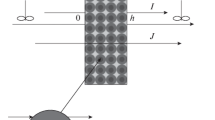
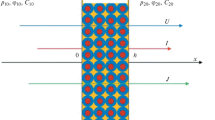
Abstract
At current densities exceeding the limiting current density, H+ and OH− ions are generated at the interface of the ion-exchange membrane with a depleted solution as a result of the dissociation of water molecules. At present, it is generally accepted that water splitting occurs in a thin (a few nanometers) layer inside the membrane, this reaction being catalytic in nature. The mathematical model of ion transport in the diffusion layer near the membrane surface has been constructed and numerically studied under conditions when dissociation and recombination processes involving water molecules and H+ and OH− ions occur simultaneously. It has been shown that in overlimiting current regimes under very high voltages, intense noncatalytic dissociation of water molecules in the extended space charge region of the depleted solution can occur irrespective of the catalytic splitting of water. Since this region has macroscopic dimensions, the rate of noncatalytic water dissociation is comparable with the rate of the corresponding catalytic process. The obtained results significantly supplement modern concepts of the mechanism of generation of H+ and OH− ions in membrane systems, showing that this process can proceed not only in accordance with the conventional mechanism with the catalytic participation of functional groups and/or other compounds, but also via the noncatalytic mechanism that has not been taken into account to the present.
Similar content being viewed by others
References
N. O. Chubyr’, A. V. Kovalenko, and M. Kh. Urtenov, Two-Dimensional Mathematical Models of Binary-Electrolyte Transport in Membrane Systems (KGTU, Krasnodar, 2012) [in Russian].
V. J. Frilette, J. Phys. Chem. 60, 435 (1956).
T. R. E. Kressman and F. L. Tye, Discuss. Faraday Soc. 21, 185 (1956).
N. W. Rosenberg and C. E. Tirrell, Ind. Eng. Chem. 49, 780 (1957).
B. A. Cooke, Electrochim. Acta 4, 179 (1961).
H. P. Gregor and M. A. Peterson, J. Phys. Chem. 68, 2201 (1964).
M. Block and J. A. Kitchener, J. Electrochem. Soc. 113, 947 (1966).
Y. Oda and T. Yawataya, Desalination 5, 129 (1968).
K. S. Spiegler, J. Leibovitz, and J. Sinkovic, Pontificiae 40, 727 (1976).
V. P. Greben, N. Y. Pivovarov, N. Y. Kovarskii, and G. V. Nefedova, Sov. J. Phys. Chem 52, 2641 (1978).
R. Simons, Desalination 28, 41 (1979).
R. Simons, Nature 280, 824 (1979).
R. Simons, Electrochim. Acta 29, 151 (1984).
V. I. Zabolotskii, N. V. Shel’deshov, and N. P. Gnusin, Russ. Chem. Rev. 57, 801 (1988).
V. I. Zabolotskii and V. V. Nikonenko, Ion Transport in Membranes (Nauka, Moscow, 1996) [in Russian].
V. I. Zabolotskii, V. V. Nikonenko, N. M. Korzhenko, et al., Russ. J. Electrochem. 38, 810 (2002).
V. I. Zabolotskii, K. A. Lebedev, and N. V. Shel’-deshov, Nauchn. Zh. KubGAU, No. 124, 210 (2016).
A. V. Kovalenko, M. Kh. Urtenov, N. M. Seidova, and A. V. Pis’menskii, Nauchn. Zh. KubGAU, No. 121, 1929 (2016).
I. Ruhinstein and L. Shtilman, J. Chem. Soc., Faraday Trans. 2 75, 231 (1979).
M. Kh. Urtenov, E. V. Kirillova, N. M. Seidova, and V. V. Nikonenko, J. Phys. Chem. 111, 14208 (2007).
A. V. Sokirko and Yu. I. Kharkats, Elektrokhimiya 25, 232 (1989).
N. D. Pismenskaya, V. V. Nikonenko, E. I. Belova, et al., Russ. J. Electrochem. 43, 307 (2007).
N. V. Sheldeshov, V. I. Zabolotskii, A. V. Bespalov, et al., Pet. Chem. 57, 518 (2017).
A. V. Kovalenko, A. M. Uzdenova, M. Kh. Urtenov, and V. V. Nikonenko, Mathematical Modeling of Physicochemical Processes in the Comsol Multiphysics 5.2 Environment (Lan’, St. Petersburg, 2017) [in Russian].
V. I. Zabolotskii, L. Novak, A. V. Kovalenko, et al., Pet. Chem. 57, 779 (2017).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © M.Kh. Urtenov, A.V. Pismensky, V.V. Nikonenko, A.V. Kovalenko, 2018, published in Membrany i Membrannye Tekhnologii, 2018, Vol. 8, No. 1, pp. 24–33.
Rights and permissions
About this article
Cite this article
Urtenov, M.K., Pismensky, A.V., Nikonenko, V.V. et al. Mathematical Modeling of Ion Transport and Water Dissociation at the Ion-Exchange Membrane/Solution Interface in Intense Current Regimes. Pet. Chem. 58, 121–129 (2018). https://doi.org/10.1134/S0965544118020056
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965544118020056