Abstract
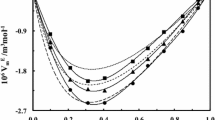
The heat capacity and density of the ternary solutions of NaI–KI–N-methylpyrrolidone (MP), NaI–RbI–MP, KI–RbI–MP, and KI–BaI2–MP at 298.15 K were studied by calorimetry and densimetry. The changes in the heat capacity and volume during the formation of the ternary systems from binary solutions were calculated and discussed. Ion association was found to dominate during the mixing of electrolyte solutions of this type in MP, with ion resolvation occasionally producing a significant effect on ion association.
Similar content being viewed by others
References
L. Yu. Rassokhina and A. N. Novikov, Russ. J. Phys. Chem. A 87, 1825 (2013).
B. V. Mikhailin, A. F. Vorob’ev, and V. A. Vasilev, Russ. J. Phys. Chem. A 56, 1183 (1982).
A. F. Kapustinskii, M. S. Stakhanova, and V. A. Vasilev, Russ. Chem. Bull. 9, 1933 (1960).
V. A. Vasilev and A. N. Novikov, Russ. J. Phys. Chem. A 67, 1253 (1993).
A. N. Novikov, O. F. Lenina, and V. A. Vasilev, Russ. J. Phys. Chem. A 83, 392 (2009).
Problems of Solution Chemistry. Ionic Solvation, Ed. by G. A. Krestov, N. P. Novoselov, I. S. Perelygin, et al. (Nauka, Moscow, 1987) [in Russian].
Y. Marcus, J. Solution Chem. 33, 549 (2004).
K. Burger, Experimental Methods for Investigation of Solvation, Ionic and Complex Formation Reactions in Nonaqueous Solutions (Akadémiai Kiadó, Budapest, 1982).
A. N. Novikov and V. A. Vasilev, Russ. J. Phys. Chem. A 80, 505 (2006).
A. N. Novikov, O. F. Lenina, and V. A. Vasilev, Russ. J. Phys. Chem. A 81, 1743 (2007).
A. N. Novikov and V. A. Vasilev, Russ. J. Phys. Chem. A 82, 1071 (2008).
Achievements and Problems of Solvation Theory: Structural and Thermodynamical Aspects, Ed. by A. M. Kutepov (Nauka, Moscow, 1998) [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.N. Novikov, L.Yu. Rassokhina, 2015, published in Zhurnal Fizicheskoi Khimii, 2015, Vol. 89, No. 12, pp. 1881–1883.
Rights and permissions
About this article
Cite this article
Novikov, A.N., Rassokhina, L.Y. Ionic molecular interactions in solutions of alkali metal iodides in N-methylpyrrolidone at 298.15 K according to calorimetry and densimetry data. Russ. J. Phys. Chem. 89, 2226–2228 (2015). https://doi.org/10.1134/S0036024415120213
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024415120213