Abstract
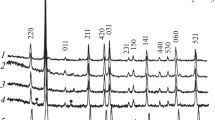
Cs2O–Al2O3 ceramic samples containing 20 and 33 mol % cesium oxide were prepared by сeramic technique and by the glycine–nitrate combustion process. The prepared samples were identified and characterized by X-ray powder diffraction and X-ray fluorescence analyses, scanning electron microscopy, and differential thermal analysis. X-ray powder diffraction and scanning electron microscopy showed that the phase composition and surface of the samples change significantly and nonmonotonically depending on the synthetic method used and the heat treatment parameters of the batch. Optimal synthetic conditions and heat treatment parameters for preparing Cs2O–Al2O3 samples were elucidated.









Similar content being viewed by others
REFERENCES
R. Prins, J. Catal. 392, 336 (2020). https://doi.org/10.1016/j.jcat.2020.10.010
G. Busca, Prog. Mater. Sci. 104, 215 (2019). https://doi.org/10.1016/j.pmatsci.2019.04.003
C. Meephoka, C. Chaisuk, P. Samparnpiboon, and P. Praserthdam, Catal. Commun. 9, 546 (2008). https://doi.org/10.3390/cryst11060690
P. S. Shreyas, B. P. Mahesh, S. Rajanna, and N. Rajesh, Mat. Tood. Proc. 45, 429 (2021). https://doi.org/10.1016/j.matpr.2020.12.1012
L. I. Podzorova, A. A. Ilyicheva, O. I. Penkova, O. S. Antonova, A. S. Baikin, and A. A. Konovalov, Inorg. Mater. 55, 671 (2019). https://doi.org/10.1134/S0020168519060128
W. Chaitree, S. Jiemsirilers, O. Mekasuwandumrong, et al., Catal. Today 164, 302 (2011). https://doi.org/10.1016/j.cattod.2010.11.004
S. V. Tsybulya and G. N. Kryukova, Phys. Rev. B 77, 024112 (2008). https://doi.org/10.1103/PhysRevB.77.024112
G. Paglia, C. E. Buckley, A. L. Rohl, et al., Phys. Rev. B 68, 144110 (2003). https://doi.org/10.1103/PhysRevB.68.144110
M. Rudolph, M. Motylenko, and D. Rafaja, IUCrJ 6, 116 (2019). https://doi.org/10.1107/S2052252518015786
B. Marí, K. C. Singh, M. Moya, et al., Opt. Quant. Electron. 47, 1569 (2015). https://doi.org/10.1007/s11082-014-9997-9
M. N. Saeed Adel, Q. A. Al-Gunaid Murad, N. K. Subramani, et al., Pol.-Plast. Tech. Eng. 57, 1188 (2018). https://doi.org/10.1080/03602559.2017.1373402
P. F. McMillan, A. Grzechnik, and H. Chotalla, J. Non-Cryst. Solids 226, 239 (1998). https://doi.org/10.1016/S0022-3093(98)00416-5
K. Fukumi, S. Sakka, and T. Kokubo, J. Non-Cryst. Solids 93, 190 (1987). https://doi.org/10.1016/S0022-3093(87)80038-8
N. Macleod, J. M. Keel, and R. M. Lambert, Catal. Lett. 86, 51 (2003). https://doi.org/10.1023/A:1022602807322
A. A. Ansari, M. A. M. Khan, M. N. Khan, and S. A. Alrokayan, J. Semicond. 32, 1 (2011). https://doi.org/10.1088/1674-4926/32/4/043001
C. Guéneau and J. L. Fleche, CALPHAD: Comput. Coupling Phase Diagrams Thermochem. 49, 67 (2015). https://doi.org/10.1016/j.calphad.2015.02.002
V. L. Stolyarova, V. A. Vorozhtcov, S. I. Lopatin, et al., Rapid Commun. Mass Spectrom. 35, e8829 (2021).https://doi.org/10.1002/rcm.9079
V. L. Stolyarova, V. A. Vorozhtcov, S. I. Lopatin, et al., Rapid Commun. Mass Spectrom. 35, e9097 (2021). https://doi.org/10.1002/rcm.9097
O. S. Kaimieva, I. E. Sabirova, E. S. Buyanova, and S. A. Petrova, Russ. J. Inorg. Chem. 67, 1211 (2022). https://doi.org/10.1134/S0036023622090054
A. E. Medveeva, E. V. Makhonina, L. S. Pechen, et al., Russ. J. Inorg. Chem. 67, 952 (2022). https://doi.org/10.1134/S0036023622070154
E. V. Babaev, Russ. J. Gen. Chem. 80, 2590 (2010). https://doi.org/10.1134/S1070363210120261
M. J. O’Donnell, C. Zhou, and W. L. Scott, J. Am. Chem. Soc. 118, 6070 (1996). https://doi.org/10.1021/ja9601245
T. L. Simonenko, N. P. Simonenko, E. P. Simonenko, et al., Russ. J. Inorg. Chem. 67, 1495 (2022). https://doi.org/10.1134/S0036023622600939
O. B. Tomilin, E. E. Muryumin, M. V. Fadin, et al., Russ. J. Inorg. Chem. 67, 431 (2022). https://doi.org/10.1134/S0036023622040192
J. Wang, H. Zhao, and Y. Wen, Electrochim. Acta 113, 679 (2013). https://doi.org/10.1016/j.electacta.2013.09.086
V. D. Zhuravlev, V. G. Vasil’ev, E. V. Vladimirova, et al., Glass Phys. Chem. 36, 506 (2010). https://doi.org/10.1134/S1087659610040164
F. Cardarelli, Materials Handbook (Springer-Verlag, London, 2008).
R.-S. Zhou and R. Snyder, Acta Crystallogr., Sect. B: Struct. Sci. 47, 617 (1991). https://doi.org/10.1107/S0108768191002719
G. Langlet, C. R. Acad. Sci. 259, 3769 (1964).
ACKNOWLEDGMENTS
The authors express their gratitude to the staff of the resource centers “X-ray Diffraction Methods of Investigation”, “Interdisciplinary Resource Centre for Nanotechnology”, “Innovative Technologies of Composite Nanomaterials”, and “Geomodel” of the science park of St. Petersburg State University.
Funding
This work was supported by Ministry of Education and Science of the Russian Federation (project No. 075-15-2021-1383).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest to disclose here.
Additional information
In commemoration of the 300th anniversary of St. Petersburg State University’s founding
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Fedorova, A.V., Stolyarov, V.A., Pavelina, M.E. et al. Ceramics of the Cs2O–Al2O3 System Prepared by Solid-Phase Technology and the Glycine–Nitrate Combustion Process. Russ. J. Inorg. Chem. 68, 911–922 (2023). https://doi.org/10.1134/S0036023623600909
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023623600909