Abstract
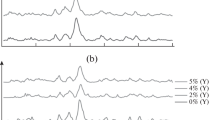
The effect of small amounts of iron(III) ions on the morphology, phase composition, and structure of the products of the hydroxyapatite (HAP) synthesis has been studied by electron microscopy, X-ray powder diffraction, and Mossbauer spectroscopy methods. It has been demonstrated that the introduction of dopant iron(III) ions into the reaction mixture at different stages of HAP formation makes it possible to control crystal growth, morphology, and phase composition. The iron ions are not incorporated into the HAP crystal structure; rather, they form their proper nanophase, as well as adsorption clusters on the HAP surface.
Similar content being viewed by others
References
S. M. Barinov and V. S. Komlev, Bioceramics Based on Calcium Phosphates (Nauka, Moscow, 2005), p. 10 [in Russian].
M. Bohner, Injury 31, D37 (2000).
M. Vallet-Regi, J. Chem. Soc., Dalton Trans., 97 (2001).
D. C. Carvalho, L. G. Pinheiro, A. Campos, et al., Appl. Catal., A: Gen., 471, 39 (2014).
Zhenping Qu, Yahui Sun, Dan Chen, and Yi Wang, J. Mol. Catal. A: Chem. 393, 182 (2014).
M. Pogosova, D. Provotorov, A. Eliseev, et al., Dyes Pigments, 113, 96 (2015).
G. Salviulo, M. Bettinelli, U. Russo, et al., J. Mater. Sci., 46, 910 (2011).
Chun-Han Hou, Sheng-Mou Hou, Yu-Sheng Hsueh, et al. Biomaterials 30, 3956 (2009).
A. Tampieri, T. D’Alessandro, M. Sandri, et al., Acta Biomater., 8, 843 (2012).
Kunfeng Zhao, Botao Qiao, Junhu Wang, et al., Chem. Commun., 47, 1779 (2012).
H. R. Low, N. Phonthammachai, A. Maignan, et al., Inorg. Chem., 47, 11774 (2008).
I. Mayer, H. Diab, and I. Felner, J. Inorg. Biochem., 129 (1992).
I. V. Melikhov, V. F. Komarov, A. V. Severin, et al., Dokl. Phys. Chem., 373, 355 (2000).
P. W. Brown, J. Am. Ceram. Soc. 75, 17 (1992).
Unified Water Analysis Methods, Ed. by Yu. Yu. Lur’e (Khimiya, Moscow, 1973) [in Russian].
S. S. Gorelik, Yu. A. Skakov, and L. N. Rastorguev, X-ray Powder and Electron-Optical Analysis (MISIS, Moscow, 2002) [in Russian].
N. C. Collier, N. B. Milestone, J. Hill, et al., Waste Manage. 26 (2006).
S. Scaccia, M. Carewska, A. D. Bartolomeo, et al., Thermochim. Acta, 383, 145 (2002).
J. O. Nriagu, Geochim. Cosmochim. Acta 36, 459 (1972).
B. M. Al-Hasni, G. Mountjoy, and E. Barney, J. Non-Cryst. Solids 380, 141 (2013).
A. S. Posner, N. C. Blumenthal, and F. Betts, in Phosphate Minerals, Ed. by G. O. Nriagu and P. B. Moore (Springer, Berlin, 1984).
J. M. Hughes, M. Cameron, and K. D. Crowley, Am. Mineral. 74, 870 (1989).
ASTM-41-224, 41-225.
J. Herrero, O. Artieda, and W. H. Hudnall, Soil Sci. Soc. Am. J. 73, 1757 (2009).
N. Prieto-Taboada, O. Gómez-Laserna, I. Martínez-Arkarazo, et al., Anal. Chem., 86, 10131 (2014).
H. Weiss and M. F. Bräu, Angew. Chem., Int. Ed. Engl. 48, 3520 (2009).
A. Y. Polyakov, A. E. Goldt, T. A. Sorkina, et al., Cryst. Eng. Commun., 14, 8097 (2012).
C. Díaz-Aguila, M. Morales, E. Baggio-Saitovitch, et al., III Congreso Internacional de Biomateriales BIOMAT’03, 2003.
E. I. Suvorova, V. V. Klechkovskaya, V. F. Komarov, et al., Crystallogr. Rep. 51, 881.
D. A. Pankratov, Inorg. Mater. 50, 82 (2014).
D. A. Pankratov, A. A. Veligzhanin and Y. V. Zubavichus, Russ. J. Inorg. Chem. 58, 67 (2013).
D. A. Pankratov and Y. M. Kiselev, Russ. J. Inorg. Chem. 54, 1451 (2009).
M. D. Dyar, E. R. Jawin, E. Breves, et al., Am. Mineral., 99, 914 (2014).
J. Mingzhi, C. Xianhao, X. Weiming, et al., Hyperfine Interact., 41, 645 (1988).
M. M. Gadgil and S. K. Kulshreshtha, J. Solid State Chem. 111, 357 (1994).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Severin, D.A. Pankratov, 2016, published in Zhurnal Neorganicheskoi Khimii, 2016, Vol. 61, No. 3, pp. 279-287.
Rights and permissions
About this article
Cite this article
Severin, A.V., Pankratov, D.A. Synthesis of nanohydroxyapatite in the presence of iron(III) ions. Russ. J. Inorg. Chem. 61, 265–272 (2016). https://doi.org/10.1134/S0036023616030190
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023616030190