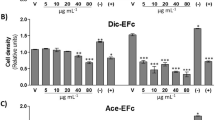
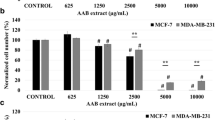
Abstract
Medicinal plant extracts have recently attracted attention of modern medical science research due to their non-lethal activity. Currently, up to 50% of the world drugs including chemotherapeutic drugs such as taxol and camptothecin are derived from natural products. Euphorbia tirucalli has a long history of usage as traditional medicine in Africa and has been widely used in the treatment of different cancers. In this study, we explore the medical properties of E. tirucalli extracts in breast cancer development. To achieve this, stems of E. tirucalli were dried, crushed and extracted with butanol, hexane or methanol (based on 1 g of dry substance in 10 mL of a solvent). The dried extracts were re-dissolved in DMSO and investigated. Composition of each extract was analyzed using liquid chromatography-mass spectroscopy (LC-MS). Extracts were found to contain different types of secondary metabolites mainly terpenes and flavonoids. Breast cancer cell lines (MCF-7 and MDA-MB 231) were treated with various concentrations of the extracts for up to 48 h. Cell viability, cell cycle, apoptosis and gene expression were analysed. In cells, extracts were found to inhibit cell proliferation in a concentration and cell type dependent manner. Analysis of the cause of antiproliferation revealed that most cells were arrested at the G0/G1 phase by p21 overexpression. In general, most pro-apoptotic genes like Bax and caspase-8 were significantly up-regulated in cells treated with plant extracts. These results suggest that the extracts might induce cell cycle arrest at G0/G1 with p21 attributing to this molecular mechanism.
Similar content being viewed by others
References
Singh Y.N. 1986. Traditional medicine in Fiji: Some herbal folk cures used by Fiji Indians. J. Ethnopharmacol. 15, 57–88.
Igoli J.O., Ogaji O.G., Tor-Anyiin T.A., Igoli N.P. 2005. Traditional medicine practise amongst the Igede people of Nigeria. Part II. Afr. J. Tradit. Complement. Altern. Med. 2, 134–152.
Rizk A.F.M. 1987. The chemical constituents and economic plants of the Euphorbiaceae. Bot. J. Linn. Soc. 94, 293–326.
Sayed M.D. 1980. Traditional medicine in healthcare. J. Ethnopharmacol. 2, 19–22.
van Wyk B.E., De Wet H., van Heerden F.R. 2008. An ethnobotanical survey of medicinal plants in the southeastern Karoo, South Africa. S. Afr. J. Bot. 74, 696–704.
Gurib-Fakim A. 2006. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Mol. Aspects Med. 27, 1–93.
Singh S.B., Barrett J.F. 2006. Empirical antibacterial drug discovery: Foundation in natural products. Biochem. Pharmacol. 71, 1006–1015.
Itokawa H., Morris-Natschke S.L., Akiyam, T., Lee K.H. 2008. Plant-derived natural product research aimed at new drug discovery. J. Nat. Med. 62, 263–280.
Pezzuto J.M., 1997. Plant-derived anticancer agents. Biochem. Pharmacol. 53, 121–133.
Siegel R., Ward E., Brawley O., Jemal A. 2011. Cancer Statistics, 2011. The impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA: Cancer J. Clin. 61, 212–236.
Reddivari L., Vanamala J., Safe S.H., Miller J.C., 2010. The bioactive compounds alpha-chaconine and gallic acid in potato extracts decrease survival and induce apoptosis in LNCaP and PC3 prostate cancer cells. Nutr. Cancer. 62, 601–610.
Idibie C.A., Davids H., Iyuke S.E. 2007. Cytotoxicity of purified cassava linamarin to a selected cancer cell lines. Bioprocess Biosyst. Eng. 30, 261–269.
Kim M.E., Ha T.K., Yoon J.H., Lee J.S. 2014. Myricetin induces cell death of human colon cancer cells via BAX/BCL2-dependent pathway. Anticancer Res. 34, 701–706.
Aboul-Fadl T., Radwan A.A., Attia M.I., Al-Dhfyan A., Abdel-Aziz H.A. 2012. Schiff bases of indoline-2,3- dione (isatin) with potential antiproliferative activity. Chem. Cent. J. 6, 49.
Emirdag-Öztürka S., Karayildirima T., Çapci-Karagözb A., Alankus-Çaliskana O., Özmenc A., Poyrazoglu-Çobanc E. 2014. Synthesis, antimicrobial and cytotoxic activities, and structure–activity relationships of gypsogenin derivatives against human cancer cells. Eur. J. Med. Chem. 82, 565–573.
