Abstract
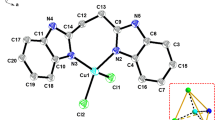
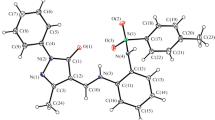
Zinc(II) complexes 2, 3, and 5 with redox-amphoteric o-indophenol ligands were prepared. The molecular structures of tetracoordinate complex 2 and hexacoordinate complex 3 were determined by single-crystal X-ray diffraction. The antioxidant properties of indophenols and their complexes were studied by cyclic voltammetry (CVA) and EPR spectroscopy. Complexation of indophenols increases the oxidation potentials by more than 0.84 V and leads to the formation of stable metal-containing radicals.





Similar content being viewed by others
REFERENCES
S. N. Lyubchenko, V. V. Litvinov, T. A. Ryskina, et al., Zh. Obshch. Khim. 60, 1618 (1990).
L. P. Olekhnovich, E. P. Ivakhnenko, S. N. Lyubchenko, et al., Ross. Khim. Zh. 48, 103 (2004).
A. N. Erickson and S. N. Brown, Dalton Trans. 47, 15583 (2018). https://doi.org/10.1039/C8DT03392G
T. Bally, Nat. Chem. 2, 165 (2010). https://doi.org/10.1038/nchem.564
G. M. Sheldrick, Acta Crystallogr., Sect A 64, 112 (2008). https://doi.org/10.1107/S0108767307043930
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian 16 (Revision A.03), Gaussian, Inc., Wallingford (CT), 2016.
W. Kohn and L. J. Sham, Phys. Rev. 140, A1133 (1965). https://doi.org/10.1103/PhysRev.140.A1133
A. D. Becke, J. Chem. Phys. 98, 5648 (1993). https://doi.org/10.1063/1.464913
M. G. Chegerev, A. A. Starikova, A. G. Starikov, et al., Mendeleev Commun. 32, 49 (2022). https://doi.org/10.1016/j.mencom.2022.01.015
L. Noodleman, J. Chem. Phys. 74, 5737 (1981). https://doi.org/10.1063/1.440939
M. Shoji, K. Koizumi, Y. Kitagawa, et al., Chem. Phys. Lett. 432, 343 (2006). https://doi.org/10.1016/j.cplett.2006.10.023
http://www.chemcraftprog.com
K. Ley and E. Müller, Chem. Ber. 89, 1402 (1956). https://doi.org/10.1002/cber.19560890607
S. N. Lyubchenko, E. P. Ivakhnenko, T. A. Ryskina, et al., Zh. Obshch. Khim. 59, 1104 (1989).
E. Evangelio and D. Ruiz-Molina, Eur. J. Inorg. Chem. 15, 2957 (2005). https://doi.org/10.1002/ejic.200500323
T. Tezgerevska, K. G. Alley, and C. Boskovic, Coord. Chem. Rev. 268, 23 (2014). https://doi.org/10.1016/j.ccr.2014.01.014
V. I. Minkin and A. G. Starikov, Russ. Chem. Bull. 64, 475 (2015). https://doi.org/10.1007/s11172-015-0891-9
I. V. Ershova, A. V. Piskunov, and V. K. Cherkasov, Russ. Chem. Rev. 89, 1157 (2020). https://doi.org/10.1070/RCR4957
V. I. Ovcharenko and O. V. Kuznetsova, Russ. Chem. Rev. 89, 1261 (2020). https://doi.org/10.1070/RCR4981
I. L. Fedushkin, O. V. Maslova, E. V. Baranov, et al., Inorg. Chem. 48, 2355 (2020). https://doi.org/10.1021/ic900022s
I. Fedushkin, O. Maslova, A. Morozov, et al., Angew. Chem., Int. Ed. 51, 10584 (2012). https://doi.org/10.1002/anie.201204452
M. G. Chegerev, A. A. Starikova, A. V. Piskunov, et al., Eur. J. Inorg. Chem. 2, 252 (2016). https://doi.org/10.1002/ejic.201501155
M. G. Chegerev, A. V. Piskunov, A. A. Starikova, et al., Eur. J. Inorg. Chem. 9, 1087 (2018).
M. G. Chegerev and A. V. Piskunov, Russ. J. Coord. Chem. 44, 258 (2018). https://doi.org/10.1134/S1070328418040036
P. Chaudhuri, M. Hess, K. Hildenbrand, et al., Inorg. Chem. 38, 2781 (1999). https://doi.org/10.1021/ic990003g
A. G. Starikov, V. I. Minkin, R. M. Minyaev, et al., J. Phys. Chem. A 114, 7780 (2010). https://doi.org/10.1021/jp101353m
G. M. Coppinger, Tetrahedron 18, 61 (1962). https://doi.org/10.1016/0040-4020(62)80024-6
E. P. Ivakhnenko, Zh. Org. Khim. 19, 886 (1983).
A. I. Prokof’ev, S. P. Solodovnikov, G. A. Nikiforov, et al., Russ. Chem. Bull. 2, 324 (1971).
E. P. Ivakhnenko, P. A. Knyazev, Yu. G. Vitkovskaya, et al., Eur. J. Inorg. Chem. 21, 2055 (2021). https://doi.org/10.1002/ejic.202100184
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation (the Government assignment in the field of scientific activity, project No. FENW-2023-0017).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Ivakhnenko, E.P., Vitkovskaya, Y.G., Lysenko, K.A. et al. Zinc(II) Chelate Complexes with Redox-Active o-Indophenols: Synthesis and Structure. Russ. J. Inorg. Chem. 68, 1159–1168 (2023). https://doi.org/10.1134/S0036023623700304
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023623700304