Abstract—
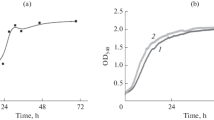
This is the first report on the effect of norepinephrine on biofilms and planktonic cultures of a human skin commensal Kytococcus schroeteri H01. A dose-depending inhibitory effect of norepinephrine on K. schroeteri was shown after 24 and 72 h of incubation on PTFE cubes. The most pronounced effect was observed at 3.5 × 10–7 M norepinephrine after 24 h of incubation (71 ± 13% of the control) and at 3.5 × 10–6 M after 72 h (78 ± 10% of the control). Biofilms formed on glass in the presence of norepinephrine were thinner and flatter than in the controls. No significant effect of norepinephrine on metabolic activity of the cells in biofilms formed on PTFE cubes was revealed. Norepinephrine probably affects initial stages of the switching to biofilm lifestyle and matrix synthesis in K. schroeteri H01.


Similar content being viewed by others
REFERENCES
Becker, K., Schumann, P., Wüllenweber, J., Schulte, M., Weil, H.P., Stackebrandt, E., Peters, G., and von Eiff, C., Kytococcus schroeteri sp. nov., a novel Gram-positive actinobacterium isolated from a human clinical source, Int. J. Syst. Evol. Microbiol., 2002, vol. 52, pp. 1609–1614.
Berridge, M.V., Herst, P.M., and Tan, A.S., Tetrazolium dyes as tools in cell biology: new insights into their cellular reduction, Biotechnol. Annu. Rev., 2005, vol. 11, pp. 127–152.
Borrel, V., Thomas, P., Catovic, C., Racine, P.J., Konto-Ghiorghi, Y., Lefeuvre, L., Duclaroir-Poc, C., Zouboulis, C., and Feuilloley, M.G., Acne and stress: impact of catecholamines on Cutibacterium acnes, Front. Med., 2019, vol. 6, p. 155.
Boukerb, A.M., Robert, M., Teteneva, N.A., Danilova, N.D., Zhurina, M.V., Mart’yanov, S.V., Plakunov, V.K., Feuilloley, M.G., and Gannesen, A.V., Draft genome sequence of Kytococcus schroeteri strain h01, isolated from human skin, Microbiol. Resour. Announc., 2019, vol. 8, art. e01081-19.
Boutcher, S.H., Maw, G.J., and Taylor, N.A., Forehead skin temperature and thermal sensation during exercise in cool and thermoneutral environments, Aviat. Space Environ. Med., 1995, vol. 66, pp. 1058–1066.
Boyanova, L., Stress hormone epinephrine (adrenaline) and norepinephrine (noradrenaline) effects on the anaerobic bacteria, Anaerobe, 2017, vol. 44, pp. 13–19.
Danilova, N.D., Solovyeva, T.V., Mart’yanov, S.V., Zhurina, M.V., and Gannesen, A.V., Stimulatory effect of epinephrine on biofilms of Micrococcus luteus C01, Microbiology (Moscow), 2020, vol. 89, pp. 493–497.
Fournière, M., Latire, T., Souak, D., Feuilloley, M.G., and Bedoux, G., Staphylococcus epidermidis and Cutibacterium acnes: two major sentinels of skin microbiota and the influence of cosmetics, Microorganisms, 2020, vol. 8, art. 1752.
Fresnoza, S., Stiksrud, E., Klinker, F., Liebetanz, D., Paulus, W., Kuo, M.F., and Nitsche, M.A., Dosage-dependent effect of dopamine D2 receptor activation on motor cortex plasticity in humans, J. Neurosci., 2014, vol. 34, pp. 10701–10709.
Gannesen A.V., Borrel V., Lefeuvre L., Netrusov A.I., Plakunov V.K., Feuilloley M.G., Effect of two cosmetic compounds on the growth, biofilm formation activity, and surface properties of acneic strains of Cutibacterium acnes and Staphylococcus aureus, MicrobiologyOpen, 2019, vol. 8, art. e00659.
Lange-Asschenfeldt, B., Marenbach, D., Lang, C., Patzelt, A., Ulrich, M., Maltusch, A., Terhorst, D., Stockfleth, E., Sterry, W., and Lademann, J., Distribution of bacteria in the epidermal layers and hair follicles of the human skin, Skin Pharmacol. Physiol., 2011, vol. 24, pp. 305–311.
Nouioui, I., Carro, L., García-López, M., Meier-Kolthoff, J.P., Woyke, T., Kyrpides, N.C., Pukall, R., Klenk, H.-P., Goodfellow, M., and Goker, R., Genome-based taxonomic classification of the phylum Actinobacteria, Front. Microbiol., 2018, vol. 9, p. 2007.
Nozhevnikova, A.N., Botchkova, E.A., and Plakunov, V.K., Multi-species biofilms in ecology, medicine, and biotechnology, Microbiology (Moscow), 2015, vol. 84, pp. 731–750.
Shah, A.S., Vijayvargiya, P., Jung, S., and Wilson, J.W., Postoperative hardware-related infection from Kytococcus schroeteri: its association with prosthetic material and hematological malignancies—a report of a case and review of existing literature, Case Rep. Infect. Dis., 2019, vol. 2019, art. 6936472.
Tan, I.J., Peeva, E., and Zandman-Goddard, G., Hormonal modulation of the immune system—a spotlight on the role of progestogens, Autoimmun. Rev., 2015, vol. 14, pp. 536–542.
Weigel, W.A. and Demuth, D.R., Qse BC, a two-component bacterial adrenergic receptor and global regulator of virulence in Enterobacteriaceae and Pasteurellaceae, Mol. Oral Microbiol., 2016, vol. 31, pp. 379–397.
Zhurina, M.V., Gannesen, A.V., Mart’yanov, S.V., Teteneva, N.A., Shtratnikova, V.Y., and Plakunov, V.K., Niclosamide as a promising antibiofilm agent, Microbiology (Moscow), 2017, vol. 86, pp. 455–462.
Funding
This work was supported by the Russian Science Foundation (grant no. 19-74-10071). Research conducted by Prof. V. K. Plakunov was sponsored by the Ministry of Science and Higher Education of the Russian Federation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Translated by A. Oleskin
Rights and permissions
About this article
Cite this article
Danilova, N.D., Geraskina, O.V., Diuvenji, E.V. et al. Inhibitory Effect of Norepinephrine on Biofilm Growth of the Human Skin Commensal Kytococcus schroeteri H01. Microbiology 90, 666–669 (2021). https://doi.org/10.1134/S0026261721050039
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261721050039