Abstract
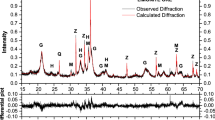
The calciothermic reduction of metals from manganese (MnNb2O6) and iron (FeNb2O6) niobates, which are analogs of natural columbite (Fe, Mn)(Nb, Ta)2O6 contained in commercial rare metal ores, is studied. Thermodynamic simulation using the HSC Chemistry software package is used to calculate the change in the Gibbs free energy, the equilibrium constants, and the thermal effects of the interaction reactions of MnNb2O6, FeNb2O6, and Nb2O5 with calcium. The probability of formation of both niobium and its alloys with iron or manganese is found to be high. The changes in the equilibrium phase compositions of the oxide–calcium interaction products as functions of temperature (in the range 1000–2500°C) and the ratios of reagents are estimated. According to the calculations of the adiabatic temperature and the specific heat effect, the reduction of metals from FeNb2O6 is feasible under “out-of-furnace” process conditions, and the reduction from MnNb2O6 would require preheating a charge to 600–800°C. During continuous heating up to 1200°C, the temperatures of the thermal effects related to reagent transformations and phase formation in the interaction products are detected by combined thermogravimetry and differential thermal analysis. The complete reduction of manganese and iron niobates is found to occur with the participation of molten calcium at a molar niobate/calcium ratio of 6 with the formation of metallic niobium and its intermetallic compounds, NbMn2 or FeNb, respectively. At excess reducing agent, solid-phase products hinder the diffusion of the calcium melt to the reaction surface, which slows down the completion of the process and promotes the development of secondary interactions between intermediate oxides (NbO, Mn0.95O, Mn2.907O4) and calcium oxide. To complete the reduction process, it is necessary to increase the temperature or the heating time.





Similar content being viewed by others
REFERENCES
T. S. Kumar, T. Balaji, S. R. Kumar, A. Kumar, and T. L. Prakash, “Issues and challenges in the preparation of niobium for strategic applications,” Int. J. Chem. Stud. 1 (2), 86–89 (2013).
C. K. Guptha and P. K. Jena, “Preparation of niobium (columbium) pentoxide,” Trans. TME-AIME 230, 1433–1438 (1964).
J. C. Sehra, D. K. Bose, and P. K. Jena, “Preparation of niobium and tantalum metal powders by calciothermic reduction of their pentoxides,” Trans. Ind. Inst. Met. 21 (1), 21–23 (1968).
M. W. D. Mendes, A. C. P. Santos, F. F. P. Medeiros, C. Alves, A. G. P. Da Silva, and U. U. Gomes, “Aluminothermic reduction of niobium pentoxide in a hydrogen plasma furnace,” Mater. Sci. Forum 514–516, 599–603 (2006).
C. A. Nunes, D. G. Pinatti, and A. Robin, “Nb–Ta alloys by aluminothermic reduction of Nb2O5/Ta2O5 mixtures and electron beam melting,” Int. J. Refract. Met. Hard Mater. 17 (4), 305–314 (1999).
C. P. D. Lazzari, O. M. Cintho, and J. D. T. Capocchi, “Kinetics of the non-isothermal reduction of Nb2O5 with aluminum,” ISIJ Int. 45 (1), 19–22 (2005).
T. V. Osinkina, S. A. Krasikov, E. M. Zhilina, S. N. Agafonov, L. B. Vedmid, and S. V. Zhidovinova, “Influence of niobium and tantalum on the phase formation during the metallothermic interaction of aluminum with titanium dioxide,” Rasplavy, No. 5, 554–561 (2018).
O. H. Toru, S. Iwata, M. Imagunbai, and M. Maeda, “Production of niobium powder by preform reduction process using various fluxes and alloy reductant,” ISIJ Int. 44 (2), 285–293 (2004).
T. S. Kumar, S. R. Kumar, M. L. Rao, and T. L. Prakash, “Preparation of niobium metal powder by two stage magnesium vapor reduction of niobium pentoxide,” J. Metallurgy 2013, 1–6 (2013).
T. Izumi, “Processing of metallic material prospect to the next generation. Powder production prospect of tantalum and niobium,” Kinzoku (Met. Sci. Technol.) 69 (10), 875–880 (1999).
I. Park, T. H. Okabe, Y. Waseda, H. S. Yu, and O. Y. Lee, “Semi-continuous production of niobium powder by magnesiothermic reduction of Nb2O5,” Mater. Trans. 42 (5), 850–855 (2001).
T. H. Okabe, S. Iwata, M. Imagunbai, and M. Maeda, “Production of niobium powder by magnesiothermic reduction of feed preform,” ISIJ Int. 43 (12), 1882–1889 (2003).
