Abstract
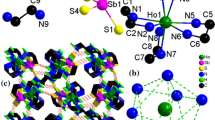
Solvate polymorphs of [Dy(acac)3(H2O)2] · Thf, [Ln(acac)3(H2O)2] · H2O · EtOH (Ln = Eu, Dy) and [Dy(acac)3(H2O)2] · 1.5MeOH, which belongs to a new structural type, are isolated as single crystals. The structures of the prepared compounds are determined by X-ray crystallography, and the effect of solvate molecules on their crystal structures is discussed.
Similar content being viewed by others
References
A. M. Kaczmarek, Y.-Y. Liu, Ch. Wang, et al., Adv. Funct. Mater. 27 (2017).
P. S. Koroteev, Z. V. Dobrokhotova, A. B. Ilyukhin, et al., J. Chem. Soc., Dalton Trans. 45, 6405 (2016).
S. Fukuzawa, K. Metoki, and S. Esumi, Tetrahedron 59, 10445 (2003).
A. J. Roche, S. A. Rabinowitz, and K. A. Cox, Tetrahedron: Asymm. 24, 1382 (2013).
S. P. Babailov, Sens. Actuators B 233, 476 (2016).
A. V. Gavrikov, P. S. Koroteev, Z. V. Dobrokhotova, et al., Polyhedron 102, 48 (2015).
A. V. Gavrikov, P. S. Koroteev, N. N. Efimov, et al., Dalton Trans. 46, 3369 (2017).
A. V. Gavrikov, N. N. Efimov, Zh. V. Dobrokhotova, et al., Dalton Trans. 46, 11806 (2017).
N. N. Efimov, P. S. Koroteev, A. V. Gavrikov, et al., Magnetochemistry 2 (2016).
W.-B. Sun, B. Yan, L.-H. Jia, et al., Dalton Trans. 45, 8790 (2016).
Sh. Zhang, H. Ke, L. Sun, et al., Inorg. Chem. 55, 3865 (2016).
S.-D. Jiang, B.-W. Wang, G. Su, et al., Angew. Chem., Int. Ed. 49, 7448 (2010).
APEX2 and SAINT (Bruker, 2007).
G. M. Sheldrick, SADABS (Univ. of Gottingen, Gottingen, 1997).
G. M. Sheldrick, Acta Crystallogr., Sect. C 71, 3 (2015).
J. W. Bunting, J. P. Kanter, R. Nelander, et al., Can. J. Chem. 73, 1305 (1995).
L. I. Martynenko, I. A. Murav’eva, N. K. Khalmurzaev, and V. I. Spitsyn, Structure, Properties, and Applications of Metal [beta]-Diketonates, Ed. by V. I. Spitsyn (Nauka, Moscow, 1978) [in Russian].
C. R. Groom and F. H. Allen, Angew. Chem., Int. Ed. 53, 662 (2014).
A. S. Batsanov, Yu. T. Struchkov, G. V. Trembovetskii, et al., Dokl. Akad. Nauk SSSR 289, 903 (1986).
J. A. Cunningham, D. E. Sands, W. F. Wagner, and M. F. Richardson, Inorg. Chem. 8, 22 (1969).
E. D. Watkins, J. A. Cunningham, T. Phillips, et al., Inorg. Chem. 8, 29 (1969).
H.-Q. Ye, Y. Peng, Z. Li, et al., J. Phys. Chem. C 117, 23970 (2013).
L. A. Aslanov, E. F. Korytnyi, and M. A. Porai- Koshits, Zh. Strukt. Khim. 12, 661 (1971).
T. Okawara, K. Ishihama, and K. Takehara, Acta Crystallogr., Sect. E 70, m258 (2014).
L. A. Aslanov, M. O. Dekaprilevich, M. A. Porai-Koshits, and V. I. Ivanov, Zh. Strukt. Khim. 8, 1106 (1967).
L. A. Aslanov, M. A. Porai-Koshits, and M. O. Dekaprilevich, Zh. Strukt. Khim. 12, 470 (1971).
S. Cheng, F. Yuguo, W. Yutian, et al., Jilin Daxue Ziran Kex. Xue. (Chin.) (Acta Sci. Nat. Univ. Jil.) 103 (1983).
L. I. Martynenko, S. A. Burova, and A. P. Pisarevskii, Koord. Khim. 21, 424 (1995).
A. L. Il’inskii, V. I. Ivanov, A. D. Khalilov, et al., Zh. Strukt. Khim. 10, 285 (1969).
A. B. Ilyukhin and A. V. Gavrikov, Private Communication. CCDC 1421242 (2015).
S. Cheng, F. Yuguo, and L. Guofa, Gaodeng Xuexiao Huaxue Xuebao (Chem. J. Chin. Univ.) 4, 769 (1983).
H. Kooijman, F. Nijsen, A. L. Spek, and F. Schip, Acta Crystallogr., Sect. 56, 156 (2000).
J. A. Cunningham, D. E. Sands, and W. F. Wagner, Inorg. Chem. 6, 499 (1967).
K. Semenenko, J. Agric. Food Chem. 22, 36 (1969).
H. Kooijman, A. L. Spek, W. Bult, and F. Nijsen, Private Communication. CCDC 1443363 (2015).
I. O. Dzyubenko, Koord. Khim. 17, 1714 (1991).
N. Y. Zharkova, S. I. Troyanov, L. I. Martynenko, et al., Russ. J. Coord. Chem. 24, 55 (1998).
F. Bai, H. Su, and F. Chang, Chem. Lett. 36, 1104 (2007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.B. Ilyukhin, A.V. Gavrikov, Zh.V. Dobrokhotova, V.M. Novotortsev, 2018, published in Zhurnal Neorganicheskoi Khimii, 2018, Vol. 63, No. 9, pp. 1161–1166.
Rights and permissions
About this article
Cite this article
Ilyukhin, A.B., Gavrikov, A.V., Dobrokhotova, Z.V. et al. New Solvate Polymorphs of Lanthanide Trisacetylacetonates: Crystal Structures of [Ln(acac)3(H2O)2] · Solv (Ln = Eu, Dy; Solv = Thf, H2O + EtOH, MeOH). Russ. J. Inorg. Chem. 63, 1186–1191 (2018). https://doi.org/10.1134/S003602361809005X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602361809005X