Abstract
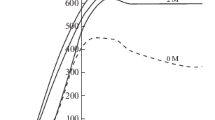
The electrochemical behavior of tungsten and a VNZhK alloy (wt %: 90 W, 7.2 Ni, 1.8 Fe, 1 Co) in 0.5–1.5 M solutions of ammonium carbonate is studied by linear voltammetry in the potentiodynamic mode. An increase in the maximum anodic current density for the oxidation of tungsten and VNZhK alloy with the ammonium carbonate concentration is revealed. The electrochemical dissolution of the VNZhK alloy wastes in a 1.0 M solution of ammonium carbonate is studied by galvanostatic electrolysis and electrolysis using sinusoidal industrial-frequency alternating current. The advantage of a step-by-step application of direct and alternating currents is substantiated. For the processing of the VNZhK alloy wastes under the step-by-step action of direct and alternating currents, the transition of tungsten from the alloy to an ammonium carbonate solution is accompanied by concentrating the iron-group metals in a microdispersed electrolysis slime. A principal technological scheme is proposed for tungsten recovery from the wastes of heavy tungsten alloys to form ammonium paratungstate as the final product.




Similar content being viewed by others
REFERENCES
B. Zeiler, A. Bartl, W. Schubert, “Recycling of tungsten: current share, economic limitations, technologies and future potential,” Intern. J. Refract. Metals Hard Mater. 98, 105546 (2021).
Z. Han, A. Golev, and M. Edraki, “A review of tungsten resources and potential extraction from mine waste,” Minerals 11, 701 (2021).
L. Shen, X. Li, D. Linberg, and P. Taskinen, “Tungsten extractive metallurgy: a review of processes and their challenges for sustainability,” Miner. Eng. 142, 105934 (2019).
A. Hool, S. van Nielen, D. Schrijvers, and S. Ganzeboom, “How companies improve critical raw material circularity: 5 use cases (findings from the international round table on materials criticality),” Miner. Econom., April (2022). https://doi.org/10.1007/S135563-022-00315-5
Technology AO Gidrometallurg (2022). http://hidromet.ru/support.html. Accessed June 29, 2022.
S. S. Kalyan Kamal, J. Vimala, Y. Sushma, P. K. Sahoo, and M. Sankaranarayana, “Large scale synthesis of nanocrystalline tungsten powders through recycling of heavy tungsten alloy scrap,” Mater. Today Commun. 11, 174–178 (2017).
H. Koohestani, “Characterization of TiO2/WO3 composite produced with recycled WO3 nanoparticles from W–Ni–Fe alloy,” Mater. Chem. Phys. 229, 251–256 (2019).
S. S. Kalyan Kamal, P. K. Sahoo, J. Vimala, B. Shanker, P. Ghosal, and L. Durai, “Synthesis of high purity tungsten nanoparticles from heavy tungsten alloy scrap by selective precipitation and reduction route,” J. Alloys Compd. 678, 403–409 (2016).
V. V. Parshutin, “Corrosion and electrochemical behavior of pseudo-alloys on the basis of tungsten and their components,” Surf. Eng. Appl. Electrochem. 44 (6), 446–461 (2008).
G. N. Shrinavasan, A. Varadharay, and J. A. M. Abdul Kader, “Anodic leaching of tungsten alloy swarf: a statistical approach,” J. Appl. Electrochem. 24, 1191–1193 (1994).
S. Hairunnisha, G. K. Sendil, J. Prabhakar Rethinaraj, G. N. Srinivasan, P. Adaikkalam, and S. Kulandaisamy, “Studies on the preparation of pure ammonium paratungstate from tungsten alloy scrap,” Hydrometallurgy 85, 67–71 (2007).
O. G. Kuznetsova, A. M. Levin, M. A. Sevastyanov, O. I. Tsybin, and A. O. Bolshikh, “Electrochemical processing of a heavy W–Ni–Fe alloy by direct and alternating current in ammonia–alkali solutions,” Russ. Metall. (Metally) 5, 586–593 (2021).
O. G. Kuznetsova, A. M. Levin, M. A. Sevastyanov, O. I. Tsybin, and A. O. Bolshikh, “Electrochemical oxidation of a heavy tungsten-containing VNZh-type alloy and its components in ammonia–alkali electrolytes,” Russ. Metall. (Metally) 5, 507–510 (2019).