M. Baba, T. Kikuchi, and R. O. Suzuki, “Niobium powder synthesized by calciothermic reduction of niobium hydroxide for use in capacitors,” J. Phys. Chem. Solids. 78, 101–109 (2015).
M. Baba, Y. Ono, and R. O. Suzuki, J. Phys. Chem. Solids. 66 (2–4), 466–470 (2005). https://doi.org/10.1016/jjpcs.2004.06.042
R. O. Suzuki, M. Aizawa, and K. Ono, J. Alloys Compd. 288 (1–2), 173–182 (1999). https://doi.org/10.1016/S0925-8388(99)00116-4
T. H. Okabe, H. Park, K. T. Jacob, and Y. Waseda, J. Alloys Compd. 288, 200–210 (1999). https://doi.org/10.1016/S0925-8388(99)00130-9
A. V. Kasimtsev, Yu. V. Levinsky, and S. N. Yudin, Tsvetn. Met., No. 1, 47–57 (2021). https://doi.org/10.r7580/tsm.2021.01.05
V. M. Orlov and M. V. Kryzhanov, Neorg. Mater. 56 (7), 774–779 (2020). https://doi.org/10.31857/S0002337X2007012X
V. M. Orlov and M. V. Kryzhanov, Neorg. Mater. 57 (1), 33–40 (2021). https://doi.org/10.31857/S0002337X20120131
A. N. Mansurova, V. M. Chumarev, L. I. Leontiev, R. I. Gulyaeva, and N. I. Selmenskikh, “Phase transformations during the interaction of Nb2O5 and FeNb2O6 with aluminum,” Metally, No. 6, 15–21 (2012).
A. Roine, HSC 6.0 Chemistry. Chemical Reactions and Equilibrium Software with Extensive Thermochemical Database and Flowsheet Simulation (Outokumpu Research Oy, Pori, 2006).
A. N. Mansurova, R. I. Gulyaeva, V. M. Chumarev, and V. P. Mar’evich, “Thermochemical properties of MnNb2O6,” J. Therm. Anal. Calorim. 101 (1), 45–47 (2010).
C. He, Y. Qin, and F. Stein, J. Phase Equilib. Diffus. 38, 771–787 (2017). https://doi.org/10.1007/s11669-017-0566-3
S. Liu, B. Hallstedt, D. Music, and Y. Du, Calphad 38, 43–58 (2012). https://doi.org/10.1016/j.calphad.2012.03.004
A. N. Mansurova, R. I. Gulyaeva, V. M. Chumarev, and S. A. Petrova, J. Alloys Compd. 695, 2483–2487 (2017). https://doi.org/10.1016/JJALLCOM.2016.11.148
Powder Diffraction File PDF4 + ICDD Release (2016).
H. M. Rietveld, J. Appl. Crystallogr., No. 2, 65–71 (1969). https://doi.org/10.1107/S0021889869006558
DIFFRAC Plus : TOPAS Bruker AXS GmbH, Ostliche. Rheinbruckenstrafie 50, D-76187 (Karlsruhe, 2008).
V. A. Podergin, Metallothermic Systems (Metallurgiya, Moscow, 1972).
H. A. Wriedt, “The Ca–O (Calcium–Oxygen) System,” Bull. Alloy Phase Diagrams 6 (4), 337–342 (1985).
V. V. Rodyakin, Calcium, Its Compounds and Alloys (Metallurgiya, Moscow, 1967).
X. Yan, P. Brož, J. Vřeštál, et al., J Alloys Compd. 865, 158715 (2021). https://doi.org/10.1016/jjallcom.2021.158715
A.R. Massive and R. J. Pérez, “Thermodynamic evaluation of the Nb–O system,” Quantum Technol. AB 2, 1–31 (2006).
E. Paul and L. J. Schwarzendruber, Byull. Alloy Phase Diagrams 7 (3), 248–254 (1986). https://doi.org/10.1007/BF02869000
Funding
This work was supported by the Russian Foundation for Basic Research (project no. 18-29-24051_mk) and was performed on the equipment of the Center for Collective Use URAL-M.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by K. Shakhlevich
Rights and permissions
About this article
Cite this article
Sergeeva, S.V., Gulyaeva, R.I., Udoeva, L.Y. et al. Thermodynamic Simulation and Experimental Investigation of the Calciothermic Reduction of Metals from Manganese and Iron Niobates. Russ. Metall. 2022, 909–918 (2022). https://doi.org/10.1134/S0036029522080146
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036029522080146