Devarajan E., Sahin A.A., Chen J.S., Krishnamurthy R.R., Aggarwal N., Brun A., Sapino A., Zhang F., Sharma D., Yang X.H., Tora A.D., Mehta K. 2002. Down-regulation of caspase 3 in breast cancer: a possible mechanism for chemoresistance. Oncogene. 21, 8843–8851.
Vorobiof D.A., Sitas F., Vorobiof G. 2001. Breast cancer incidence in South Africa. J. Clin. Oncol. 19, 125–127.
Gehl J., Boesgaard M., Paaske T., Vittrup Jensen B., Dombernowsky P. 1996. Combined doxorubicin and paclitaxel in advanced breast cancer: Effective and cardiotoxic. Ann. Oncol. 7, 687–693.
Aljabarin K.L., Farraj M., Jabarin B., Feng Z.J. 2014. Antimicrobial susceptibility of Euphorbia tirucalli extracts and the effect of latex on growth and viability of mouse breast cancer cells EMT-6. IOSR J. Dental Med. Sci. 13, 59–63.
Betancur-Galvis L.A., Morales G.E., Forero J.E., Roldan J. 2002. Cytotoxic and antiviral activities of Colombian medicinal plant extracts of the Euphorbia genus. Mem. Inst. Oswaldo Cruz. 97, 541–546.
Miglietta A., Gabriel L., Appendino G., Bocca C. 2002. Biological properties of jatrophane polyesters, new microtubule-interacting agents. Cancer Chemother. Pharm. 51, 67–74.
Spiridonov N.A., Konovalov D.A., Arkhipov V.V. 2005. Cytotoxicity of some Russian ethnomedicinal plants and plant compounds. Phytother. Res. 19, 428–432.
Thafeni M., Sayed Y., Motadi L. 2012. Euphorbia mauritanica and Kedrostis hirtella extracts induces cell death in lung cancer cells. J. Mol. Biol. 39, 10785–10794.
Amirghofran Z., Bahmani M., Azadmehr A., Javidnia K. 2006. Induction of apoptosis in leukemia cell lines by Linum persicum and Euphorbia cheiradenia. J. Cancer. Res. Clin. Oncol. 132, 427–432.
Ke N., Wang X., Xu X., Abassi Y.A. 2010. The xCELLigence system for real-time and label-free monitoring of cell viability. Methods Mol. Biol. 740, 33–43.
Moniri M.R., Young A., Reinheimer K., Rayat J., Dai L.J., Warnock G.L. 2015. Dynamic assessment of cell viability, proliferation and migration using real time cell analyzer system (RTCA). Cytotechnology. 67, 379–386.
Meindl C., Absenger M., Roblegg E., Fröhlich E. 2013. Suitability of cell-based label-free detection for cytotoxicity screening of carbon nanotubes. Biomed. Res. Int. 2013, 564804.
Moela P., Choene M.S., Motadi L.R. 2014. Silencing RBBP6 (Retinoblastoma Binding Protein 6) sensitises breast cancer cells MCF7 to staurosporine and camptothecin- induced cell death. Immunobiology. 219, 593–601.
Liu X., Li Q., Dowdell K., Fischer E.R., Cohen J.I. 2012. Varicella-zoster virus ORF12 protein triggers phosphorylation of ERK1/2 and inhibits apoptosis. J. Virol. 86, 3143–3151.
Szarc vel Szic K., Op De Beeck K., Ratman D., Wouters A., Beck I.M., Declerck K., Heyninck K., Fransen E., Bracke M., De Bosscher K., Lardon F., van Camp G., Vanden Berghe W. 2014. Pharmacological levels of withaferin A (Withania somnifera) trigger clinically relevant anticancer effects specific to triple negative breast cancer cells. PLoS ONE. 9, e87850.
Looi C.Y., Arya A., Cheah F.K., Muharram B., Leong K.H., Mohamad K., Wong W.F., Rai N., Mustafa M.R. 2013. Induction of apoptosis in human breast cancer cells via caspase pathway by vernodalin isolated from Centratherum anthelminticum (L.) seeds. PLoS ONE. 8, e56643.
Fulda S., Debatin K.M. 2006. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene. 25, 4798–4811.
Fulda S. 2009. Tumor resistance to apoptosis. Int. J. Cancer. 124, 511–515.