K. Vadasdi, “Effluent-free manufacture of ammonium paratungstate (APT) by recycling the by-products,” Intern. J. Refract. Metals Hard Mater. 13, 45–59 (1995).
V. R. Guriev, “Investigation and development of technologies for waste processing of the production of refractory and heavy nonferrous metals using electrochemical methods,” Extended Abstract of Cand. Sci. (Eng.) Dissertation, Vladikavkaz, 2001.
V. A. Reznichenko, A. A. Palant, G. I. Anufrieva, R. A. Guriev, and V. K. Gavrilov, “Investigation of the electrochemical dissolution of multiphase tungsten-based alloys,” Izv. Akad. Nauk SSSR, Met., No. 2, 32–35 (1985).
V. Kovalenko and V. Kotoc, “Investigation of the anodic behavior of W-based superalloy for electrochemical selective treatment,” East.-Europ. J. Enter. Technol. 108 (6/12), 61–66 (2020).
V. Kovalenko and V. Kotoc, “Selective anodic treatment or W(WC)-based superalloy scrap,” East.-Europ. J. Enter. Technol. 85 (1/5), 53–58 (2017).
G. V. Chernyak and K. B. Povarova, Tungsten in Ammunition (TsNIIKhM, Moscow, 2014).
O. G. Kuznetsova and A. M. Levin, “Electrochemical behavior of components of binding phases of heavy tungsten alloys in ammonium carbonate solutions,” in Proceedings of International Scientific Practical Conference on the Development and Application of Science Intensive Technologies in Interests of Modernization of Modern Society (AETERNA, Ufa, 2022), Part 2, pp. 8–11.
C. D. Vanderpool and T. K. Kim, “Electrolytic method for producing ammonium paratungstate from cemented tungsten carbide,” US Patent 504408, 1990.
O. G. Kuznetsova, A. M. Levin, M. A. Sevost’yanov, O. I. Tsybin, and A. O. Bol’shikh, “Electrical conductivity of tungsten-containing ammonium hydroxide solutions,” Russ. Metall. (Metally), No. 9, 971–975 (2019).
A. M. Levin, O. G. Kuznetsova, and M. A. Sevost’yanov, “Electroconductivity of tungsten-containing aqueous solutions of ammonium carbonate,” in Proceedings of International Scientific Practical Conference on the Theory and Practice of Scientific Activity Modernization under Digitalization Conditions (OMEGASCIENCE, Ufa, 2021), pp. 25–28.
E. Lassner, “From concentrates and scrap to highly pure ammonium paratungstate (APT),” Intern. J. Refract. Metals Hard Mater. 13, 35–44 (1995).
O. G. Kuznetsova, A. M. Levin, M. A. Sevastyanov, O. I. Tsybin, and A. O. Bolshikh, “Electrochemical processing of heavy tungsten alloy wastes for obtaining a microdispersed iron–nickel base powder by using alternating current,” IOP Conf. Ser.: J. Phys. 1942, 012056 (2020).
S. L. Berezina, V. N. Goryacheva, N. N. Dvulichanskaya, V. I. Ermolaeva, and L. E. Slyn’ko, “Anodic behavior of tungsten with different structures in an alkaline electrolyte,” Usp. Sovrem. Estestvoznan. Khim. Nauki, No. 4, 7–11 (2017).
A. B. Kilimnik and E. Yu. Ostrozhkova, Electrochemical Synthesis of Nanodispersed Powders of Metal Oxides (TGTU, Tambov, 2012).
Yu. A. Abramenko, V. V. Dem’yan, I. Yu. Zhukova, and E. N. Panina, “Electrochemical synthesis of nanostructured powders of tungsten and molybdenum oxides during electrolysis by alternating asymmetric sinusoidal current in a solution of potassium alkali,” in Proceedings of International Scientific Practical Conference on Actual Problems of Science and Technology (DSTU, Rostov-on-Don, 2017), pp. 178, 179.
Funding
This work was carried out in terms of state assignment no. 075-00715-22-00.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Kuznetsova, O.G., Levin, A.M., Konushkin, S.V. et al. Electrochemical Processing of the Wastes of Heavy Tungsten Alloys in Ammonium Carbonate Solutions. Russ. Metall. 2023, 7–12 (2023). https://doi.org/10.1134/S003602952301007X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602952301007X