V., Vyahhi, N., and Tesler, G., QUAST: quality assessment tool for genome assemblies, Bioinformatics, 2013, vol. 29, pp. 1072–1075.
Wada, H., Avelange-Macherel, M.H., and Murata, N., The desA gene of the cyanobacterium Synechocystis sp. strain PCC6803 is the structural gene for delta 12 desaturase, J. Bacteriol., 1993, vol. 175, pp. 6056–6058.
Sakamoto, T., Los, D.A., Higashi, S., Wada, H., Nishida, I., Ohmori, M., and Murata, N., Cloning of Δ3 desaturase from cyanobacteria and its use in altering the degree of membrane-lipid unsaturation, Plant Mol. Biol., 1994, vol. 26, pp. 249–263.
Tasaka, Y., Gombos, Z., Nishiyama, Y., Mohanty, P., Ohba, T., Ohki, K., and Murata, N., Targeted mutagenesis of acyl-lipid desaturases in Synechocystis: evidence for the important roles of polyunsaturated membrane lipids in growth, respiration and photosynthesis, EMBO J., 1996, vol. 15, pp. 6416–6425.
Chintalapati, S., Prakash, S., Gupta, P., Ohtani, S., Suzuki, I., Sakamoto, T., Murata, N., and Shivaji, S., A novel Δ9 acyl-lipid desaturase, DesC2, from cyanobacteria acts on fatty acids esterified to the sn-2 position of glycerolipids, Biochem. J., 2006, vol. 398, pp. 207–214. doi 10.1042/BJ20060039
Shih, P.M., Wu, D., Latifi, A., Axen, S.D., Fewer, D.P., Talla, E., Calteau, A., Cai, F., Tandeau de Marsac, N., Rippka, R., Herdman, M., Sivonen, K., Coursin, T., Laurent, T., Goodwin, L., et al., Improving the coverage of the cyanobacterial phylum using diversity-driven genome sequencing, Proc. Natl. Acad. Sci. USA, 2013, vol. 110, pp. 1053–1058.
Panpoom, S., Los, D.A., and Murata, N., Biochemical characterization of a Δ12 acyl-lipid desaturase after overexpression of the enzyme in Escherichia coli, Biochim. Biophys. Acta, 1996, vol. 1390. pp. 323–332.
Raetz, C.R., Enzymology, genetics, and regulation of membrane phospholipid synthesis in Escherichia coli, Microbiol. Rev., 1978, vol. 42, pp. 614–659.
Feng, Y. and Cronan, J.E., Escherichia coli unsaturated fatty acid synthesis complex transcription of the fabA gene and in vivo identification of the essential reaction catalyzed by FabB, J. Biol. Chem., 2009, vol. 284, pp. 29526–29535.
Starikov, A.Yu., Userbaeva, A.A., Lapina, S.S., Mironov, K.S., Maslova, I.P., Pchelkin, V.P., Zayadan, B.K., Sinetova, M.A., and Los, D.A., Substrate specificity of acyl-lipid Δ9-desaturase from Prochlorothrix hollandica cyanobacterium producing myristoleic acid, Russ. J. Plant Phys., 2017, vol. 64, pp. 560–565.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Fiziologiya Rastenii, 2018, Vol. 65, No. 4, pp. 270–278.
The article was translated by authors.
Rights and permissions
About this article
Cite this article
Starikov, A.Y., Usserbaeva, A.A., Mironov, K.S. et al. Substrate Specificity of Acyl-Lipid Δ9-Desaturase from Cyanobacterium sp. IPPAS B-1200, a Cyanobacterium with Unique Fatty Acid Composition. Russ J Plant Physiol 65, 490–497 (2018). https://doi.org/10.1134/S102144371804009X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S102144371804009X