Shapiro G.I., Harper J.W. 1999. Anticancer drug targets: cell cycle and checkpoint control. J. Clin. Invest. 104, 12.
Jansen B., Wacheck V., Heere-Ress E., Schlagbauer- Wadl H., Hoeller C., Lucas T., Hoermann M., Hollenstein U., Wolff K., Pehamberger H. 2000. Chemosensitisation of malignant melanoma by BCL2 antisense therapy. Lancet. 356, 1728–1733.
Kastan M.B., Onyekwere O., Sidransky D., Vogelstein B., Craig R. 1991. Participation of p53 protein in the cellular response to DNA damage. Cancer Res. 51, 6304–6311.
Agarwal M.L., Agarwal A., Taylor W.R., Stark G.R. 1995. p53 controls both the G2/M and the G1 cell cycle checkpoints and mediates reversible growth arrest in human. J. Cell Biol. 92, 8493–8497.
Choene M.S., Motadi L.R. 2012. Anti-proliferative effects of methanolic extract of Kedrostis foetidissima in breast cancer cell lines. Mol. Biol. 1, 107. doi 10.4172/2168-9547.1000107
Mahassni S.H., Al-Reemi R.M. 2012. Apoptosis and necrosis of human breast cancer cells by an aqueous extract of garden cress (Lepidium sativum) seeds. Saudi J. Biol. Sci. 20, 131–139.
Hsieh Y., Chang C., Wan C., Chen C, Yi-Han Chiu, Y., Leu Y., Peng K. 2013. Euphorbia formosana root extract induces apoptosis by caspase-dependent cell death via Fas and mitochondrial pathway in THP-1 human leukemic cells. Molecules. 18, 1949–1962.
Ujiki M.B., Ding X.Z., Salabat M.R., Bentrem D.J., Golkar L., Milam B., Talamonti M.S., Bell R.H., Iwamura T., Adrian T.E. 2006. Apigenin inhibits pancreatic cancer cell proliferation through G2/M cell cycle arrest. Mol. Cancer. 5, 76.
Krifa M., Alhosin M., Muller C.D., Gies J., Chekir-Ghedira L., Ghedira K., Mély Y., Bronner C., Mousli M. 2013. Limoniastrum guyonianum aqueous gall extract induces apoptosis in human cervical cancer cells involving p16INK4A re-expression related to UHRF1 and DNMT1 down-regulation. J. Exp. Clin. Cancer Res. 32, 30.
Williams G.T., Smith C.A. 1993. Molecular regulation of apoptosis: genetic controls on cell death. Cell. 74, 777–779.
Raisova M., Hossini A.M., Eberle J., Riebeling C., Wieder T., Sturm I., Daniel P.T., Orfanos C.E., Geilen C.C. 2001. The Bax/Bcl-2 ratio determines the susceptibility of human melanoma cells to CD95/Fas-mediated apoptosis. J. Invest. Dermatol. 117, 333–340.
Alshatwi A.A., Hasan T.N., Shafi G., Syed N.A., Al- Assaf A.H., Alamri M.S., Al-Khalifa A.S. 2012. Validation of the antiproliferative effects of organic extracts from the green husk of Juglans regia L. on PC-3 human prostate cancer cells by assessment of apoptosis-related genes. Evid. Based Complement. Alternat. Med. 2012, 103026.
Huppertz B., Frank H.-G., Kaufmann P. 1999. The apoptosis cascade: Morphological and immunohistochemical methods for its visualization. Anat. Embryol. 200, 1–18.
Brauns S.C., Dealtry G., Milne P., Naude R., Van De Venter M. 2005. Caspase-3 activation and induction of PARP cleavage by cyclic dipeptide cyclo(Phe-Pro) in HT-29 cells. Anticancer Res. 25, 4197–4202.
Yaacob N.S., Hamzah N., Kamal N.N.N.M., Abidin S.A.Z., Lai C.S., Navaratnam V., Norazmi M.N. 2010. Anticancer activity of a sub-fraction of dichloromethane extract of Strobilanthes crispus on human breast and prostate cancer cells in vitro. BMC Complement. Altern. Med. 10, 42.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Molekulyarnaya Biologiya, 2016, Vol. 50, No. 1, pp. 115–128.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Choene, M., Motadi, L. Validation of the antiproliferative effects of Euphorbia tirucalli extracts in breast cancer cell lines. Mol Biol 50, 98–110 (2016). https://doi.org/10.1134/S0026893316010040
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893316010040